 |
 |
 |
| |
Acute HCV Treatment/HIV+ MSM/HCV Screening/New HCV Therapies: "Determinants of Response to Pegylated Interferon and Ribavirin for Acute Hepatitis C Infection in Patients with Human Immunodeficiency Virus"
|
| |
| |
Reported by Jules Levin
IDSA 2013 Oct 2-6 San Francisco, CA
Leah Burke, M.D.1, Daniel Fierer, M.D.2, David Cassagnol, M.S.3, Rosanne Hijdra, B.S.4, Wouter Van Seggelen, M.D.4, and Kristen Marks, M.D.5
1New York Presbyterian Hospital - Weill Cornell, New York, NY, 2Mount Sinai School of Medicine, New York, NY, 3Weill Cornell Medical College, New York, NY, 4Amsterdam Medical Center, Amsterdam, Netherlands, 5Weill Medical College of Cornell University, New York, NY
from Jules: NEW THERAPIES & HCV Screening: by this December 2 new therapy regimens are expected to be approved: Janssen's Simeprevir(protease inh)+Peg/Rbv and Gilead's Sofosbuvir+Peg/Rbv for 12 weeks. In 2014 we expect 2 new IFN-free regimens to be approved: Abbvie's ABT450(protease)+ABT267(NS5A)+ABT333(non-nuc polymerase) +/- RBV, and Gilead's Sofosbuvir(nucleotide)+Ledipasvir(NS5A) +/- Rbv. Both these regimens are in large global phase 3 studies now, in phase 2 the Abbvie regimen found 97% SVR rates with 12 weeks therapy, the Gilead regimen 95-100% SVR with 12 weeks therapy. BMS is expected to enter phase 3 with their IFN & Rbv free 3-drug oral regimen of Daclatasvir+Asunaprevir and their non-nuc polymerase inhibitor BMS325, in phase 2 SVR rates were 89-94% with patient discontinuations considered failures. Merck is developing a 2-drug oral regimen with their 2 2nd generation drugs, protease MK5172+MK8472 with early very promising results. Janssen just purchased GSK's potent NS5A inhibitor which they will combine in an IFN-free regimen including their protease Simeprevir & presumably their non-nuc polymerase inh TMC055. Boehringer Ingelheim recently announced preliminary results from an initial study of their 3-drug oral regimen of their protease Faldaprevir + their non-nuc polymerase inh Deleobuvir + the potent Presidio NS5A inhibitor PPI-668: 97% (28/29) of patients achieved rapid virologic response at 4 weeks and 100% of patients (13/13) who have completed all 12 weeks of the investigational regimen had undetectable levels of hepatitis C virus at the end of treatment. Roche will present results soon at AASLD from their ongoing ANNAPURNA Study which looks at their IFN-free regimen which includes their protease Danoprevir, their non-nuc polymerase inh ANA-598 + their nuke Mericitabine with & without Rbv. Clearly HCV cure is around the corner with 12 weeks therapy without Pegylated interferon and for some without Rbv. However, it is estimated as many as 75% of those with HCV have not been tested & remain undiagnosed, so HCV screening programs are a major missing ingredient in trying to give everyone an opportunity to be cured. Another major concern is that many with HCV are considered the "aging HCV population", they have had HCV for 20-30 years, perhaps undiagnosed, and advancing to serious liver disease including advanced cirrhosis and liver cancer, so HCV screening programs are needed to identify these patients so they can receive care and treatment as soon as possible. Unfortunately the federal govt has not made adequate commitments to fund large-scale screening projects. On a State & City level no State or City has committed large-scale funding to screen on a large scale, however in New York state now, sitting on Go Cuomo's desk is a bill passed by the State Legislature to provide screening for "Baby boomers", we hope the governor signs this bill soon & other States follow this first Statewide screening bill.
EASL: New Oral HCV Drugs at EASL - Report 4A
HCV Economics 101 - (10/13/13)
http://www.e-attend.com/Event/DownloadFile?http://1aec793686d3222bb329-999d00328f5ec7bc3f57388404b171c3.r91.cf1.rackcdn.com/mp4/IDWeek13-0207_BurkeLeah.mp4
from Jules;
This is a retrospective cohort of 75 HIV+ patients in NYC examining treatment for acute HCV infection with only peginterferon+ribavirin. Below is a link to a study treating connected acute HCV with triple therapy including telaprevir+Peg/Rbv presented earlier this year in March at CROI. ·Since 2000, there have been reported epidemics of acute HCV in HIV-infected MSM predominantly in Western Europe, Australia, and the USA. This issue has received little attention in the USA, but in July this published study in CID looked at HCV in MSM in the HIV MACS Cohort-----
(MACS Study) Incident Hepatitis C Virus Infection in Men Who Have Sex With Men: A Prospective Cohort Analysis, 1984-2011 'HIV-infection (6-fold), unprotected receptive anal intercourse (3-fold) increase risk for HCV acquisition, low CD4, syphils' - (09/20/13)
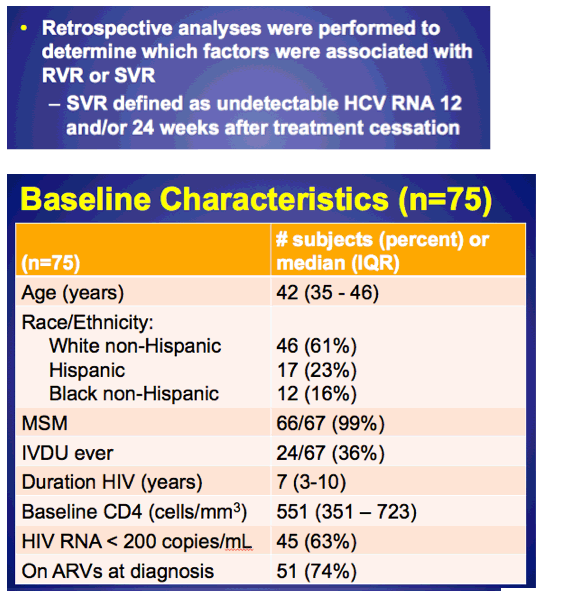
Our study aimed to describe treatment outcomes in a relatively large North American cohort with acute HCV and HIV. From January 2004 - January 2013, 75 HIV-infected males with acute HCV were treated with pIFN + RBV via ongoing NYC cohort studies at Weill Cornell Medical Center and Mount Sinai. Subjects were identified through referral from primary care and HIV physicians, city STD and public health clinics, and community outreach in high-risk populations. Retrospective analyses were performed to determine which factors were associated with RVR or SVR
-SVR defined as undetectable HCV RNA 12 and/or 24 weeks after treatment cessation
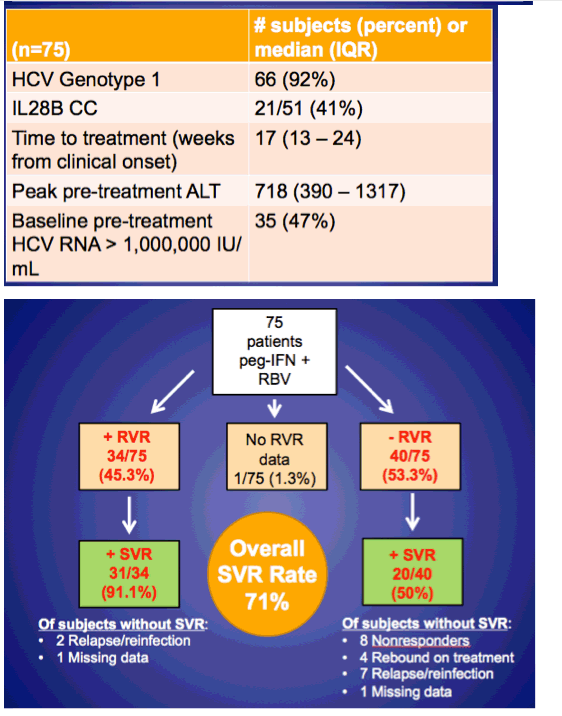
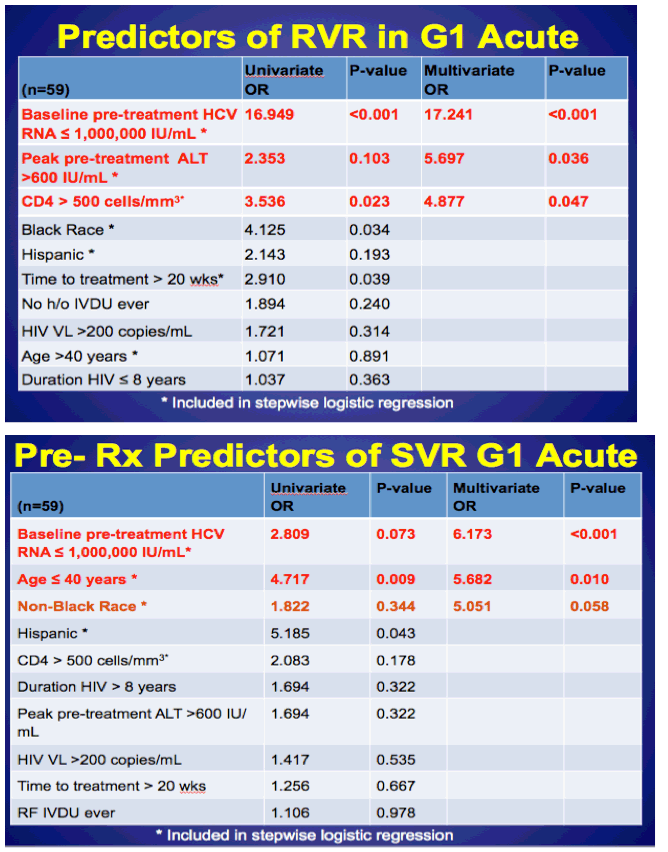
Average age was 42 (35-56). Most were MSM (99%), 61% white, 16% black, 23% hispanic. 36% were identified as IDU ever. Baseline CD4 551, 74% on ART and 63% <200 c/ml. 92% genotype 1. Average time to HCV treatment 17 weeks, peak pre-treatment ALT 718 (390-1317). 45% (34/75) achieved RVR and 91% of these (31/34) achieved SVR, but 53% (40/75) did not achieve RVR, of these 20/40 went on to achieve an SVR, for an overall SVR rate of 71%. In the slides below the authors characterized 9 patients with "relapse/reinfection". The authors found these predictors of RVR in Gt1 acute HCV: baseline pre-treatment HCV RNA <1 million IU/ml (multivariate OR 17.241 p-value <0.001); peak pre-treatment ALT >600 IU/ml (multivariate OR 5.697 p-value 0.036); and CD4 >500 (multivariate OR 4.877, p-value 0.047). see slide, apparently black race was a univariate predictor but not a multivariate predictor. Pre-treatment predictors of SVR for GT1 acute HCV: baseline pre-treatment HCV RNA <1 million IU/ml; age <40, non-black race (see slide). RVR was a predictor of SVR (odds ratio 13.5 p=.003), when included in MV model, age <40 was the only other significant predictor (OR 5.8 p 0.01). The authors summarization included:
Consideration should me made to the addition of a DAA to pIFN + RBV:
1)Prior to initiating treatment if poor prognostic factors
-CD4 ≤500 cells/mm3, peak pre-treatment ALT ≤600 IU/mL, age >40 years, baseline pre-treatment HCV RNA of > 1,000,000 IU/mL, black race
OR
2)On treatment if no RVR
CROI: Treatment of Acute HCV in HIV-infected MSM Incorporating Telaprevir - (03/13/13)









|
| |
|
 |
 |
|
|