 |
 |
 |
| |
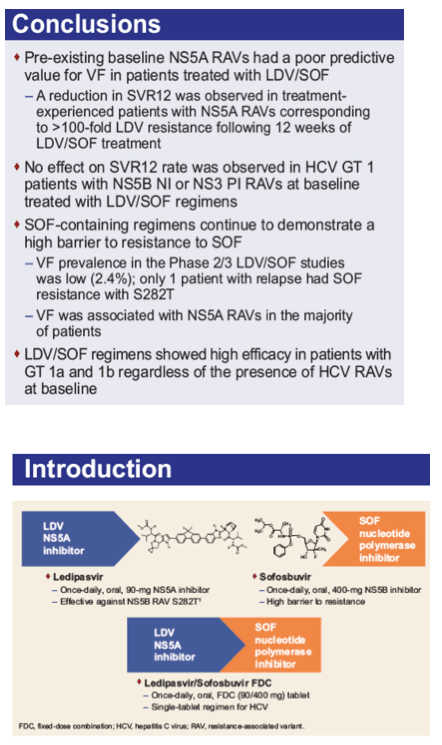
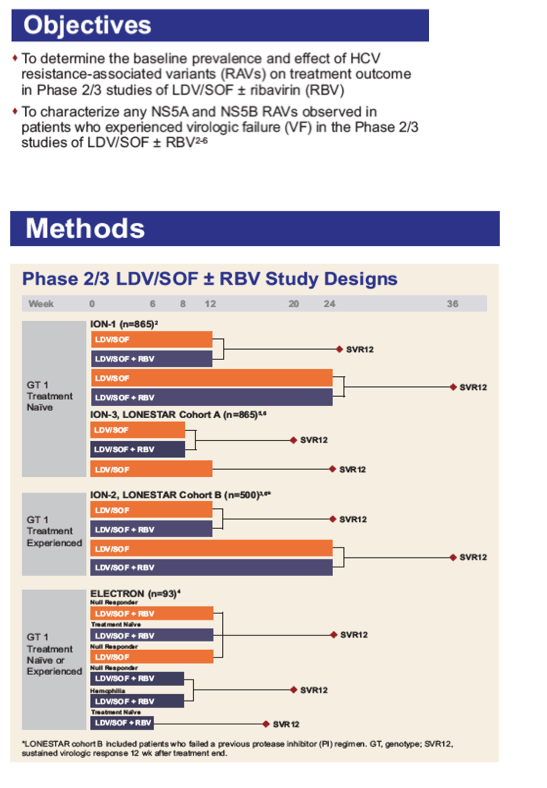
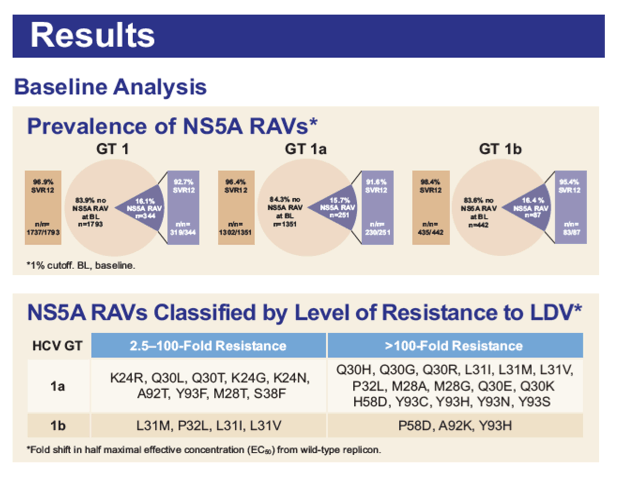
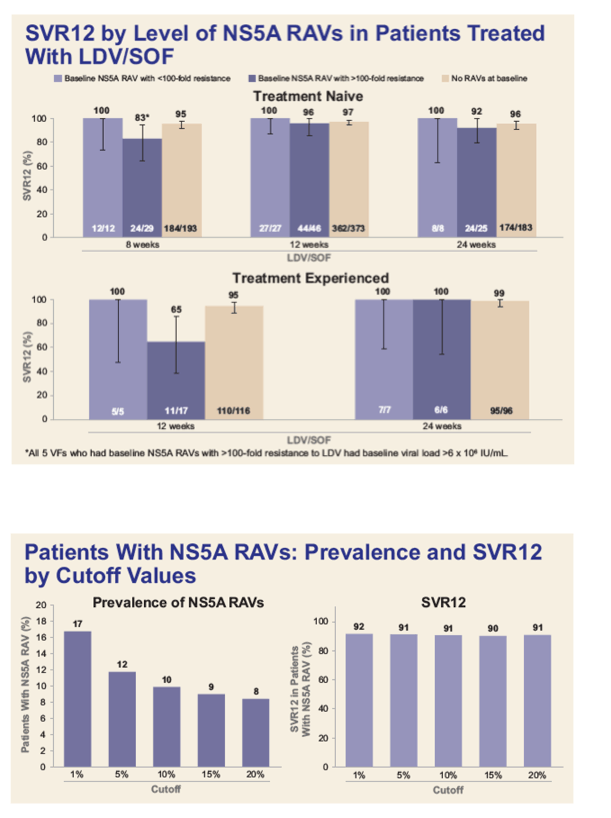
Baseline and Post-Baseline Resistance Analyses of Phase 2/3 Studies of Ledipasvir/Sofosbuvir ± RBV
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
Christoph Sarrazin,1 Hadas Dvory-Sobol,2 Evguenia Svarovskaia,2 Brian Doehle,2 Joseph F. McCarville,2 Phillip S. Pang,2 Nezam Afdhal,3
Kris V. Kowdley,4 Edward J. Gane,5 Eric J. Lawitz,6 John G. McHutchison,2 Michael D. Miller,2 Hongmei Mo2 1Medizinische Klinik
1, Goethe University Hospital, Frankfurt, Germany; 2Gilead Sciences, Inc., Foster City, CA; 3Harvard Medical School, Beth Israel Deaconess Medical Center, Boston, MA; 4Swedish Medical Center, Seattle, WA; 5New Zealand Liver Transplant Unit, Auckland City Hospital, Auckland, New Zealand; 6Texas Liver institute, University of Texas Health Science Center, San Antonio, TX
AASLD: An Integrated Safety and Efficacy Analysis of >500 Patients With Compensated Cirrhosis Treated With Ledipasvir/Sofosbuvir With or Without Ribavirin - (11/11/14)
AASLD: Ledipasvir/Sofosbuvir With Ribavirin for the Treatment of HCV in Patients With Decompensated Cirrhosis: Preliminary Results of a Prospective, Multicenter Study - (11/12/14)
AASLD: Ledipasvir/Sofosbuvir Fixed-Dose Combination Is Safe and Efficacious in Cirrhotic Patients Who Have Previously Failed Protease-Inhibitor Based Triple Therapy SIRIUS Trail - (11/11/14)

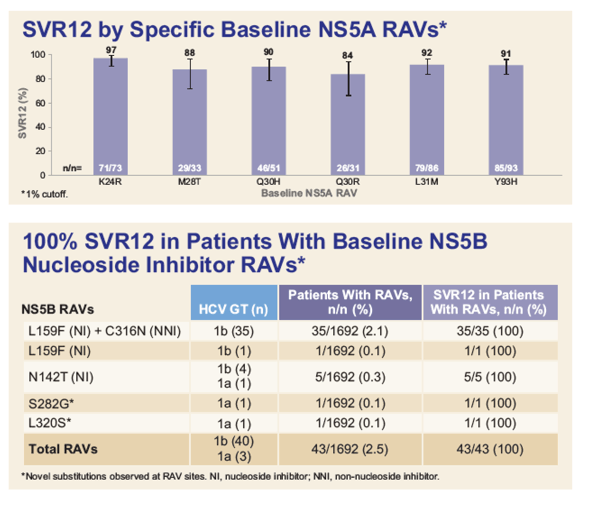
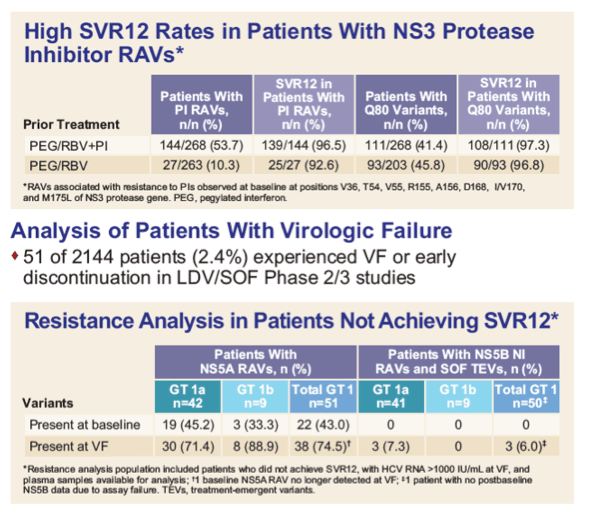
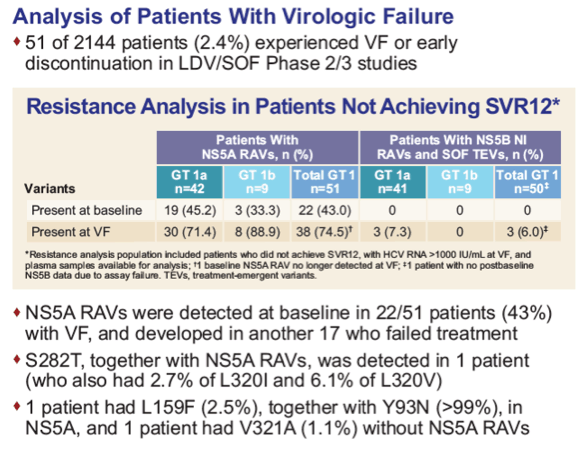
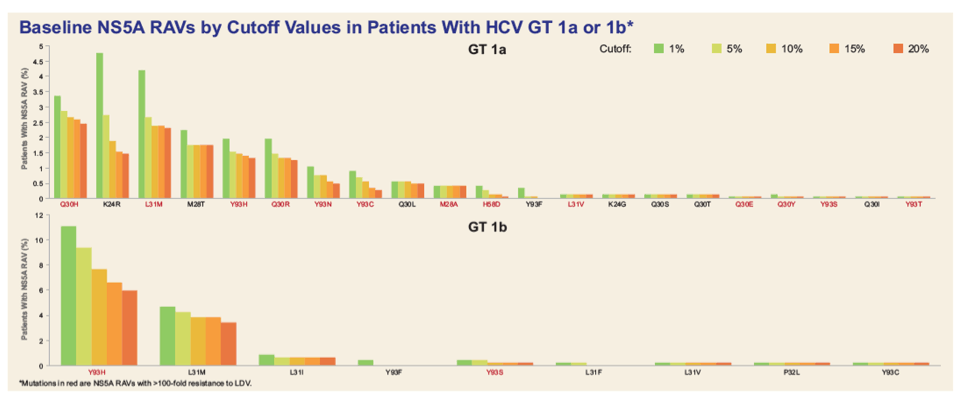
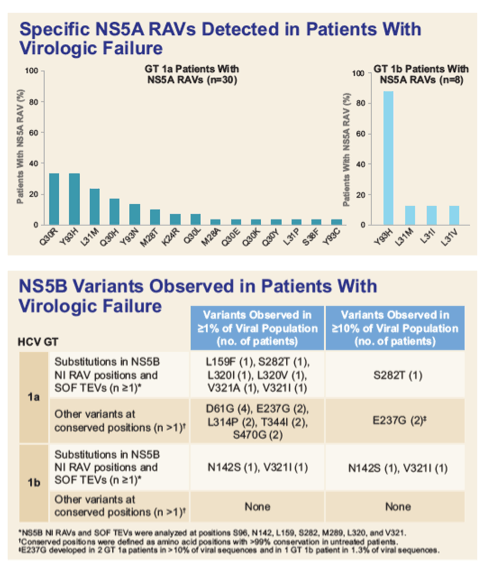
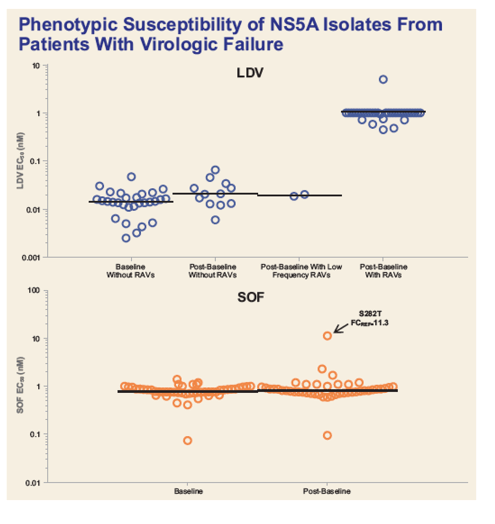



|
| |
|
 |
 |
|
|