 |
 |
 |
| |
Tenofovir Montherapy in Chronic Hepatitis B Patients
with Genotypic Resistance to Previous Antiviral Therapy - a Cohort Study
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston

Program Abstract:
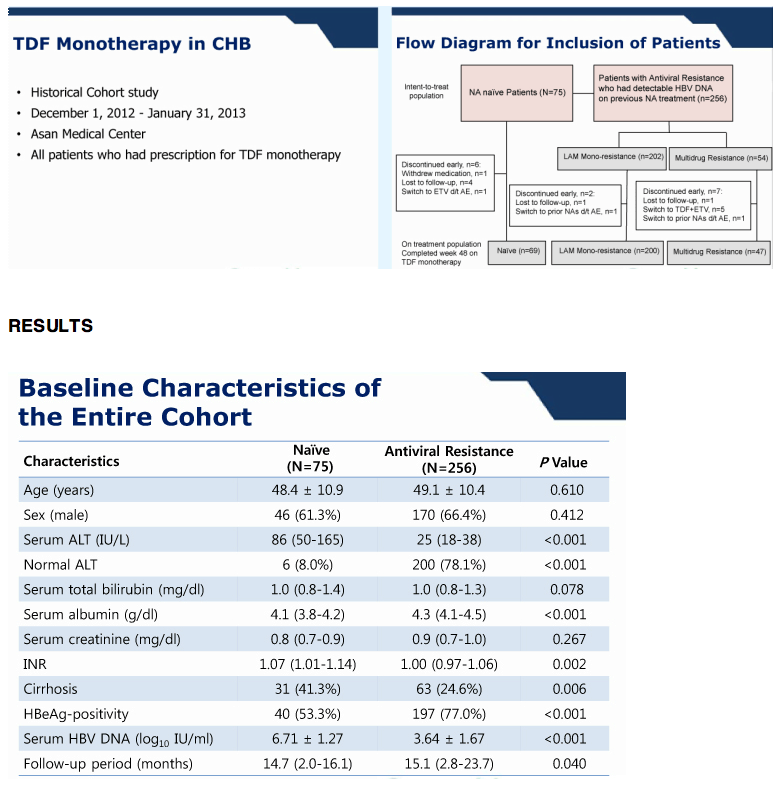
Background/Aims: The antiviral effect of monotherapy with tenofovir disoproxil fumarate (TDF) is controversial for patients with hepatitis B virus (HBV) resistant to nucloes(t)ide analogues. Methods: Patients who were treated with TDF monotherapy for HBV with documented genotypic resistance mutations were included.
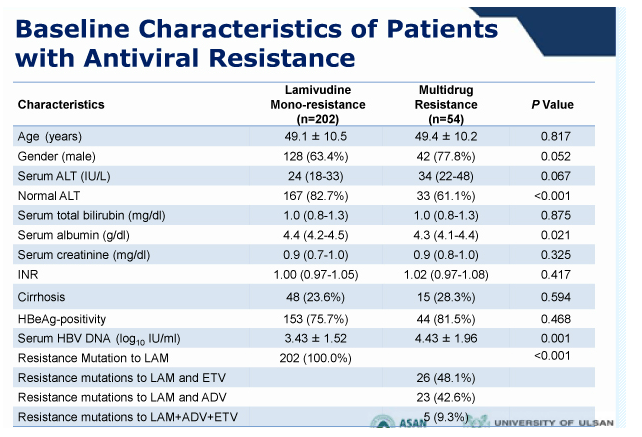
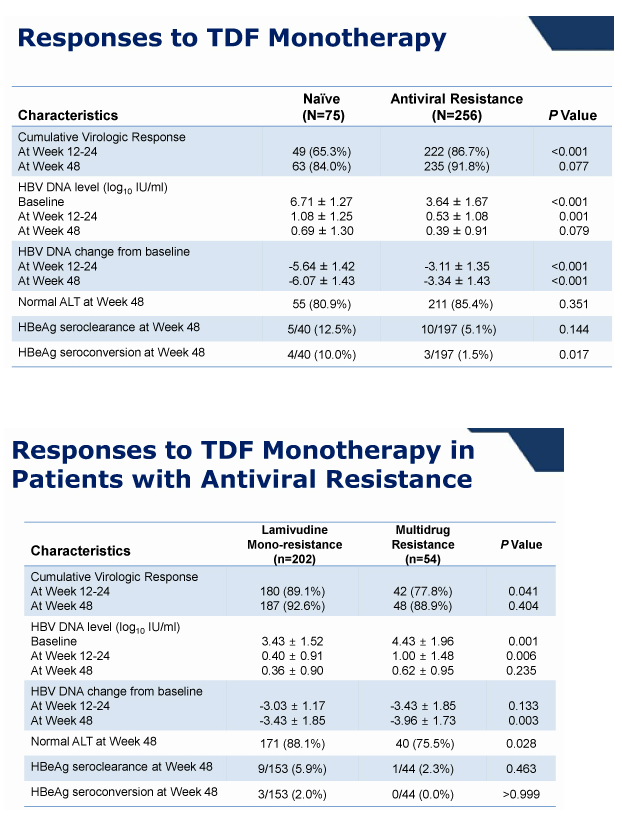
Results: At baseline, 325 (81%) patients had mono-resistance to lamivudine, while others had multi-drug resistance to lamivudine and adefovir (n=43, 10%) or lamivudine and entecavir (n=32, 8%). Most (287, 72%) were being treated with combination therapies including lamivudine, adefovir, and entecavir. Their mean HBV DNA level was 2.5 +/- 2.0 log10 IU/mL.
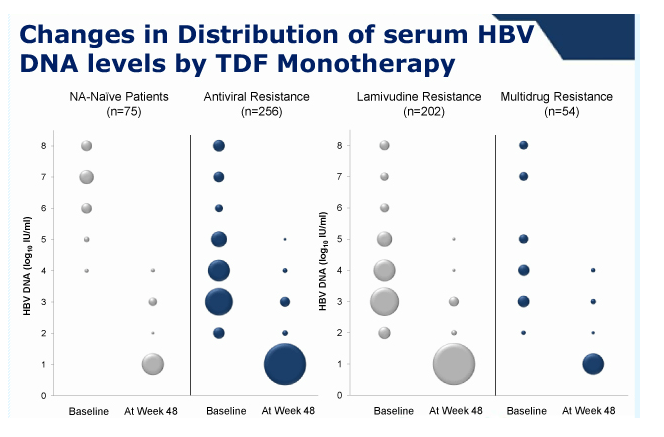
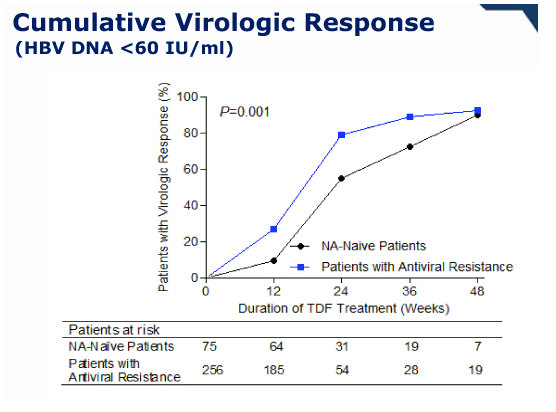
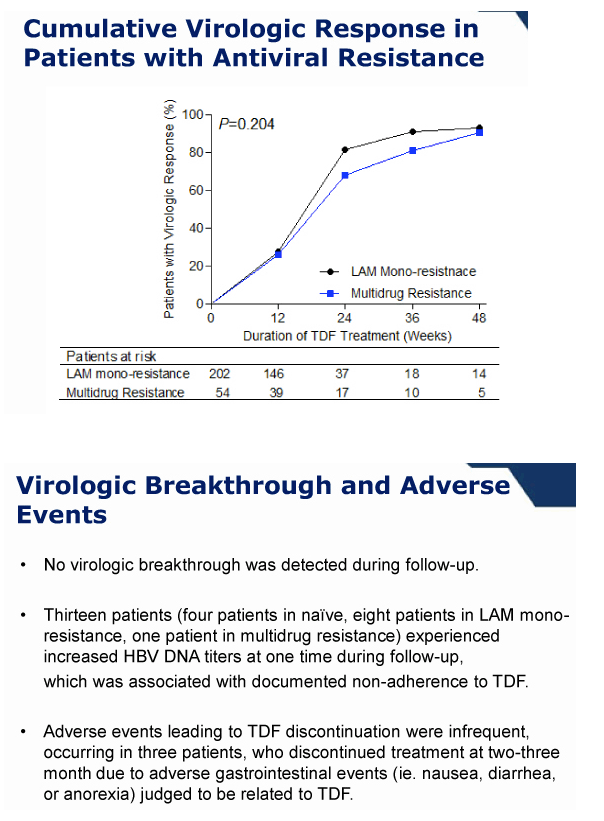
During 1 year of TDF monotherapy, 378 (95%) patients cumulatively achieved virological response (VR, HBV DNA <60 IU/mL) by modified intention-to-treat analysis. The rates of VR were not different between patients with mono-resistance to lamivudine and those with multi-drug resistance (95.2% vs. 92.0%, P =0.27). Sixteen patients who did not achieve VR also showed a significant reduction in their mean HBV DNA levels from baseline (-3.42 log10 IU/mL, P <0.01). Five patients experienced viral breakthrough, and all were related with low adherence to medication. The rate of HBV DNA undetectability was not influenced by the degree of previous resistance mutations (P >0.05). Four patients discontinued TDF because of gastrointestinal symptoms (n=3) or for pregnancy (n=1). Otherwise, no significant clinical or laboratory adverse event was reported.
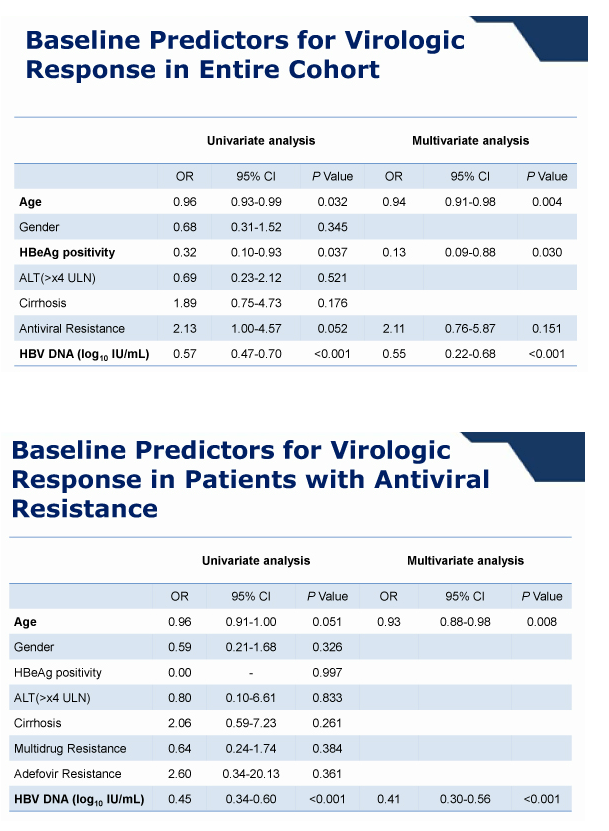
Conclusions: In this cohort study, most patients infected with HBV that has genotypic resistance mutations to lamivudine, adefovir, or entecavir rapidly achieved VR within 48 weeks of TDF monotherapy, regardless of the nature of previous resistance mutations. No patient developed additional resistance mutations.


|
| |
|
 |
 |
|
|