 |
 |
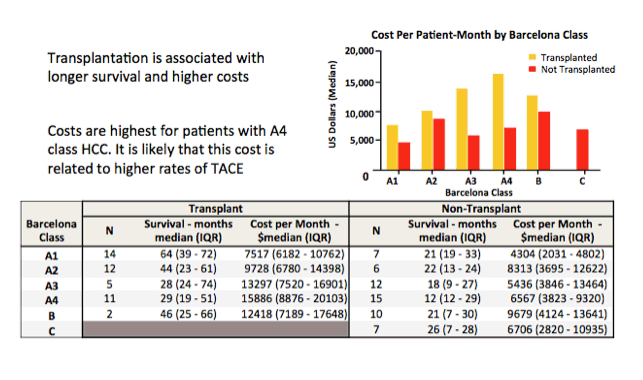
 |
| |
Direct Costs of Care for Hepatocellular Carcinoma in patients with Hepatitis C cirrhosis
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
Andreea M Catana1, Elliot Tapper1, Daniel Mansuri1,Nidhi Sethi1, Annie Vong1, Saurabh Sethi1, Nezam H. Afdhal1
1. Hepatology, Beth Israel Deaconess Medical Center, Boston, MA,United States.
Funded by Gilead Sciences

Program abstract
Hepatitis C is the commonest cause of hepatocellular cancer (HCC) in the US and the incidence is expected to increase further as the HCV population ages and develops more cirrhosis. Management of HCC is very heterogenous with multiple non-surgical and surgical options. The true cost of care of the HCV patient with HCC is unknown. AIMS: To evaluate the total direct health care costs of different approaches to HCC care in HCV patients in a major referral and transplant center.
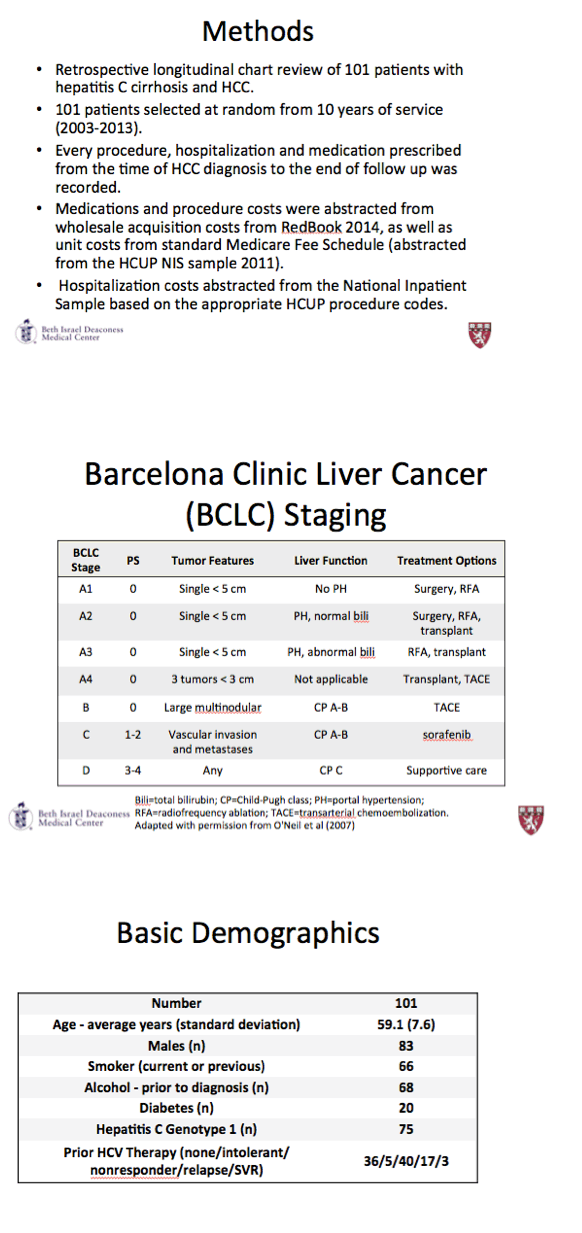
METHODS: 101 patients were randomly selected by computer from a list of all HCC patients with HCV between 2003 and 2013. All patients were biopsy-proven HCC or met UNOS OPTN criteria. Patients were categorized by the primary treatment modality of TACE, Cyberknife radiotherapy, radiofrequency abalation (RFA), chemotherapy or resection. Patients could have multiple treatment modalities and also go on to liver transplant, which is considered as a separate modality for cost determination. The direct cost includes the cost of the procedure, imaging, hospitalizations and all subsequent care of the HCC patient until either death or transplant including cost of HCV treatment and immunosuppression post-transplant. Costs were derived from the Medicare fee schedule abstracted from the HCUP NIS sample 2011. Medication costs used were wholesale acquisition costs (Redbook 2014).
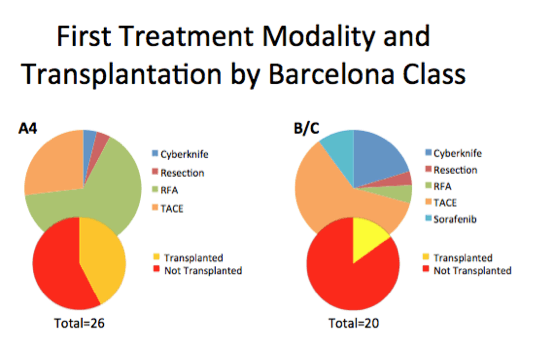
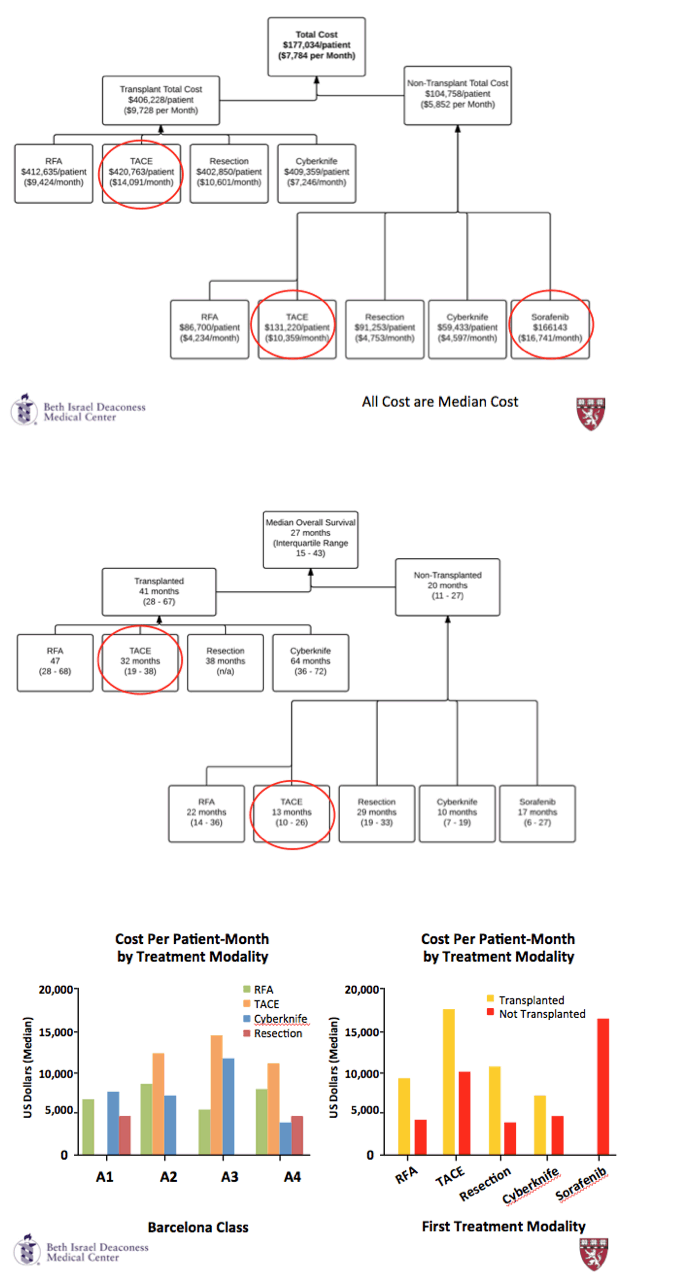
RESULTS: 101 patients, 82 male mean age 59years (range 49-82) were included. All had HCV cirrhosis at diagnosis with a median CTP score of 7 ( range 5-11) and a median MELD of 8. Genotype 1 (74%) and genotype 3 (16%) were predominant. 31 patients were HCV treatment naïve, 65 treatment failures and 4 had had a prior SVR. Majority of HCC were detected through cross-sectional radiological screening programs. Liver staging using the Barcelona score was A1 20%; A2 18%; A3 16% and A4 27%; B 12% and C 7%. Tumor size was mean 2.8cms with a range from 1 - 14cms. Mean follow up was 32 months with a range from 4 - 118 and 37 patients have died. Initial primary treatment modalities were RFA 53%; TACE 26%; Cyberknife 10%, resection 8% and chemotherapy 2%. 43 patients went on to liver transplantation. Calculated overall cost of HCC care for this group of patients was $22,030,108 for a mean cost per patient of $218,120. The 43 patients who underwent transplant accounted for $17,025,037 of the overall costs at $395,000 per transplanted patient compared to $5,817,300 for the non transplant patients for a mean cost of $100,299 per patient.
CONCLUSIONS: Pharmaco-economic studies of HCV treatment need to model real life estimations of true direct cost of HCC care.

Initial primary treatment modalities were RFA 53%; TACE 26%; Cyberknife 10%, resection 8% and chemotherapy 2%.
Initial primary treatment modalities for A1 Barcelona Class were: RFA 60%, Resection 30%, CK 5%, transplant 5% none got TACE or sorafenib
Initial primary treatment modalities for A2 Barcelona Class were: RFA 66,67%, TACE 27,78%, CK 5.56%, none resection or sorafenib or transplant
Initial primary treatment modalities for A3 Barcelona Class were: RFA 62.5%, TACE 18.75%, CK 18.75%, none resection, sorafenib or transplant
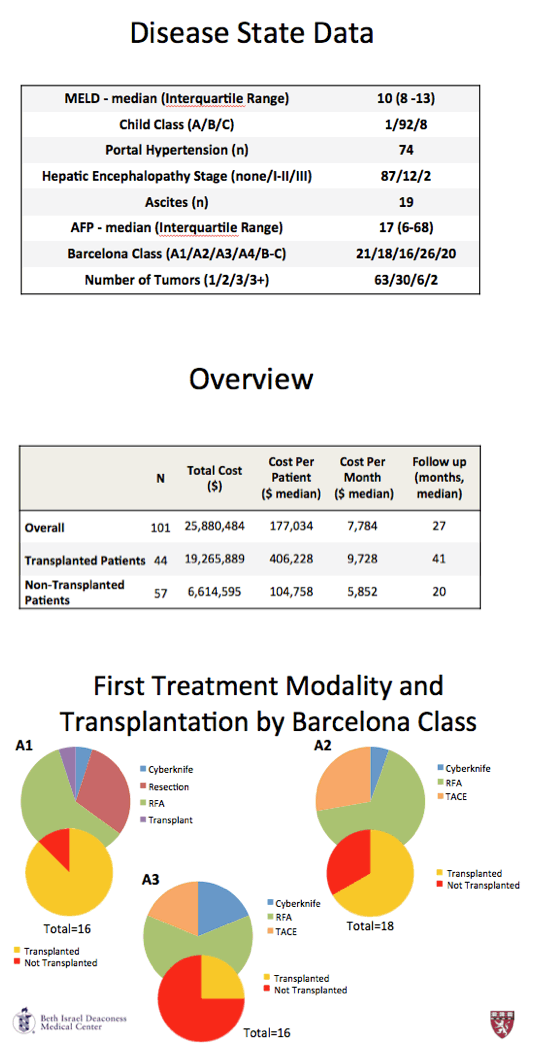
Initial primary treatment modalities for A4 Barcelona Class were: RFA 65.38%, TACE 26.92%, resection 3.85%, CK 3.85%, none transplant or sorafenib
Initial primary treatment modalities for B/C Barcelona Class were: TACE 60%, CK 20%, sorafenib 10%, resection and RFA 5%, none transplant
|
| |
|
 |
 |
|
|