 |
 |
 |
| |
Cost-Effectiveness of Novel Hepatitis C Drug Regimens
Among Treatment-Experienced U.S. Veterans
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
Alexis P. Chidi, S.S. Rogal, C.L. Bryce, M.J. Fine, C.B.
Good, L. Myaskovsky, V.K. Rustgi, A. Tsung, K.J. Smith
University of Pittsburgh & VA Pittsburgh Healthcare System





Program Abstract
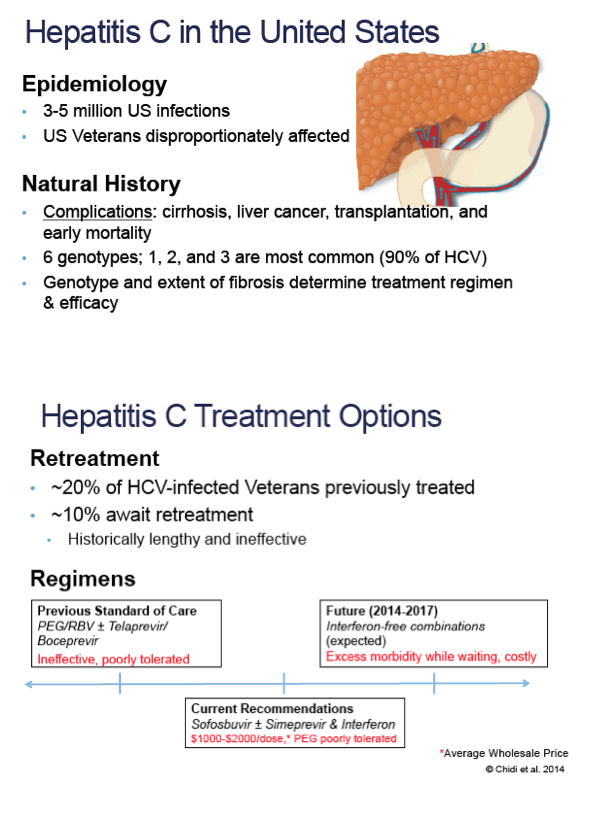
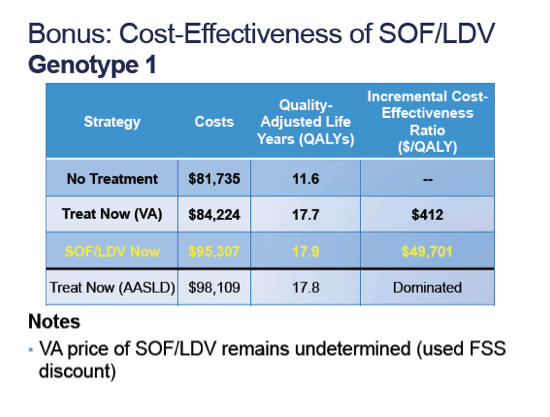
Background: It remains unclear whether treatment-experienced patients (partial- or null-responders) with hepatitis C (HCV) should begin treatment with current sofosbuvir (SOF)-based regimens or wait for all-oral, interferon-free regimens expected in 2015.
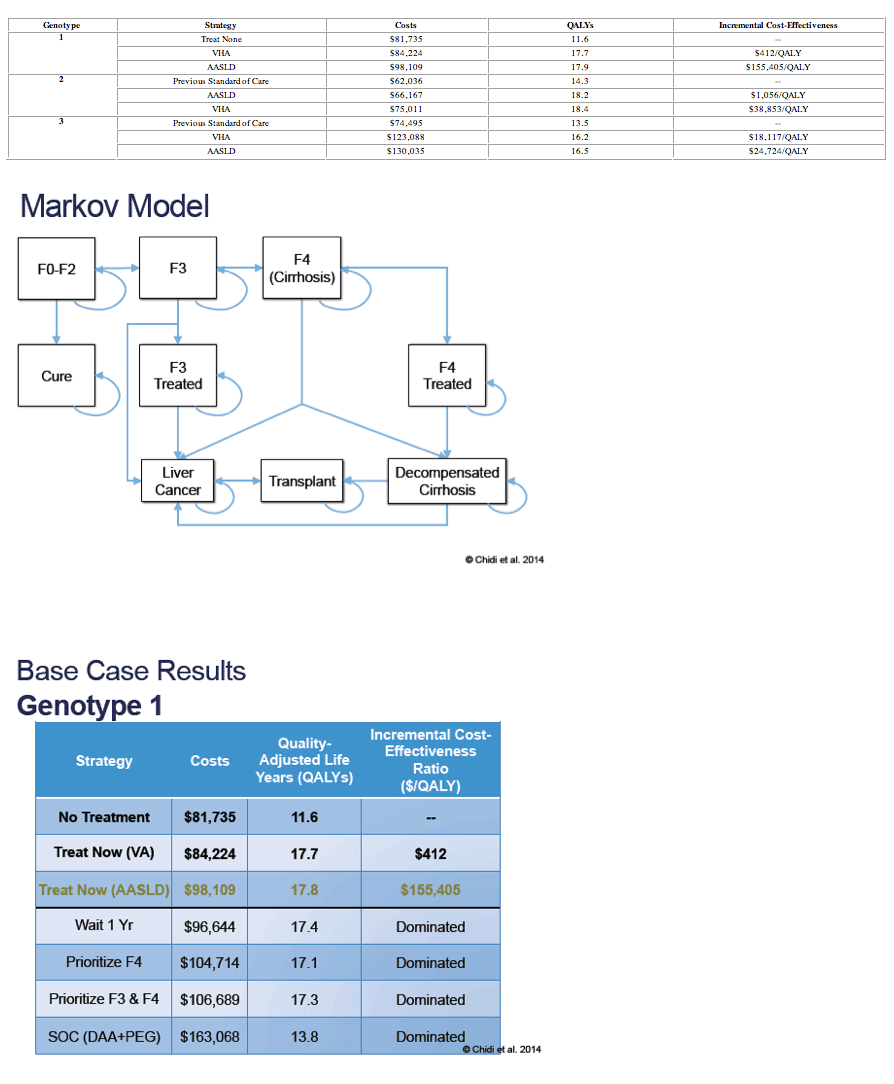
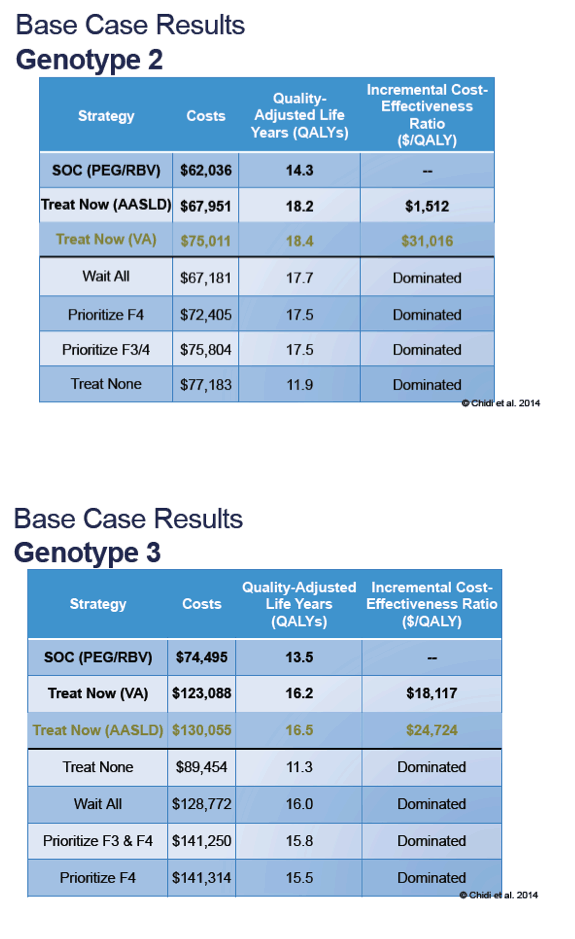
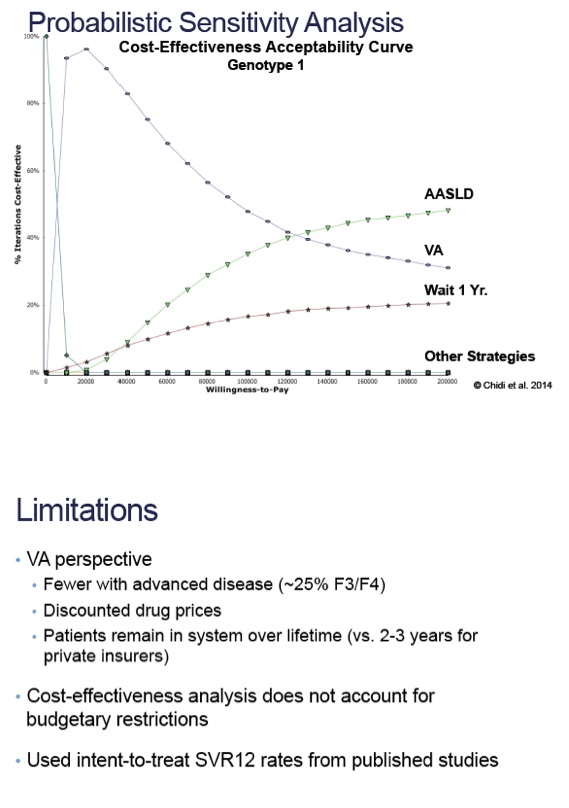
Methods: We used a Markov model with one-year cycle length for a cohort of 50-year old Veterans with genotype 1, 2, or 3 HCV to compare treating: (1) all with current SOF regimens using American Association for the Study of Liver Disease/Infectious Disease Society of America (AASLD) recommendations; (2) METAVIR F3-4 disease with AASLD recommendations and F0-2 disease in one year with future all-oral regimens; (3) all with SOF regimens using Veteran's Health Administration (VHA) guidelines [AASLD alternative recommendation of SOF with pegylated-interferon/ribavirin (PEG/RBV) for PEG-eligible genotypes 1 & 2, wait to treat F0-3 genotype 3]; (4) all with future all-oral regimens in one year; or (5) only cirrhotic (F4) patients. For comparison, we included the previous standard of care (PEG/RBV ± telaprevir/boceprevir) and no treatment. We modeled the natural history of HCV and cirrhosis, assuming progression, morbidity, and mortality risks were lower after sustained virologic response (SVR). Analyses used a VHA perspective, with a 3% annual discount rate and lifetime horizon. We varied model inputs in probabilistic sensitivity analyses.
Results: Preferred strategies included AASLD guidelines for genotypes 1 ($155,405/QALY) and 3 ($24,724/QALY), and VHA guidelines for genotype 2 ($38,853/QALY) [see Table], which were dominant (less costly, more effective) compared to waiting for all-oral regimens or treating based on fibrosis score. Results were sensitive to SVRs for SOF/PEG/RBV, SOF/simeprevir ± RBV and SOF/RBV, costs of future all-oral regimens, and strategies for treating genotype 3.
Conclusion: For treatment-experienced U.S. Veterans, using current SOF-based regimens cost less and was more effective than waiting to treat with future all-oral therapies, regardless of genotype or METAVIR fibrosis score.
|
| |
|
 |
 |
|
|