 |
 |
 |
| |
No Differences in the Efficacy of Fixed-Dose Combination Ledipasvir/Sofosbuvir in Patients According to
Fibrosis Stage Determined by Liver Biopsy or Laboratory Biomarker in Phase 3 Clinical Trials
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
Stuart Gordon,1 Michael Fried,2 Paul Kwo,3 Jenny C. Yang,4 Yanni Zhu,4 Rob Hyland,4 Phillip S. Pang,4 John G. McHutchison,4 K. Rajender Reddy,5 Patrick Marcellin6
1Henry Ford Health System, Detroit, MI; 2University of North Carolina at Chapel Hill Liver Center, Chapel Hill, NC; 3Indiana University School of Medicine, Indianapolis, IN; 4Gilead Sciences, Inc., Foster City, CA; 5University of Pennsylvania, Philadelphia, PA; 6Hôpital Beaujon, University Paris-Diderot, Clichy, France
AASLD: Retreatment of Patients Who Failed Prior Sofosbuvir-Based Regimens With All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir Plus Ribavirin for 12 Weeks - (11/20/14)
AASLD: Virologic Response Rates to All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir Regimens Are Similar in Patients With and Without Traditional Negative Predictive Factors in Phase 3 Clinical Trials - (11/17/14)
AASLD: Ledipasvir/Sofosbuvir Fixed-Dose Combination Is Safe and Efficacious in Cirrhotic Patients Who Have Previously Failed Protease-Inhibitor Based Triple Therapy - (11/11/14)
AASLD: An Integrated Safety and Efficacy Analysis of >500 Patients With Compensated Cirrhosis Treated With Ledipasvir/Sofosbuvir With or Without Ribavirin - (11/11/14)
AASLD: Ledipasvir/Sofosbuvir With Ribavirin for the Treatment of HCV in Patients With Decompensated Cirrhosis: Preliminary Results of a Prospective, Multicenter Study - (11/12/14)

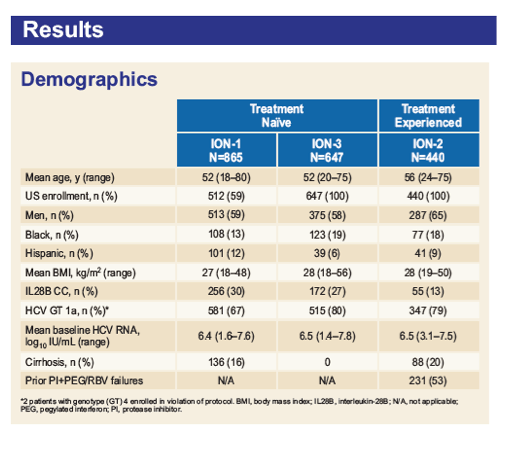
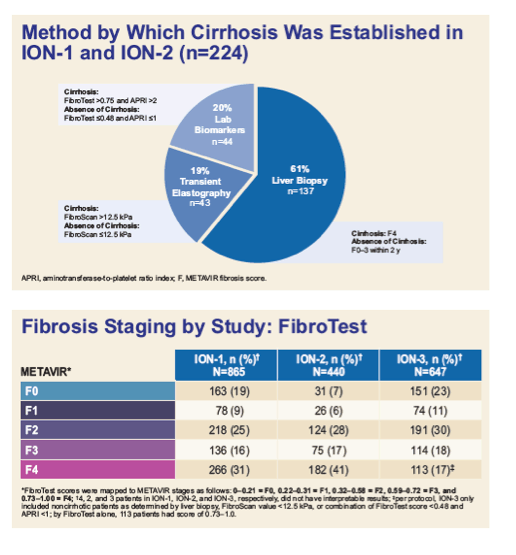
*FibroTest scores were mapped to METAVIR stages as follows: 0-0.21 = F0, 0.22-0.31 = F1, 0.32-0.58 = F2, 0.59-0.72 = F3, and
0.73-1.00 = F4; 4, 2, and 3 patients in ION-1, ION-2, and ION-3, respectively, did not have interpretable results; per protocol, ION-3 only
included noncirrhotic patients as determined by liver biopsy, FibroScan value <12.5 kPa, or combination of FibroTest score <0.48 and
APRI <1; by FibroTest alone, 113 patients had score of 0.73-1.0.
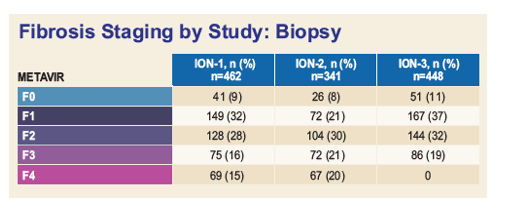
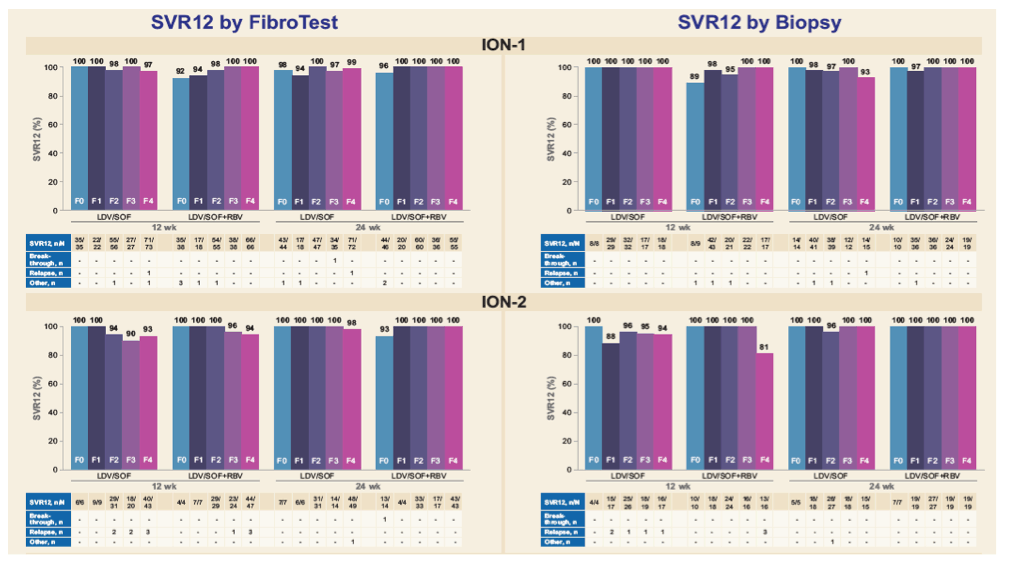
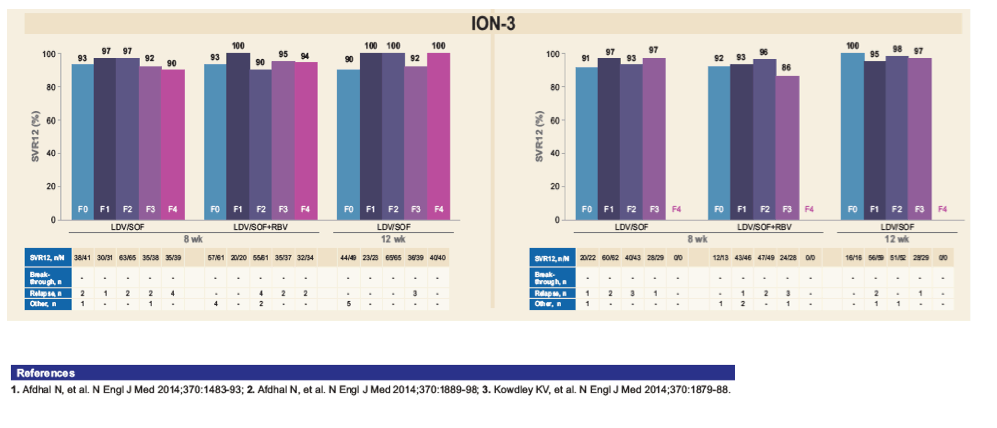
|
| |
|
 |
 |
|
|