 |
 |
 |
| |
High Sustained Response Rates With Sofosbuvir + Ribavirin for 12 or 24 Weeks for Patients With HCV Genotype 2 or 3 Infection: The VALENCE Trial
|
| |
| |
Reported by Jules Levin
Asian Pacific Association for the Study of the Liver (APASL), March 12-15, 2014, Brisbane, Australia
Christophe Hezode,1 Stefan Zeuzem,2 Riina Salupere,3 Alessandra Mangia,4 Robert Flisiak,5 Robert H. Hyland,6 Ari Illeperuma,6 Evguenia S. Svarovskaia,6 Diana M. Brainard,6 William T. Symonds,6 John G. McHutchison,6 Ola Weiland,7 Hendrik W. Reesink,8 Peter Ferenci,9 Geoffrey M. Dusheiko,10 Rafael Esteban11
1Centre Hospitalier Universitaire Henri Mondor de CrŽteil, France; 2Johann Wolfgang Goethe University, Frankfurt, Germany; 3Tartu University Hospital, Tartu, Estonia; 4Casa Sollievo della Sofferenza Hospital, San Giovanni Rotondo, Italy; 5Medical University of Bialystok, Poland; 6Gilead Sciences, Inc., Foster City, CA, USA; 7Karolinska Institutet, Karolinska University Hospital Huddinge, Stockholm, Sweden; 8Academic Medical Center, Amsterdam, the Netherlands; 9Medical University of Vienna, Austria; 10Royal Free and University College School of Medicine, Royal Free Hospital, London, United Kingdom; 11Hospital Universitario Vall dŐHebron, Barcelona, Spain



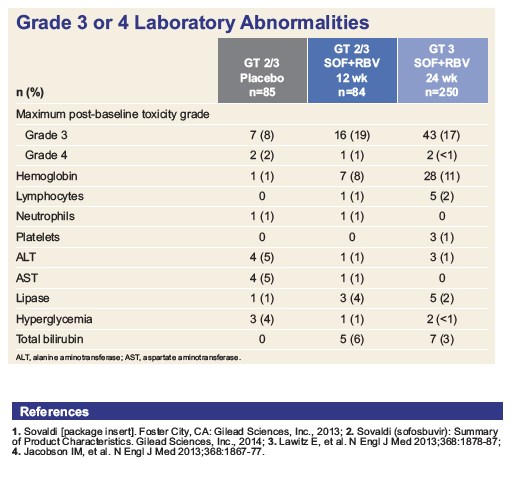
|
| |
|
 |
 |
|
|