 |
 |
 |
| |
Inflammation in Acute HIV Infection Correlates with Blood and Gut CD4 T-cell Loss and HIV Viral Burden
|
| |
| |
"these data suggest that days matter when halting viral replication to reduce the viral reservoir, intestinal CD4 depletion, and inflammation......Plasma levels of biomarkers of inflammation, microbial translocation (MT), fibrosis and coagulation correlated positively with plasma HIV viremia.....and with sigmoid HIV DNA levels"
Reported by Jules Levin
CROI 2014 March 3-6 Boston, MA
Netanya Sandler1,2, Jintanat Ananworanich3,4, Suteeraporn Pinyakorn3,4, Adam Rupert5, Alexandra Schuetz6, James LK Fletcher3, Daniel Douek1, Nicolas Chomont7, Jerome Kim8, Irini Sereti1 on behalf of the RV254/SEARCH010 Study Group
1NIAID/NIH, Bethesda, MD, USA, 2University of Texas Medical Branch, Galveston, TX, USA, 3SEARCH, The Thai Red Cross AIDS Research Center, Bangkok, Thailand," 4HIV-NAT, The Thai Red Cross AIDS Research Center, Bangkok, Thailand, 5SAIC-Frederick, Inc. NCI-FCRDC, Frederick, MD, USA, "
6Armed Forces Research Institute of Medical Sciences - US Component, Bangkok, Thailand, 7Vaccine and Gene Therapy Institute Florida, Port St. Lucie, Florida, USA, " 8US Military HIV Research Program, Walter Reed Army Institute of Research, Silver Spring, MD

Program Abstract:
Background: Biomarkers of inflammation, coagulation and fibrosis are associated with morbidity and mortality in HIV infected persons. Understanding
the changes in these biomarkers during sequential stages of acute HIV infection (AHI) before seroconversion could illuminate the course of early HIV
pathogenesis.
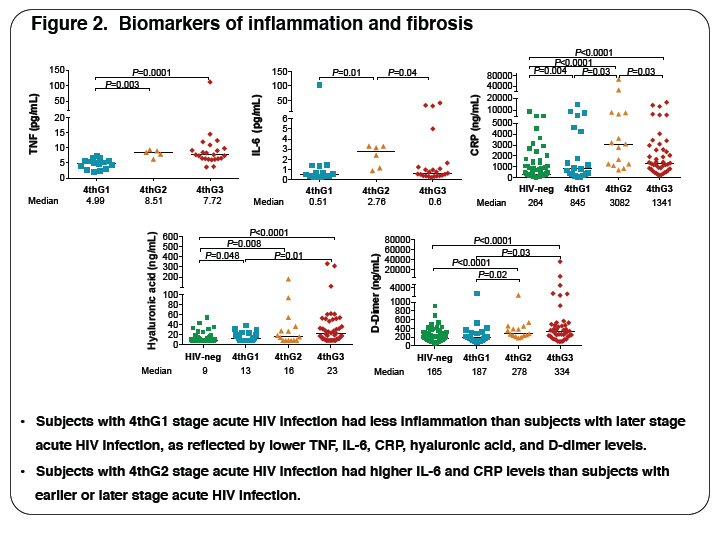
Methodology: Plasma levels of D-dimer, hyaluronic acid (HA), C-reactive protein (CRP), soluble CD14 (sCD14), IL-6 and TNFα were measured from
78 AHI and 109 HIV uninfected (HIV-) Thai participants by ELFA (D-Dimer) and ELISA methods, respectively. 42 AHI participants had sigmoid biopsies.
Cell-associated HIV DNA and RNA in peripheral blood and sigmoid colon were measured by PCR and RT-PCR. Subjects were divided by 4thG staging: 1
(4th generation (G) EIA-, 3rdG EIA-), 2 (4thG +, 3rdG -) and 3 (4thG +, 3rdG +, Western blot - or indeterminate). Median values were compared between
groups by non-parametric methods and Spearman correlations were used for associations between variables.
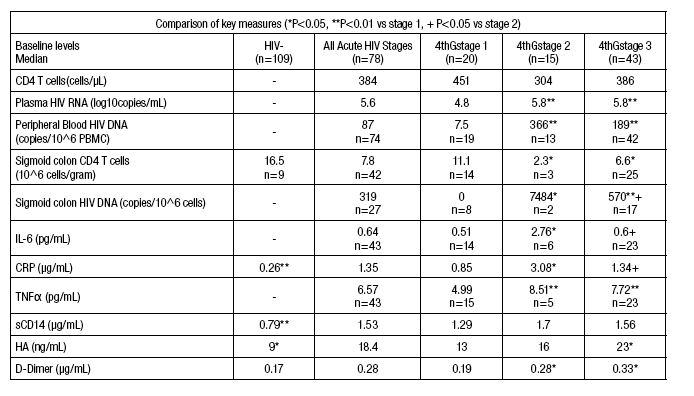
Results: The median age in AHI versus control subjects was 28 vs. 27 years. Median time since HIV exposure was 12, 16 and 18 days in stages 1, 2 and
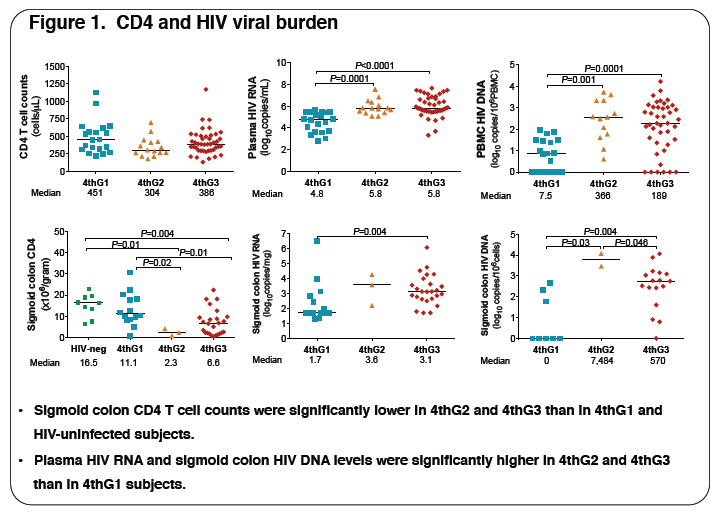
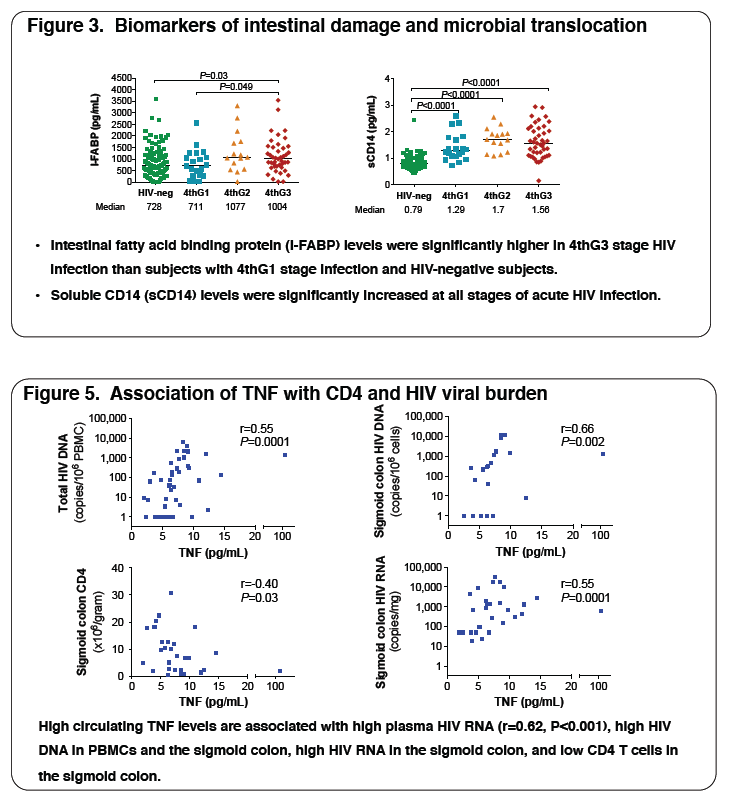
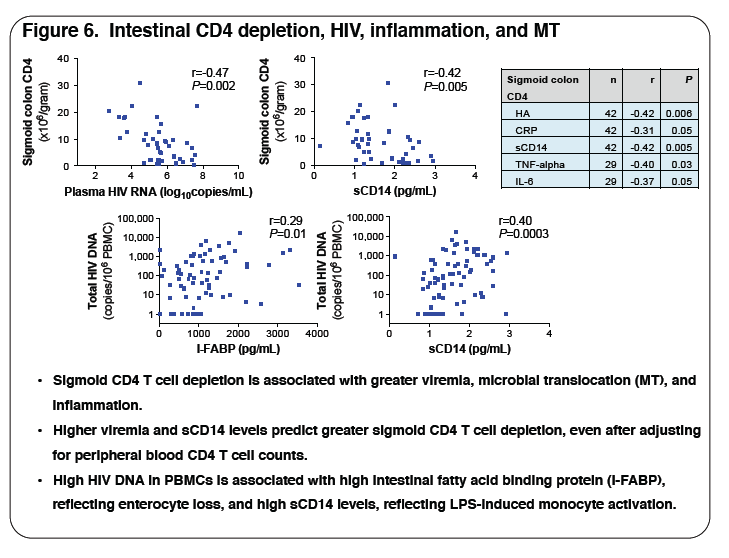
3, respectively. The table illustrates key data. Plasma levels of biomarkers of inflammation, microbial translocation (MT), fibrosis and coagulation correlated positively with plasma HIV viremia: TNFα (r=0.62, P<0.001), sCD14 (r=0.25, P=0.03), HA (r=0.28, P=0.01) and D-dimer (r=0.33, P=0.003) and with
sigmoid HIV DNA levels: TNFα (r=0.66, P=0.002) and CRP (r=0.44, P=0.02). Increased inflammation and MT correlated with low circulating CD4 T cell
counts: TNFα (r=-0.31, P=0.04) and CRP (r=-0.25, P=0.03), and low sigmoid CD4 T cell numbers: TNFα (r=-0.40, P=0.03), CRP (r=-0.31, P=0.05),
sCD14 (r=-0.42, P=0.005) and IL-6 (r=-0.37, P=0.05). High HA levels (r=-0.42, P=0.006), reflecting fibrosis, correlated with low sigmoid CD4 T cell
numbers. Subjects with detectable peripheral blood integrated HIV DNA at week 24 had higher baseline TNFα (8.9 vs. 6.4 pg/mL, P=0.02), sCD14 (2.2
vs. 1.3 μg/mL, P=0.01) and D-dimer (0.41 vs. 0.24 μg/mL, P=0.01) levels.
Conclusions: Biomarkers of inflammation, coagulation and fibrosis are elevated in AHI before seroconversion. The temporal course of these biomarkers
suggests that peak viral burden and inflammation trigger a pro-thrombotic and pro-fibrotic response. Stage 2 may represent the peak of immunologic
destruction and thus a critical point for early intervention to prevent further immune decay and ultimately HIV disease.

|
| |
|
 |
 |
|
|