 |
 |
 |
| |
Stopping TDF Before Renal Impairment Boosts Chance of Improving eGFR
|
| |
| |
CROI 2014, March 3-6, 2014, Boston
Mark Mascolini
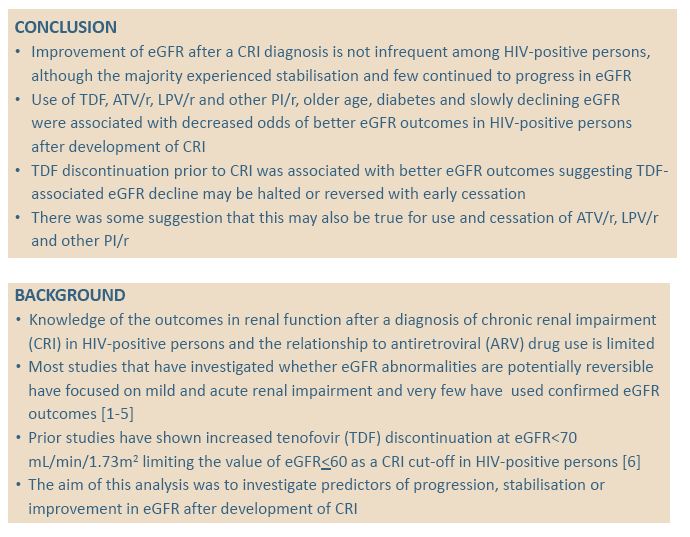
People who stopped tenofovir (TDF) before they had chronic renal impairment (CRI) had better chances of rebounding estimated glomerular filtration rate (eGFR) after impairment, according to a large D:A:D cohort analysis [1]. Findings suggested the same may be true for early stopping of ritonavir-boosted protease inhibitors (PIs). Almost one quarter of people with CRI saw their eGFR improve after reaching that point, about 70% had stabilized eGFR, and 8% got worse.
Although CRI affects many antiretroviral-treated people, D:A:D investigators noted that little is known about what happens to them after their kidneys become impaired and what factors influence whether they get better, get worse, or stay the same. Most studies addressing these questions focused on mild or acute renal impairment, the D:A:D team observed, and few used confirmed eGFR outcomes. To learn more about these issues in the large prospective D:A:D cohort, they conducted this study.
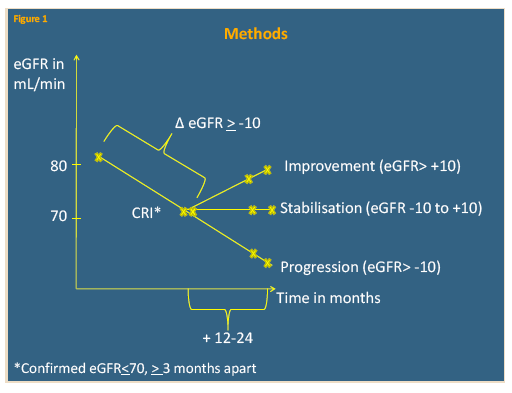
D:A:D involves more than 49,000 HIV-positive people in Europe, Australia, and the United States. This analysis focused on people who had an initial eGFR above 80 mL/min after January 1, 2004 and later had CRI, defined as consecutive eGFRs at or below 70 mL/min at least 3 months apart. The D:A:D team compared the median of all eGFRs in the 12 to 24 months after CRI with the median eGFR that initially defined eGFR. Everyone had at least two eGFRs after CRI.
The researchers grouped eGFR changes into three clusters: progression (decrease more than -10 mL/min), stabilization (-10 to +10 mL/min), and improvement (increase more than +10 mL/min). Logistic regression to assess odds of improvement after CRI considered use or discontinuation of antiretrovirals commonly associated with renal impairment, various demographic and HIV-related factors, HCV status, and traditional renal risk factors such as diabetes and hypertension.
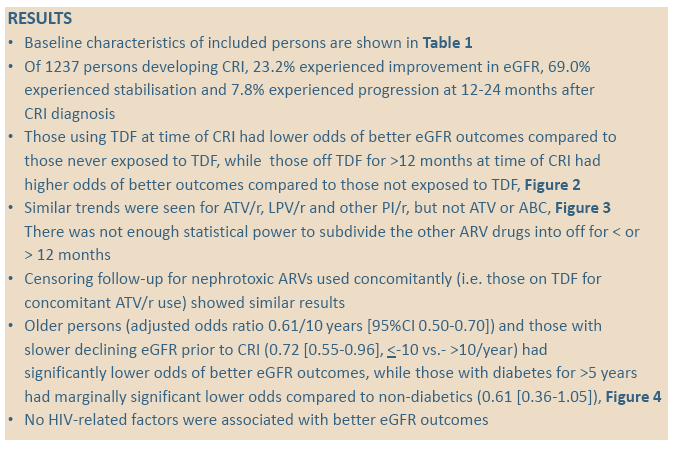
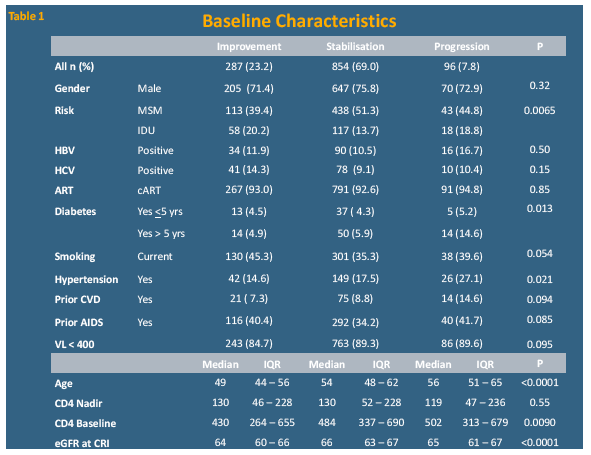
The analysis included 1237 people who reached CRI, of whom 287 (23.2%) improved, 854 (69.0%) stabilized, and 96 (7.8%) got worse. Median age was significantly younger in people with eGFR improvement than in people who stabilized or got worse (49 vs 54 vs 56, P < 0.0001) and the improvers had a lower median eGFR when classified as CRI (64 vs 66 vs 65, P < 0.0001). Median baseline CD4 count was significantly lower in improvers than in the other two groups (430 vs 484 vs 502, P = 0.0090). A significantly lower proportion of improvers than stabilizers or progressors (4.9%, vs 5.9% vs 14.6%, P = 0.013) had diabetes for more than 5 years, and a significantly lower proportion had hypertension (14.6% vs 17.5% vs 27.1%, P = 0.021).
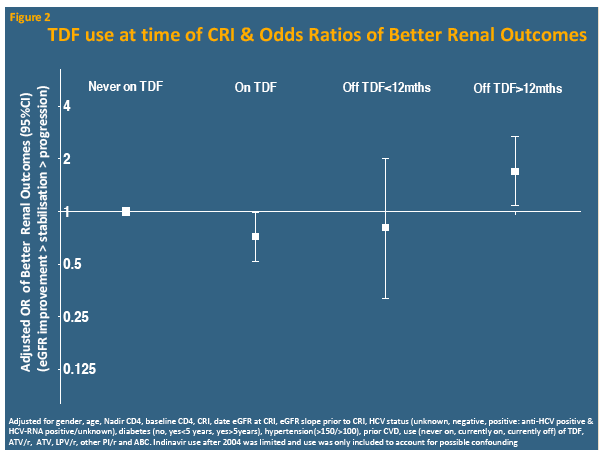
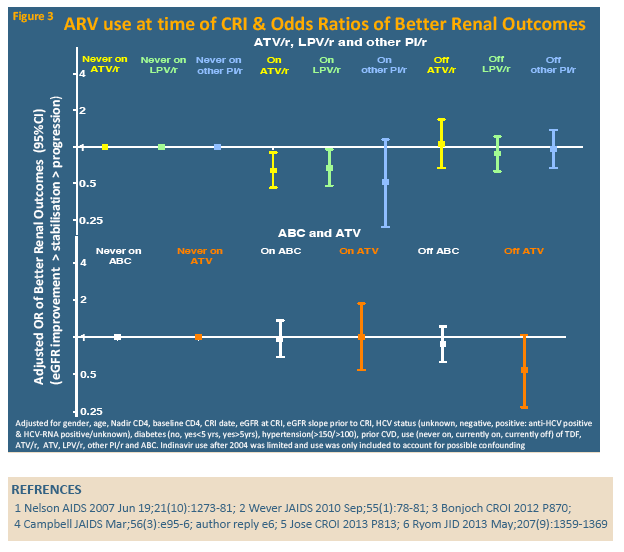
Compared with people who never took tenofovir, those on tenofovir at CRI had independently lower odds of subsequently improving eGFR, while those off tenofovir for more than 12 months had almost doubled odds of improving eGFR. The analysis showed similar trends for atazanavir/ritonavir, lopinavir/ritonavir, and other boosted PIs (grouped together), but nor for unboosted atazanavir or abacavir: People taking these drugs when they reached CRI had less chance of later eGFR improvement, while those off these drugs more than 12 months before CRI had better odds of later eGFR improvement.
Older people had significantly lower odds of improving eGFR after CRI: for every 10 years of age, they had almost a 40% lower chance (adjusted odds ratio [aOR] 0.61, 95% confidence interval [CI] 0.50 to 0.70). People whose eGFR dropped more slowly before CRI had almost a 30% lower chance of eGFR improvement after CRI (aOR 0.72, 95% CI 0.55 to 0.96, for eGFR change < -10 versus > -10 per year). And people with diabetes for more than 5 years had marginally significantly lower odds of post-CRI improvement in eGFR (aOR 0.61, 95% CI 0.36 to 1.05). This analysis disclosed no HIV-related factors linked to improving eGFR after CRI.
The D:A:D team concluded that rebounding eGFR is not rare after CRI, though almost 70% do not improve and 8% have further eGFR deterioration. They believe the association between stopping tenofovir 12 months before CRI and subsequent improvement suggests tenofovir-induced drops in eGFR "may be halted or reversed with early cessation." The same may be true for boosted PIs, though that evidence is not as strong.
Reference
1. Ryom L, Mocroft A, Kirk O, et al. Predictors of progression, stabilisation, or improvement of eGFR after chronic renal impairment. CROI 2014. Conference on Retroviruses and Opportunistic Infections. March 3-6, 2014. Boston. Abstract 792.
---------------------------------------

Predictors of Progression, Stabilisation or Improvement of eGFR After Chronic Renal Impairment
L Ryom1, A Mocroft2, O Kirk1, P Reiss3, M Ross4, O Moranne5, P Morlat6, C Smith2, CA Fux7, S de Wit8, A d'Arminio Monforte9, W El Sadr10, CA Sabin2, A Phillips2, M Law11 and JD Lundgren1 for the D:A:D study group
1CHIP, Dept of Infectious Diseases, Section 2100, Rigshospitalet, University of Copenhagen, Denmark; 2Research Department of Infection and Population Health, UCL, London, United Kingdom; 3Academic Medical Center, Division of Infectious Diseases and Department of Global Health, University of Amsterdam, The Netherlands;
4Division of Nephrology, Mount Sinai School of Medicine, New York, USA; 5Nephrology Department, Public Health Department, CHU Nice, France; 6UniversiteaL Bordeaux Segalen, INSERM U 897, CHU de Bordeaux, France; 7Clinic for Infectious Diseases and Hospital Hygiene, Kantonsspital Aarau, Switzerland; 8CHU Saint-Pierre,
Department of Infectious Diseases, Brussels, Belgium; 9Dipartimento di Scienze della Salute, Clinica di Malattie Infectitive e Tropicali, Azienda Ospedaliera-Polo Universitario San Paolo, Milan Italy; 10ICAP-Columbia University and Harlem Hospital, New York, United States; 11The Kirby Institute, UNSW, Sydney, Australia


|
| |
|
 |
 |
|
|