 |
 |
 |
| |
The Clinical Impact of Viral Load Copy Years in Antiretroviral-Naïve HIV Seroconverters
|
| |
| |
Reported by Jules Levin
CROI 2014 March 3-6 Boston, MA
Matthias an der Heiden1, Alexander Zoufaly2, Caroline Sabin3, Jan van Lunzen4, Hans-Jurgen Stellbrink5, Barbara Gunsenheimer-Bartmeyer1, Philippe Vanhems6, Santiago Perez-Hoyos7, Genevieve Chene8 and Osamah Hamouda1, on behalf of CASCADE Collaboration in EuroCoord
1 Robert Koch-Institute, Department for Infectious Disease Epidemiology, Berlin, GERMANY, 2Universitatsklinikum Hamburg-Eppendorf, Hamburg, GERMANY, 3University College London, Institute of Epidemiology & Health, London, UNITED KINGDOM, 4Universitatsklinikum Hamburg-Eppendorf, Infektiologie, Hamburg, GERMANY, 5Infektionsmedizinisches Centrum Hamburg, Hamburg, GERMANY, 6Centre Hospitalier Universitaire de Lyon, Infection Control, Lyon, FRANCE, 7Institut de Recerca Hospital Vall Hebron, Servei de Medicina Preventiva i Salut Publica, Barcelona, SPAIN, 8UniversitE Victor Segalen Bordeaux 2, Institut de SantE Publique, d'EpidEmiologie et de DEveloppement, Bordeaux, FRANCE

from Jules: the authors suggest below that CD4 >1000 appears perhaps protective compared to CD4 500-1000 but this was not statistically significant in the multivariate analysis perhaps due to small numbers of patients

Program Abstract:
Background: Ongoing viral replication may contribute to immune activation which may have deleterious effects additional to CD4 loss before antiretroviral
therapy (ART) is started. A measure of the cumulative exposure to HIV replication (viral load copy years, VCY) may therefore add important prognostic
information in chronic HIV infection.
Methodology: Data from CASCADE, a cohort collaboration of HIV-1 seroconverters, were used. All ART-naïve patients >15 years with seroconversion (SC)
dates after 1997, who had viral load (VL) and CD4 measured within 4-12 months following SC were included. Patient follow-up began 4 months after SC
and was censored at earliest of ART start, CD4 <200 cells/μl, or when there were no VL and CD4 measurements for >12 months. VCY was approximated
by the time-updated product of VL and the duration to the next VL measurement. A multivariable Poisson model with outcome clinical AIDS or death was
constructed including age at SC as a continuous variable and sex, risk group, current CD4 count and VL, and VCY as categorical variables. To understand
the qualitative impact of VCY better, we also used B-splines to model VCY continuously.
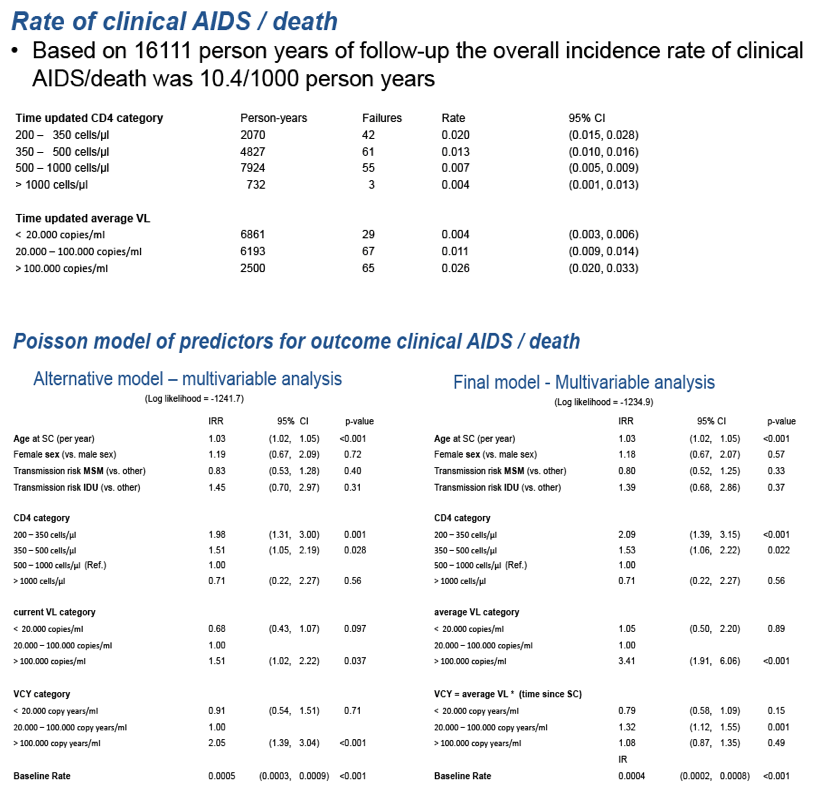
Results: Of 5770 patients with 13,330 years of follow-up, 110 (2%) acquired AIDS or died, 2351 (59%) started ART and CD4 count dropped below 200
cell/μl in 160 patients. In the adjusted Poisson model, older age (rate ratio 1.04 per year [95% confidence interval 1.02-1.06]), injection drug use (2.76
[1.25-6.10]) and having a CD4 count of 200-350 cells/μl (rate ratio 1.93 [1.16-3.23]) compared to 500-1000 cells/μl were associated with a higher rate
of clinical AIDS or death. Compared to VCY category 20,000-50,000 copies°-y/mL, rates of clinical AIDS/death were higher for those with VCY 100,000-200,000
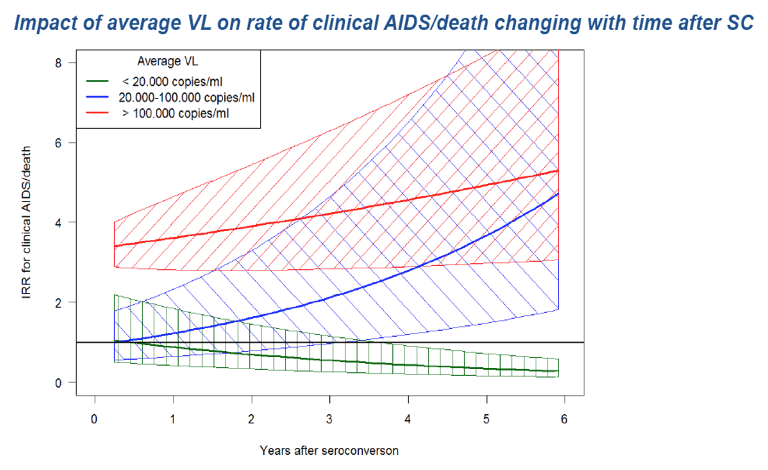
copies°-y/mL (2.4 [1.1-5.2]) and >200,000 copies°-y/mL (4.9 [2.3-10.1]). Modelling log10(VCY) continuously using B-splines in the otherwise unchanged Poisson
model is depicted in Figure 1 for 25 year old males with a CD4 count of 350-500 cells/μl and current VL > 60,000 copies/mL.
Conclusions: VCY appears to provide prognostic information on the risk of clinical AIDS/death additional to current VL and CD4 in naive patients.
Choice on when to start ART may also be influenced by the duration and extent of previous HIV replication.

|
| |
|
 |
 |
|
|