 |
 |
 |
| |
The HDAC inhibitor romidepsin is safe and
effectively reverses HIV-1 latency in vivo as measured by standard clinical assays
|
| |
| |
webcast
http://pag.aids2014.org/flash.aspx?pid=4968
Dowload the PDF here
Dowload the PDF here
Reported by Jules Levin
20th International AIDS Conference, July 20-25, 2014, Melbourne
Ole Schmeltz Sogaard, MD PhD
Department of Infectious Diseases, Aarhus
University Hospital, Denmark

-Potential cART and RMD: Tested in a TZM-bl assay. Using a range of concentrations of PI, NRTIs, NNRTIs and INT, we did not see any see reduced sensitivity to the antiviral effect of these drugs when co-administered with HDACis including RMD and PAN
- Potential RMD and PI interaction: Romidepsin is metabolized by CYP3A4. In recently conducted drug interaction clinical trial evaluating the strong CYP3A4 inhibitor, ketoconazole, showed co-administration of 8mg/m2 romidepsin (4-hour infusion) with ketoconazole, resulted in an overall increase in romidepsin exposure by approximately 25% and 10% for AUC0-∞ and Cmax, respectively, compared to romidepsin. As a comparison, ritonavir has been shown to have an inhibitory potency on CYP3A4 slightly less than that of ketoconazole (von Moltke, Greenblatt et al. 1998). Given the lower dosing of romidepsin in the current study and the estimated <25% increase in romidepsin (AUC0-∞) with ritonavir regimens, subjects on ritonavir boosted protease inhibitors will be allowed in the current study but will be monitored closely for toxicity related to increased romidepsin exposure. BUT PHARMACOKINETICS VS PHARMACODYNAMICS (H3 acetylation). Highest VL blip in a NNRTI treated pt, least and most side effects observed in two PI patients.

-------------------------------
Program abstract
Background: In a recently published ex vivo study, the latency reversing agent (LRA) romidepsin induced HIV expression in resting CD4+ T cells isolated from patients undergoing combination antiretroviral treatment (cART). In light of this exciting finding, we evaluated the effects of romidepsin on measures of viral transcription and plasma viremia in vivo.
Methods: In a phase I/II clinical trial, six aviremic HIV-infected adults received intravenous romidepsin (5 mg/m2) once weekly for 3 weeks while maintaining cART. We used flow cytometry to determine H3 histone acetylation levels in lymphocytes as a cellular measure of the pharmacodynamic response to romidepsin. Changes in intracellular viral transcription were quantitated by cell-associated unspliced HIV-1-RNA (CA-US HIV-1-RNA) using digital droplet PCR in unfractionated CD4+ T cells. Plasma HIV-1-RNA was analyzed by a standard clinical viral load assay (Cobas Taqman) and a transcription-mediated amplification (TMA) assay (Procleix Ultrio Plus). Safety was evaluated at each study visit. Baseline values were compared with post-infusion values using Wilcoxon signed-rank tests. Binary outcomes were analyzed using two-sided binomial exact tests.
Results: All 6 patients (5 males, 1 female) completed three romidepsin infusions. H3 histone acetylation increased rapidly (max 17.7 fold relative to baseline) within the first hours following each romidepsin administration and then decreased between day 3 and 7 day post-infusion. Concurrently, CA-US HIV-1-RNA levels increased significantly from baseline during treatment (2.1-3.9 fold after 2nd infusion; p=0.03). Importantly, viral load increased from "undetectable" at baseline to readily quantifiable levels at multiple post-infusion timepoints in 5 of 6 patients (range 46-103 copies/mL after 2nd infusion, p=0.007). Plasma HIV-1-RNA was also detected by TMA more frequently at post-infusion timepoints vs. baseline (p=0.03 after 2nd infusion). Furthermore, the emergence of quantifiable plasma HIV-1-RNA corresponded directly with the cyclic romidepsin infusions. Adverse events (all grade 1-2) were consistent with the known side effects of romidepsin and HDAC inhibitors in general.
Conclusions: Romidepsin safely induced HIV-1 transcription resulting in plasma viremia that was readily quantified with standard commercial assays. Our data show that potent in vivo latency reversal is possible with a single LRA. A trial combining romidepsin and therapeutic vaccination is ongoing.
----------------------------
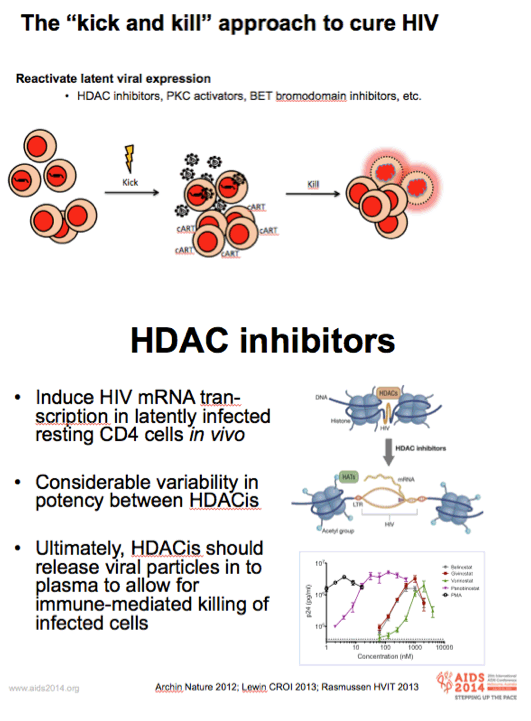
-Acetylation of the lysine residues on the histone tails relaxes the chromatin structure and allows for gene expression, whereas deacetylation of the histone tails leads to condensation of the chromatin structure and silencing of gene expression.
-In accordance with this mechanism, HDACis have been shown to induce HIV transcription in latently infected cells
-Many HDACis, considerable difference in their effect on HIV gene expression

-In the present study, we have used the HDACi, romidepsin
-Much credit for the advancement of this drug in HIV latency should be given to Romas Geleziunas, who together with his team at Gilead and external collaborators have done a tremendous amount of in vitro/ex vivo work with RMD
PLOS Pathogens, Wei, April 2014: Histone Deacetylase Inhibitor Romidepsin Induces HIV Expression in CD4 T Cells from Patients on Suppressive Antiretroviral Therapy at Concentrations Achieved by Clinical Dosing (pdf attached above for download)
http://www.plospathogens.org/article/info:doi/10.1371/journal.ppat.1004071
Nature Medicine, Bullen, 23 March 2014.....New ex vivo approaches distinguish effective and ineffective single agents for reversing HIV-1 latency in vivo (pdf attached above for download)


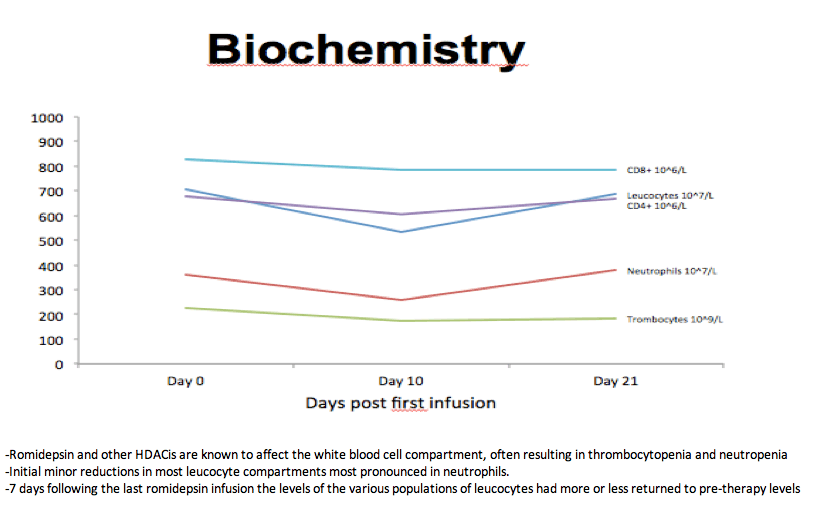
-Romidepsin and other HDACis are known to affect the white blood cell compartment, often resulting in thrombocytopenia and neutropenia
-Initial minor reductions in most leucocyte compartments most pronounced in neutrophils.
-7 days following the last romidepsin infusion the levels of the various populations of leucocytes had more or less returned to pre-therapy levels

-The degree of histone acetylation is a direct measure of the pharmacodynamic effect of an HDACi on cells. Thus, to closely describe the chain of events leading from RMD administration to activation of HIV-1 transcription, we determined lymphocyte histone H3 acetylation at each study visit.
-This graft shows the mean H3 acetylation for the 6 patients as a group
-Emphasize dosing and sampling schedule
- The lasting effect of RMD compared to other HDACi may be related to its unique intracellular pharmacology and interaction with HDAC enzymes. RMD acts as an intracellular prodrug that undergoes reduction of its intramolecular disulfide bond upon entering cells. The released free sulfhydryl groups tightly interact with the Zn2+ ion in the active site of various target HDAC isoforms, a mechanism of inhibition that does not apply to VOR or PNB.
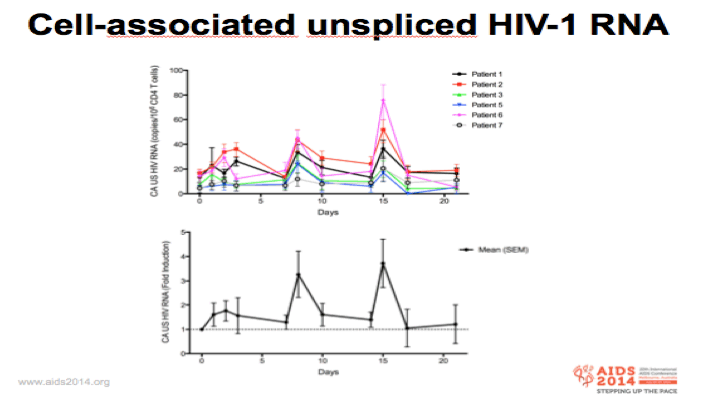
-We determined intracellular HIV RNA levels in total CD4+ cells by ddPCR as a measure of HIV transcriptional activity
-Top graft shows the actual changes in HIV RNA for each individual and the bottom graft shows fold-changes from pre-therapy level for the whole group
-We observed significant increases in CA US HIV RNA in all 6 patients
-Highest levels observed after the 2nd and 3rd dosing (mean 3.5 fold increase)
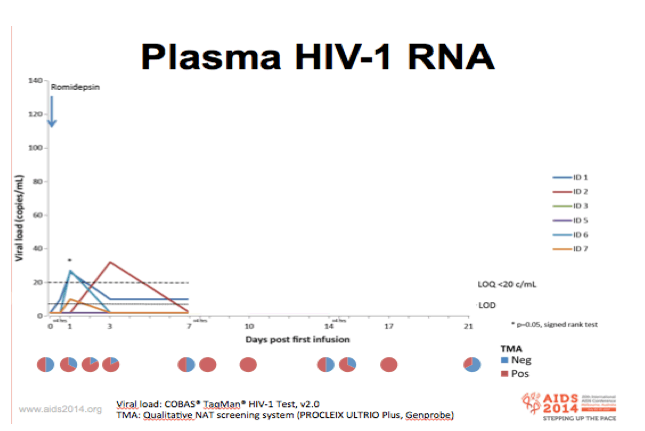
-Plasma viral RNA was determined using the standard COBAS taqman assay
-"Take 1 infusion at a time"
-5 of 6 patients reached quantifiable plasma RNA levels during the 3 infusions
-Using the same qualitative transcription mediated assay that blood bank use for screening of donor blood for early signs of HIV infections, we noted that 50% of the pre-infusion samples were positive for plasma HIV RNA, whereas more than 90% were positive 3 days post-infusion
-Collectively, these data may be represent convincing evidence of a reactivating agent that "kicks" the latently infected cells out of latency
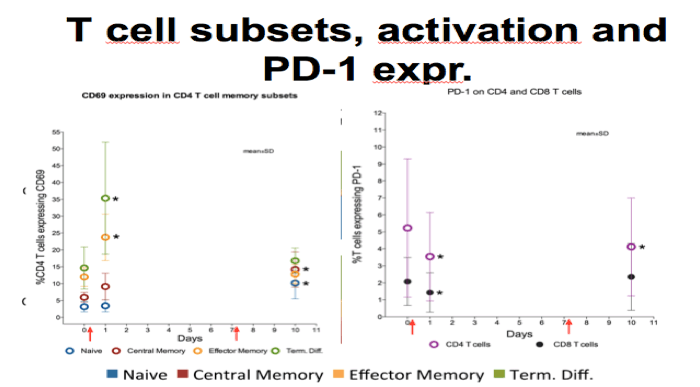
- RMD induced immediately changes in the composition of the peripheral CD4+ and CD8+ compartment
-Shift towards larger proportions of naive cells and fever effector and central memory cells. However, changes had almost reversed by day 10
-CD4 T cell activation as measured by CD69 increased primarily in the effector memory and term diff subsets which is consistant with ex vivo findings
-Expression of the exhaustion marker PD1 decreased initially in both CD4+ and CD8+
-Collectively, these changes may impact measurements of the HIV reservoir and the chance of clearing reactivated cells
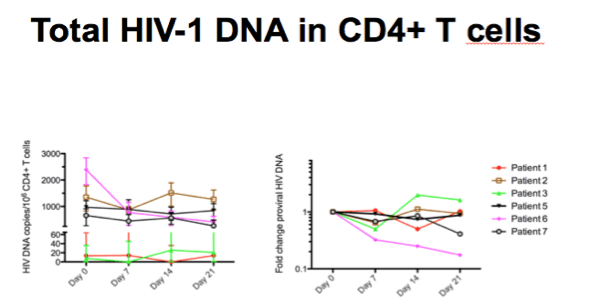
-Individual HIV DNA levels in total CD4+ cells measured by qPCR shown left, fold changes from pre-therapy level shown to the right
-One person had an 80% decrease in HIV DNA but on a group level there was no change from baseline to day 21
|
| |
|
 |
 |
|
|