 |
 |
 |
| |
Early Data Show Good Adherence to Intermittent PrEP by MSM in French Trial
|
| |
| |
20th International AIDS Conference, July 20-25, 2014, Melbourne
Mark Mascolini
High-risk men who have sex with men (MSM) enrolled in a placebo-controlled trial generally adhered well to intermittent preexposure prophylaxis (PrEP) with tenofovir/emtricitabine (TDF/FTC) taken before and after sex [1]. More than 80% of men in the active drug arm had detectable tenofovir and FTC in plasma samples.
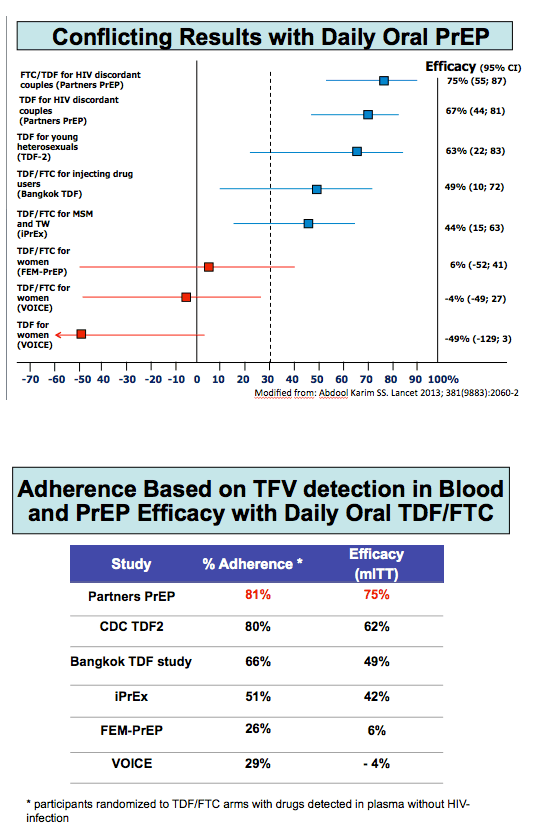
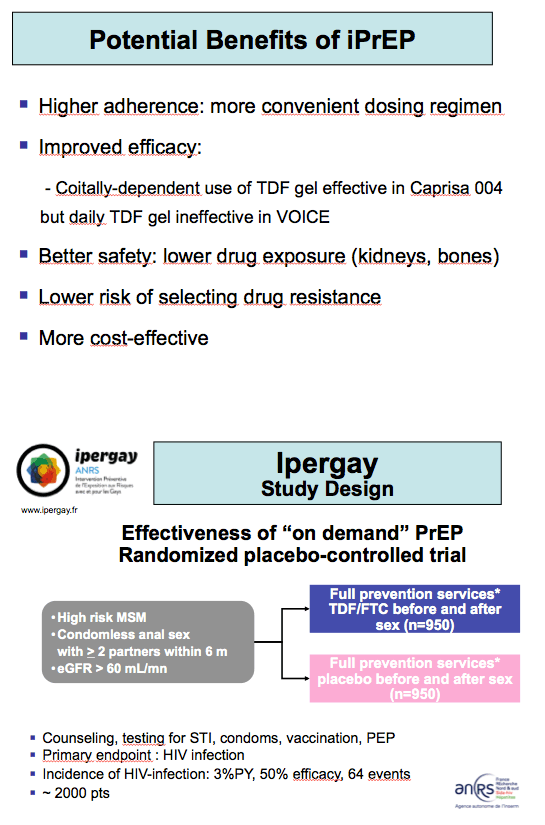
Data from five successful and two unsuccessful TDF/FTC PrEP trials indicate that good adherence to daily oral PrEP is essential to avoiding HIV acquisition. Because daily adherence remains a challenge for many people who might benefit from PrEP, French ANRS Ipergay investigators tested the feasibility of taking TDF/FTC only before and after sex. They noted that tenofovir vaginal gel lowered HIV acquisition risk in the CAPRISA 004 trial [2], whose participants applied the gel before and after sex, but not in the VOICE trial [3], which called for daily tenofovir gel application.
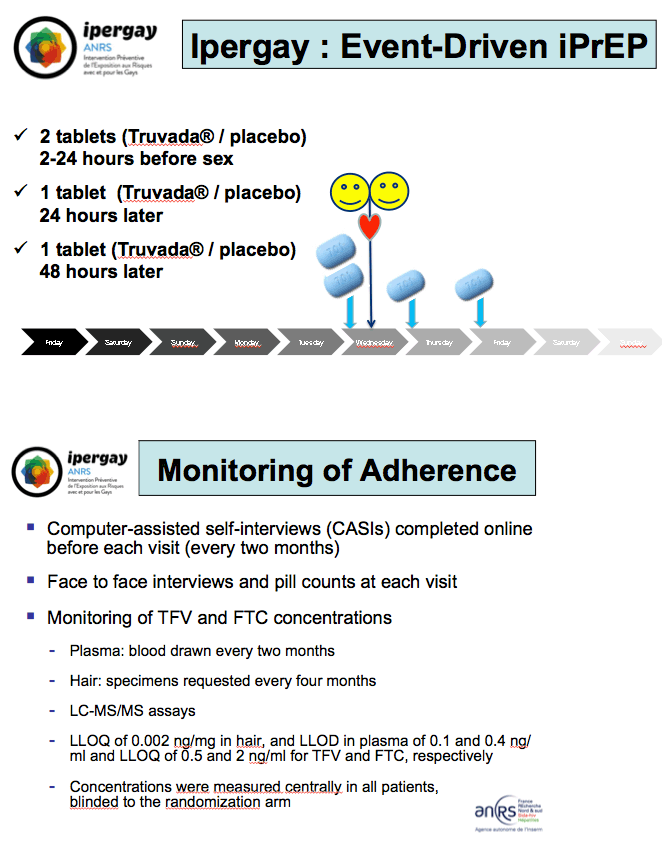
Ipergay enrolls MSM who report condom-free anal sex with 2 or more partners in the past 6 months [1,4]. ANRS investigators ultimately hope to enroll 1900 men, randomizing half to take oral TDF/FTC before and after sex or to take placebo. All participants receive counseling, sexually transmitted infection testing, condoms, and (when necessary) postexposure prophylaxis. The regimen consists of two TDF/FTC tablets (coformulated as Truvada) 2 to 24 hours before sex, one tablet within 24 hours after sex, and another tablet within 48 hours after sex. The trial's pilot phase aims to assess adherence and feasibility.
The investigators rigorously assess adherence via (1) computer-assisted self-interviews completed online every 2 months, (2) face-to-face interviews and pill counts at each study visit, and (3) monitoring tenofovir and FTC concentrations in plasma collected every 2 months and in hair samples clipped every 4 months.
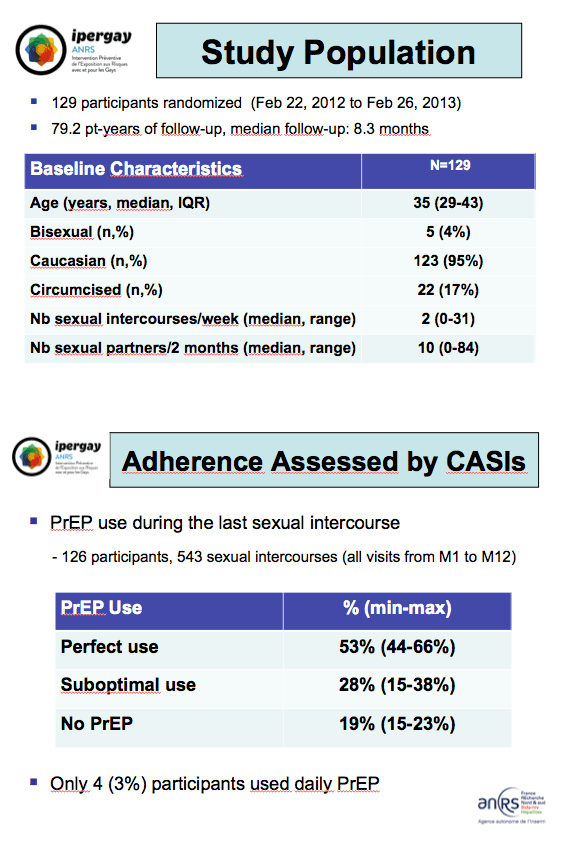
This analysis involved 129 men randomized from February 2012 through February 2013. The group had a median age of 35 years (interquartile range 29 to 43), 123 men (95%) were Caucasian, and 22 (17%) were circumcised. Participants reported a median of 2 acts of sexual intercourse (range 0 to 31) in the past week and a median of 10 partners (range 0 to 84) in the past 2 months. All told, 126 men reported 543 acts of sexual intercourse during 1 year.
Through a median follow-up of 8.3 months, 53% of study participants reported perfect PrEP use before and after their last sex-always taking TDF/FTC as scheduled. Another 28% reported "suboptimal use"-taking the pill but not always on schedule. The remaining 19% reported not using PrEP. Only 4 men (3%) took TDF/FTC daily. Among 578 responses to the question whether they used PrEP since their last visit, 76% used PrEP with each sex act, 11% used PrEP "often," 4% used it "from time to time," and 9% did not use PrEP. Median time between the study visit and last PrEP use was 1.1 days.
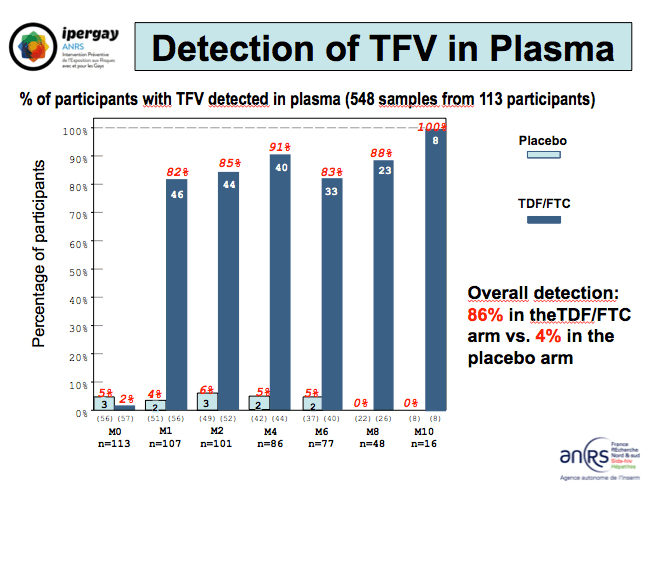
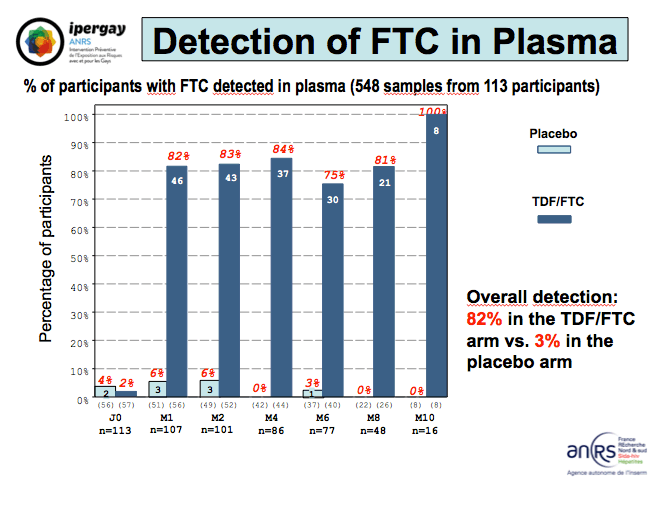
In 548 plasma samples from 113 men, tenofovir could be detected in 86% of samples from men randomized to take the drugs and in 4% of men randomized to placebo. Overall FTC detection rates in plasma measured 82% in the active PrEP arm and 3% in the placebo arm. Preliminary hair drug level results involved 59 samples that could be used to measure drugs. Tenofovir could be detected in 19 of 37 samples (51%) from men randomized to TDF/FTC and in 3 of 22 samples (14%) from men randomized to placebo.
While calling adherence findings to date "very encouraging," Ipergay investigators cautioned that the results "are not a demonstration of the efficacy of 'on demand' PrEP." But they judged that these adherence results are strong enough to support the trial's efficacy phase.
References
1. Fonsart J, Capitant C, Spire B, et al. High adherence rate to intermittent oral PrEP with TDF/FTC among high risk MSM (ANRS Ipergay). AIDS 2014. 20th International AIDS Conference. July 20-25, 2014. Melbourne. Abstract THAC0103.
2. Abdool Karim Q, Abdool Karim SS, Frohlich JA, et al. Effectiveness and safety of tenofovir gel, an antiretroviral microbicide, for the prevention of HIV infection in women. Science. 2010;329:1168-1174. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3001187/
3. Marrazzo J, Ramjee G, Nair G, et al. Pre--exposure prophylaxis for HIV in Women: daily oral tenofovir, oral tenofovir/emtricitabine, or vaginal tenofovir gel in the VOICE Study (MTN 003). 20th Conference on Retroviruses and Opportunistic Infections. March 3-6, 2013. Atlanta. Abstract 26LB. http://www.natap.org/2013/CROI/croi_09.htm
4. ClinicalTrials.gov. On demand antiretroviral pre-exposure prophylaxis for HIV infection in men who have sex with men (IPERGAY). http://clinicaltrials.gov/ct2/show/NCT01473472




|
| |
|
 |
 |
|
|