 |
 |
 |
| |
Predictors of Neurocognitive Decline and Rebound in US CHARTER Group
|
| |
| |
20th International AIDS Conference, July 20-25, 2014, Melbourne
Mark Mascolini
Not taking antiretroviral therapy (ART), markers of poor general health, and neuropsychiatric comorbidities predicted faster neurocognitive decline in a 436-person CHARTER cohort analysis [1]. Higher premorbid IQ, markers of good general health, and lack of depression predicted earlier neurocognitive improvement in these people with HIV.
CHARTER cohort investigators noted that 30% to 50% of HIV-positive people have HIV-associated neurocognitive disorder (HAND) despite access to usually suppressive antiretroviral therapy [2]. Neurocognitive function can fall as people age on ART, but it can also improve. Because most longitudinal studies of neurocognitive change in people with HIV have at most 1 year of follow-up, CHARTER investigators conducted this study, with a long-term goal of identifying strategies that prevent neurocognitive decline or foster its improvement.
The CHARTER cohort includes HIV-positive people from six sites across the United States. Participants complete 15 tests in seven domains. The investigators calculated neurocognitive change as the average z-score for this test battery (sRCS). The z-scores, the CHARTER team explained, "reflect how well or poorly the person performed at follow-up, relative to normal expectation for someone with the same baseline neurocognition and other relevant characteristics" such as age and education.
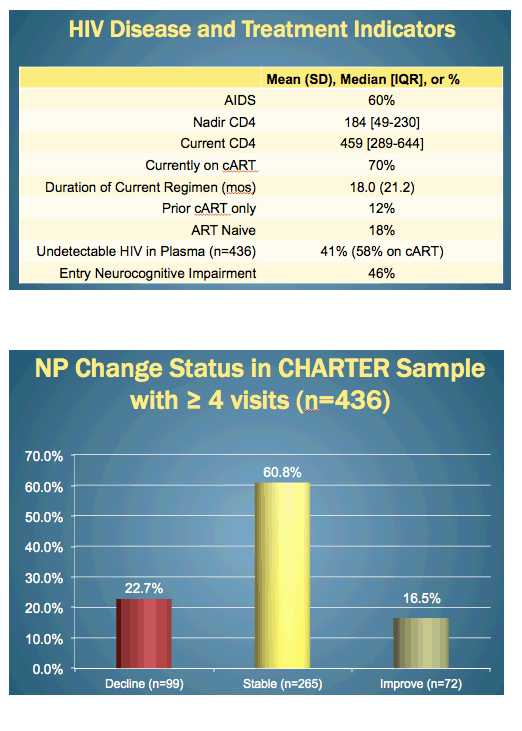
This analysis involved 436 people who made four to seven study visits through an average follow-up of 35 months. Age averaged 43.9 years and education 12.9 years. While 80% of cohort members were men, 43% were Caucasian. Median nadir CD4 count stood at 184 and current count at 459. While 18% of cohort members had never taken antiretrovirals, 12% had taken them but stopped, and 70% were currently on treatment for an average 18 months. Only 41% of the whole group had an undetectable viral load, including 58% on ART. Almost half of the study group, 46%, had neurocognitive impairment when entering CHARTER.
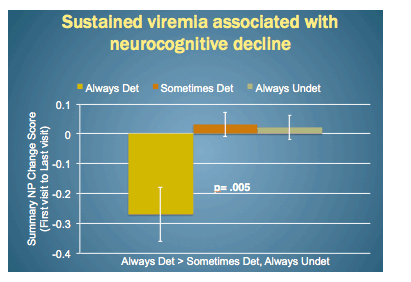
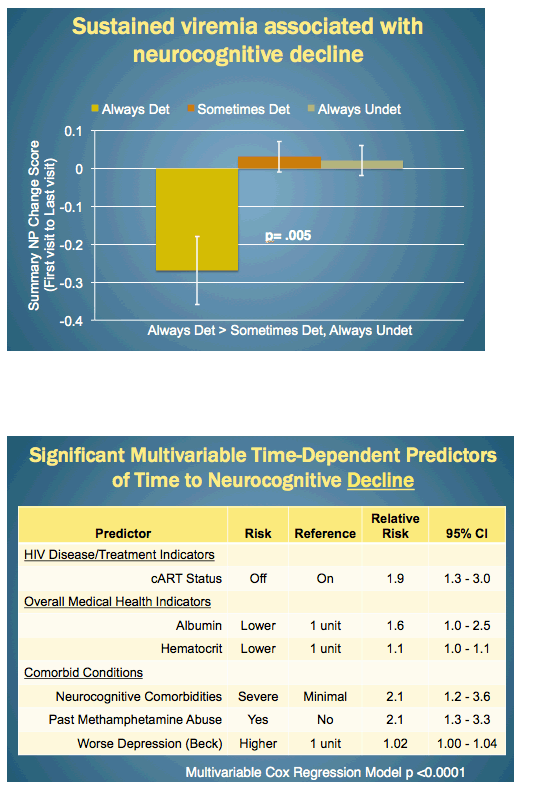
During a median follow-up of 3 years (interquartile range 1.5 to 4), 99 people (23%) had declining neuropsychological performance, 72 (17%) improved, and 265 (61%) remained stable. The summary score dropped more than -0.25 during follow-up in people with an always-detectable viral load, while improving slightly in people with a sometimes-detectable load or an always-undetectable load, a significant difference (P = 0.005).
Multivariable Cox regression analysis identified six predictors of earlier neurocognitive decline at the following relative risks (RR) (and 95% confidence intervals):
-- Off vs on ART: RR 1.9 (1.3 to 3.0)
-- Every 1-unit lower albumin: RR 1.6 (1.0 to 2.5)
-- Every 1-unit lower hematocrit: RR 1.1 (1.0 to 1.1)
-- Severe vs minimal neurocognitive comorbidities: RR 2.1 (1.2 to 3.6)
-- Past methamphetamine abuse: RR 2.1 (1.3 to 3.3)
-- Worse depression per 1 unit on Beck scale: RR 1.02 (1.00 to 1.04)
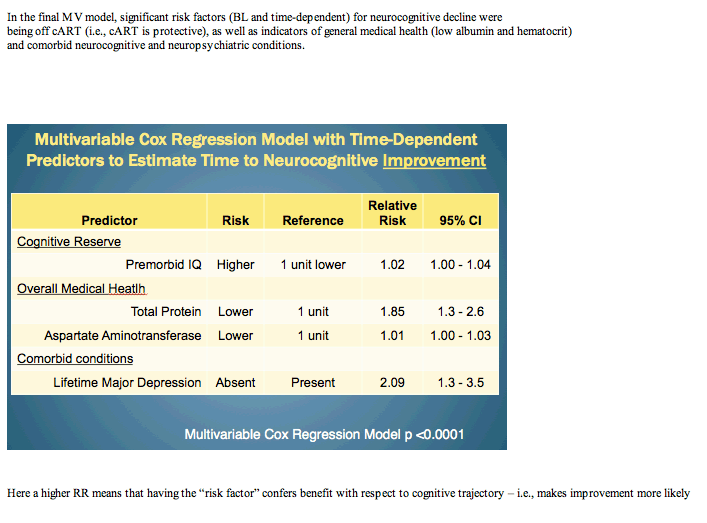
Four factors predicted earlier neurocognitive improvement:
-- Each 1-unit higher premorbid IQ: RR 1.02 (1.00 to 1.04)
-- Each 1-unit lower total protein: RR 1.85 (1.3 to 2.6)
-- Each 1-unit lower aspartate aminotransferase: RR 1.01 (1.00 to 1.03)
-- Lack of lifetime major depression: RR 2.09 (RR 1.3 to 3.5)
The CHARTER team proposed that "enhancing neurocognitive function in people living with AIDS needs attention not only to optimizing ART, but also to comorbidities, particularly substance use and depression, as well as general medical health."
In a separate NATAP report, longer analysis of a larger US cohort, ALLRT, reflected CHARTER's antiretroviral treatment result in finding that more time on ART and bigger CD4 gains protected against neurocognitive decline (measured in a different way) [3]. The ALLRT study found that a history of stroke boosted chances of neurocognitive decline.
References
1. Heaton R, Franklin D, Deutsch R, et al. Neurocognitive change in the era of HIV combination antiretroviral therapy: a longitudinal CHARTER study. AIDS 2014. 20th International AIDS Conference. July 20-25, 2014. Melbourne. Abstract THAB0103.
2. Heaton RK, Clifford DB, Franklin DR Jr, et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology. 2010;75:2087-2096.
3. Smurzynski M, Yang M, Robertson K, et al. Predictors of impaired neurocognitive performance during follow-up among ART-naive individuals initiating ART in ACTG clinical trials. AIDS 2014. 20th International AIDS Conference. July 20-25, 2014. Melbourne. Abstract THAB0106.

Previous studies have demonstrated that good neurocognitive function is important for better QOL and resilience in the face of growing older with HIV infection.
Our results imply that in addition to optimizing cART, in order to enhance neurocognitive function in the large number of aging PLWHA, we also need greater attention to neuropsychiatric comorbidities, particularly substance abuse and depression.
Ultimately, studies should begin to assess the impact of interventions for these comorbidities on ARV adherence and overall health status

In the U.S., only about 30% of HIV-infected individuals actually benefit optimally from ART by virtue of having an undetectable VL. Of the remaining 70%, a considerable proportion (ranging from 10-50% of those receiving ART) fail to attain virologic suppression, and many more do not receive ART and therefore have active viral replication for a variety reasons. People with SMI, many of whom require treatment with SGAs, are disproportionately represented among those who do not benefit from ART, in many cases because of poor health services utilization and poor adherence to medications. We'll come back to this point in the context of considering whether the increased risk of CV disease is attributable to ART, HIV itself, or both.
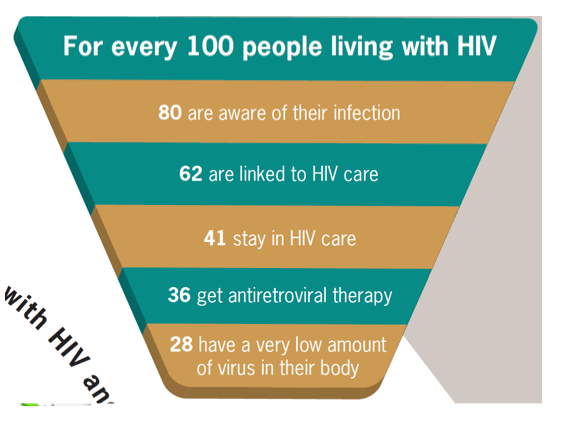
A U.S. Centers for Disease Control and Prevention analysis, published on November 29, 2011, indicates that for every 100 people living with HIV in this country, only 80 are aware of their infection, 62 have entered into care and 41 actually stay in HIV care.
Another study of available data, published in a March 2011 issue of Clinical Infectious Diseases, paints a similar picture: Of the 1.1 million estimated to be living with HIV in the U.S., roughly 874,000 have been diagnosed, 656,000 have been linked to HIV care and 437,000 have been retained in HIV care.
In 2010, a CDC analysis of 50 studies conducted between 1996 and 2009 indicated that 30 to 40 percent of people with HIV fail to enter or stay in care after their initial HIV diagnosis.



The outcome here, rather than reaching a threshold of impairment or a simple difference score is instead whether the subject ever met during the follow-up interval a criterion for clinically significant cognitive decline as defined byÉ
Published, regression-based norms for NC change were used to generate overall change status (decline vs. stable vs. improved) at each study visit. Survival analysis was used to examine the predictors of time to NC change.
In order to determine NC change, we generated a Z-score for each of 15 neuropsychological variables based on published normative data10. These Z-scores reflect how well or poorly the person performed at follow-up, relative to normal expectation for someone with the same baseline NC and other relevant characteristics (e.g., age, education). The Z-scores were then averaged to provide a summary regression change score (sRCS). The top 5% of the sRCS distribution of the normative sample defined the "improve" range and the bottom 5% defined the "decline" range10. The remaining middle 90% were defined as "stable". NC change status from baseline was generated for each follow-up visit. The individual visit change status for each participant was then merged into an overall change status: 1) Decline: if a participant had at least one "decline" status and no "improve" status across visits; 2) Improve: if a participant had at least one "improve" status and no "decline" status; 3) Stable: if a participant had no "decline" or "improve" NC change status (all visits "stable"). Two participants out of an original cohort of 438 met criteria for both "improve" and "decline" during the follow-up period (at different visits) and were thus excluded from analyses.



|
| |
|
 |
 |
|
|