 |
 |
 |
| |
High Rate of NPEP Completion, Good Safety, With Raltegravir Plus TDF/FTC
|
| |
| |
IDWeek 2014, October 8-12, 2014, Philadelphia
Mark Mascolini
More than 4 in 5 people in a Houston study completed nonoccupational postexposure prophylaxis (NPEP) with raltegravir plus tenofovir/emtricitabine (TDF/FTC) [1]. African Americans proved less likely to complete their NPEP course.
Antiretrovirals are recommended for NPEP, but the University of Texas team that conducted this study noted that suggested regimens have not kept pace with currently available possibilities. They have been using raltegravir plus TDF/FTC in people with possible sexual exposure to HIV.
This prospective cohort study focused on safety and tolerability of that regimen, as well as completion rate. From May 2011 through July 2013, the Houston team enrolled 103 people 18 or older who had vaginal, anal, or other mucosal exposure to ejaculate rectal or cervicovaginal secretions from an HIV-positive person or a high-risk partner with an unknown HIV status. Exposure occurred within the previous 72 hours.
At their first visit, volunteers who tested negative for HIV received a 14-day supply of raltegravir plus TDF/FTC and a 24-hour phone number they could call to discuss adverse events or side effects. At a follow-up visit 10 to 14 days after the first visit, participants received the remaining 14-day supply of antiretrovirals and health workers assessed adherence. Participants had additional follow-up visits with HIV testing at 28 days, 3 months, and 6 months.
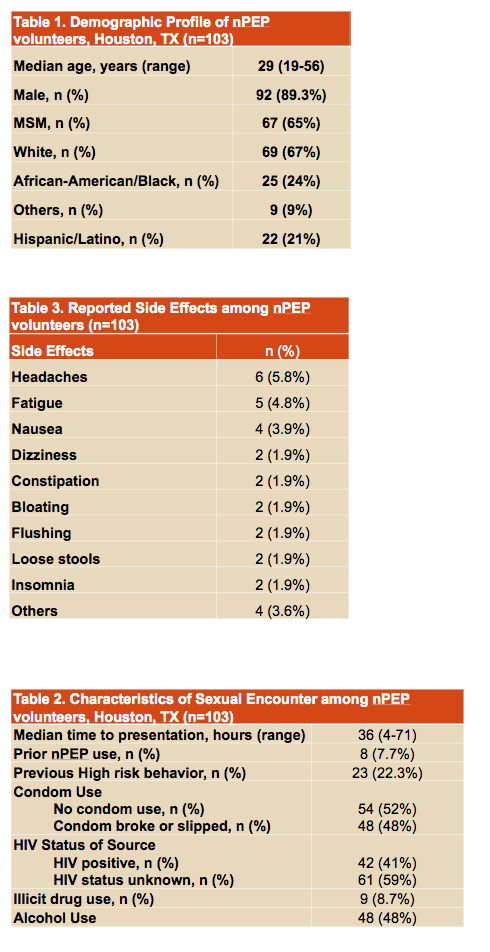
During the study period 103 people enrolled, including 92 men (89%), 67 men who have sex with men (65%), 69 whites (67%), and 25 African Americans (24%). Twenty-two people (21%) identified themselves as Hispanic. Median time to seeking NPEP was 36 hours (range 4 to 71) and 8 people (8%) had used NPEP before. Fifty-four people said they had not used a condom during sex (52%), while the others said the condom broke or slipped. Forty-two people (41%) knew their sex partner had HIV, and the rest did not know.
Participants reported only grade 1 side effects, including headaches in 6, fatigue in 5, and nausea in 4. Eleven people reported missing NPEP doses, but only one or two doses.
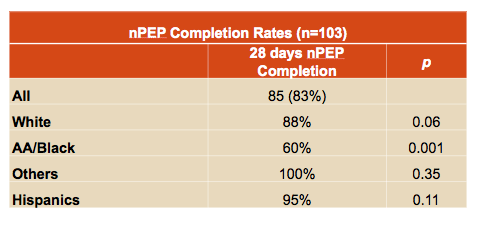
Fifty-five people (53%) completed all study visits, and 85 (83%) completed their 28-day NPEP course, including 95% of Hispanics, 88% of whites, and 60% of African Americans. Compared with other races and ethnicities, the 28-day completion rate was significantly lower in African Americans (P = 0.001). No volunteers who completed all study visits had HIV seroconversion by the 6-month follow-up visit.
The researchers concluded that raltegravir plus TDF/FTC is a well-tolerated NPEP regimen with high rates of adherence and treatment completion. They proposed that "efforts on prevention strategies need to continue to focus on the high-risk groups, particularly those with low adherence to treatment completion such as African Americans."
Reference
1. Vigil KJ, Simmons P, Luna K, Martinez ML, Hasbun R, Arduino R. Raltegravir plus tenofovir DF and emtricitabine for non-occupational postexposure prophylaxis (nPEP): African-Americans are at higher risk of non-completion of nPEP. IDWeek 2014. October 8-12, 2014, Philadelphia. Abstract 1529.



|
| |
|
 |
 |
|
|