 |
 |
 |
| |
In Vitro Drug Interaction Profile of the HIV Integrase Inhibitor, GSK1265744, and Demonstrated Lack of Clinical Interaction with Midazolam
|
| |
| |
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HI V and Hepatitis Therapy; May 19-21, 2014; Washington, DC
Reese M, Ford S, Bowers G, Humphreys J, Webster L, Gould E, Polli J, Generaux G, Johnson M, Clark P, Watson C, Lou Y, Piscitelli S
GlaxoSmithKline, Research Triangle Park, NC USA; King of Prussia, PA USA
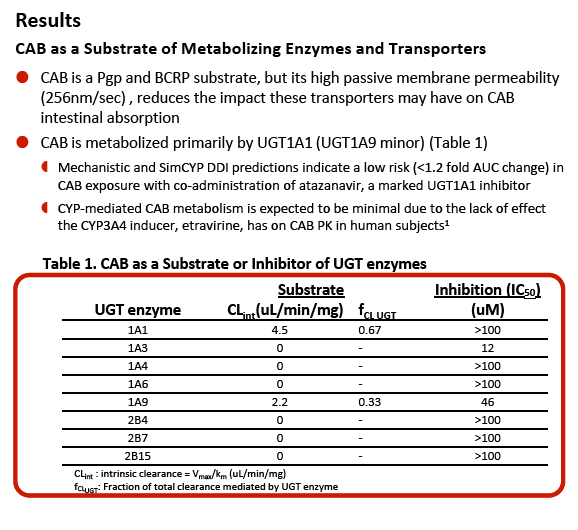
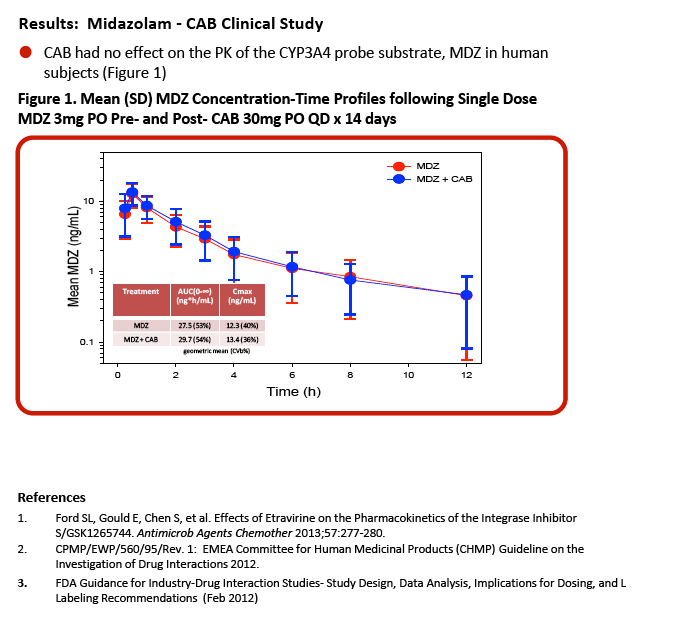
"GSK1265744 (cabotegravir; CAB) is a potent HIV integrase inhibitor in Phase 2 clinical development as an oral and long-acting (LA) injectable formulation for the treatment and prevention of HIV infection .........objectives of this study ........Evaluate the potential for CAB to cause or be subject to clinical drug interactions based on in vitro drug metabolizing enzyme and transporter data and reported clinical interaction studies ........Evaluate the effect of CAB on the PK of .......midazolam (MDZ), a CYP3A4 clinical probe, in human subjects"



|
| |
|
 |
 |
|
|