 |
 |
 |
| |
Raltegravir Levels With Once-Daily Dosing in French Observational Study
|
| |
| |
15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy, May 19- 21, 2014, Washington, DC
Mark Mascolini
All but 3 people in a 71-patient French observational study maintained a viral load below 50 copies for 48 weeks after switching from a stable suppressive regimen to a combination containing 800 mg of raltegravir once daily [1]. About 1 in 5 study participants had a 24-hour raltegravir concentration (C24h) below 22 ng/mL (45 nM), the proposed cutoff for in vivo activity.
The randomized QDMRK trial determined that once-daily raltegravir is not noninferior to twice-daily dosing as a first-line regimen including tenofovir/emtricitabine [2]. "Despite high response rates with both regimens," QDMRK researchers advised, "once-daily raltegravir cannot be recommended in place of twice-daily dosing." In Spain, ODIS trial investigators enrolled people with a sub-50-copy viral load while taking a protease inhibitor and randomized them to once- or twice-daily raltegravir [3]. After 24 weeks of raltegravir, they found a trend toward a higher risk of virologic failure with once-daily raltegravir in participants with nucleoside-associated resistance mutations when they started the integrase inhibitor. However, once-daily raltegravir failed in only 1 of 148 people without baseline nucleoside resistance.
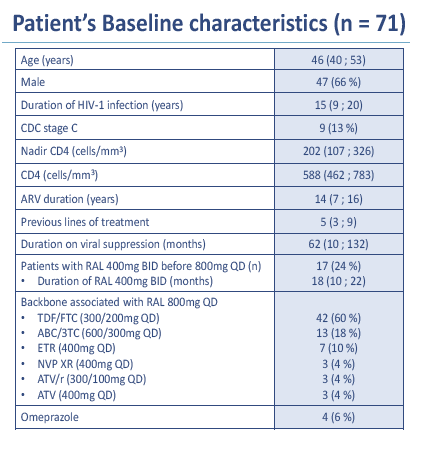
Gilles Peytavin and colleagues in Paris have been studying 71 people who switched from a suppressive regimen to once-daily raltegravir--two 400-mg tablets of the licensed oral compressed tablet (OCT) formulation [1]. This multicenter observational study involves adults with a viral load below 50 copies for at least 6 months and no integrase inhibitor experience except 400 mg of raltegravir twice daily. No one had a history of virologic failure with raltegravir. Median age stood at 46 years (interquartile range [IQR] 40 to 53), and 47 cohort members (66%) were men.
Viral suppression duration upon switching to once-daily raltegravir averaged 62 months, and median CD4 count was 588 (IQR 462 to 783). Participants had taken antiretrovirals for a median of 14 years (IQR 7 to 16) and had taken a median of 5 regimens (IQR 3 to 9). Seventeen of these 71 people (24%) had taken twice-daily raltegravir for an average 8 months. Forty-seven people switched to raltegravir because of tolerance problems with their current regimen, 6 switched because of drug-drug interactions, and 4 switched to simplify their regimen.
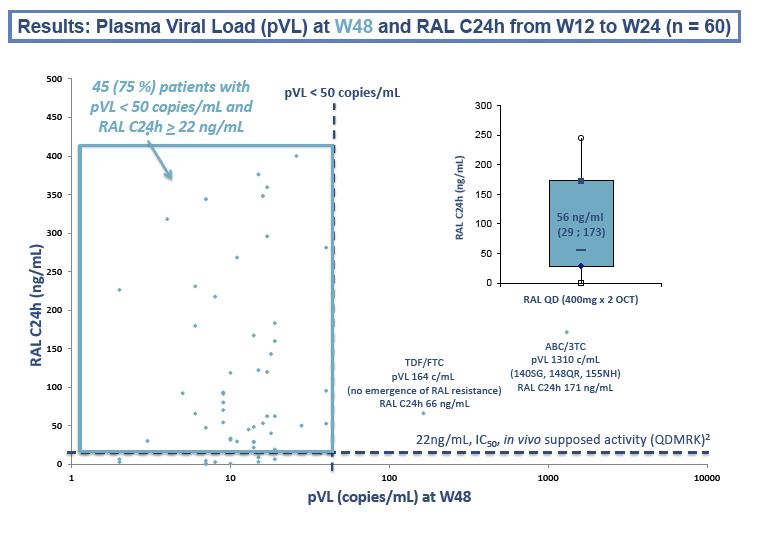
After 24 weeks of once-daily raltegravir, 70 of 71 people (99%) maintained a viral load below 50 copies. After 48 weeks, 2 more people had a virologic failure [4]. All three people with virologic failure had a previous failure while taking two nucleos(t)ides, and 2 of them with genotyping available had detectable baseline nucleos(t)ide-related mutations. Raltegravir mutations (Q148R/K, N155H, and G140S) emerged upon failure in these 2 people.
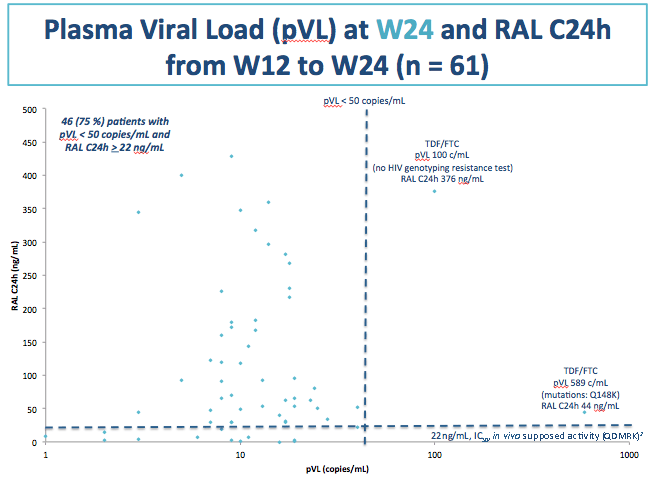
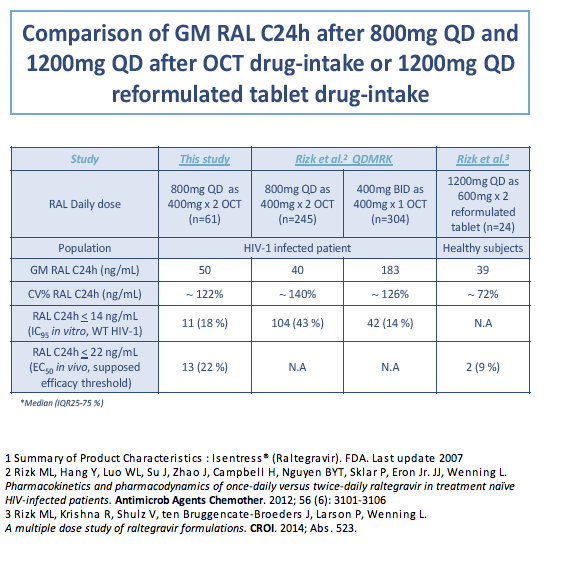
The Paris team determined geometric mean raltegravir 24-hour concentration (C24h) in 61 study participants as 50 ng/mL (coefficient of variation [CV] 122%). The researchers figured that 11 participants (18% of 61) had a C24h below 14 ng/mL (31 nM), the in vitro 95% inhibitory concentration against wild-type HIV in 50% human serum. Thirteen participants (21%) had a C24h below 22 ng/mL (45 nM), the assumed efficacy threshold in vivo.
Recent research by Merck investigators in healthy volunteers determined that 1200 mg of raltegravir once daily as three OCT tablets or as two reformulated 600-mg tablets taken fasting yielded 5-day geometric mean trough concentrations of 40 ng/mL (CV 53%) and 39 ng/mL (72%) [5].
References
1. Belissa E, Caby F, Bonmarchand M, et al. Pharmacokinetics of raltegravir (800 mg) once-daily in switching strategies in HIV-1-infected patients with suppressed viremia. 15th International Workshop on Clinical Pharmacology of HIV and Hepatitis Therapy. May 19-21, 2014. Washington, DC. Abstract PP_02.
2. Eron JJ Jr, Rockstroh JK, Reynes J, et al. Raltegravir once daily or twice daily in previously untreated patients with HIV-1: a randomised, active-controlled, phase 3 non-inferiority trial. Lancet Infect Dis. 2011;11:907-915. Erratum in: Lancet Infect Dis. 2011;11:895. Dosage error in article text. http://www.ncbi.nlm.nih.gov/pubmed/21933752
3. Vispo E, Barreiro P, Maida I, et al. Simplification from protease inhibitors to once- or twice-daily raltegravir: the ODIS trial. HIV Clin Trials. 2010;11:197-204.
4. Caby F, Bonmarchand M, Soulie C, et al. Efficacy of raltegravir once daily in switching strategies in HIV-1 infected patients with suppressed viraemia. 14th European AIDS Conference. October 16-19, 2013. Brussels. Abstract BPD1/7. http://www.natap.org/2013/EACS/EACS_25.htm
5. Rizk ML, Krishna R, Schultz v, et al. A multiple dose study of raltegravir formulations. CROI 2014. Conference on Retroviruses and Opportunistic Infections. March 3-6, 2014. Boston. http://www.natap.org/2014/CROI/croi_200.htm






|
| |
|
 |
 |
|
|