 |
 |
 |
| |
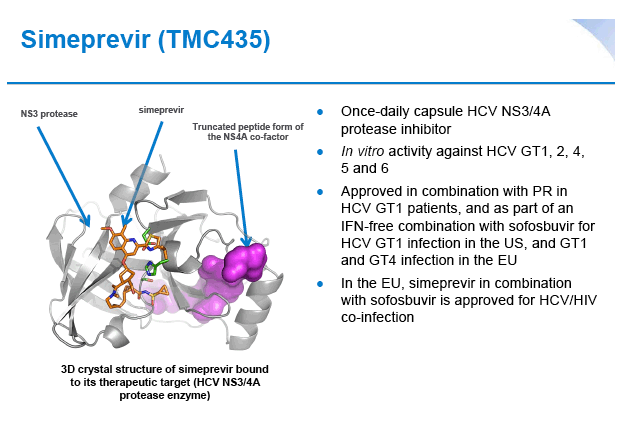
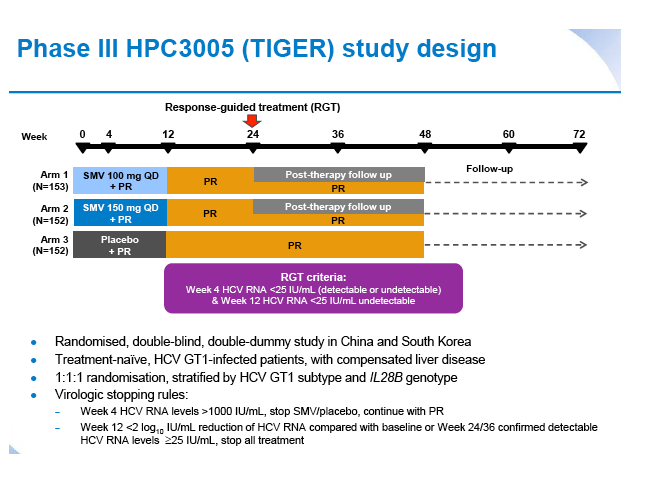
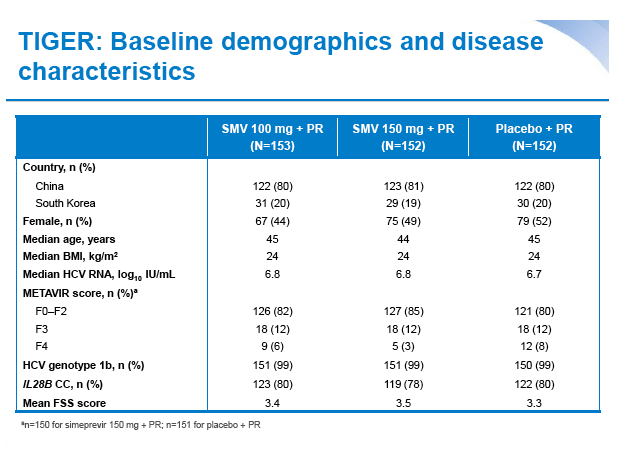
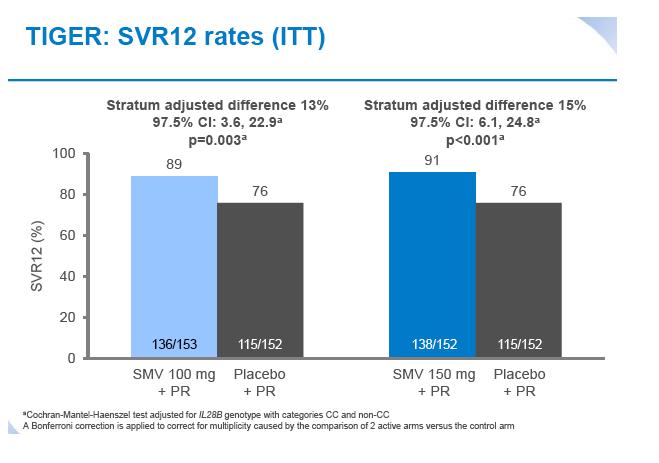
Simeprevir plus peginterferon/ribavirin in treatment-naïve patients with chronic hepatitis C virus genotype 1 infection: Results from the Phase III TIGER study conducted in East Asian patients living in China and Korea
|
| |
| |
Reported by Jules Levin
APASL 2015
24th Conference of the Asian Pacific Association for the Study of the Liver (APASL), 12-15 March 2015, Istanbul, Turkey
Wei L1, Han T2, Yang D3, Heo J4, Shang J5, Cheng J6, Chen X7, Xie Q8, Kim J9, Kalmeijer R10, Ouwerkerk-Mahadevan S11, Lenz O12, Sinha R13, Li M14, Scott J15, Peeters M12, Witek J13
1Peking University People's Hospital and Peking University Hepatology Institute and Peking Key Laboratory for Hepatitis C and Immune Therapy, Beijing, China; 2Tianjin Third Central Hospital, Tianjin, China; 3Union Hospital of Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China; 4Pusan National University and Medical Research Institute, Busan, Republic of Korea; 5Henan Provincial People's Hospital, Zhengzhou, China; 6Beijing Ditan Hospital, Capital Medical University, Beijing, China; 7Beijing You An Hospital, Capital Medical University, Beijing, China; 8Ruijin Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China; 9Gachon University Gil Medical Center, Incheon, Republic of Korea; 10Janssen Global Services LLC, Titusville, NJ, USA; 11Janssen Research & Development BVBA, Beerse, Belgium; 12Janssen Infectious Diseases BVBA, Beerse, Belgium; 13Janssen Research & Development LLC, Titusville, NJ, USA; 14China R&D and Scientific Affairs, Xi'an Janssen Pharmaceutical Company, Beijing, China; 15Janssen Global Services, LLC, High Wycombe, UK

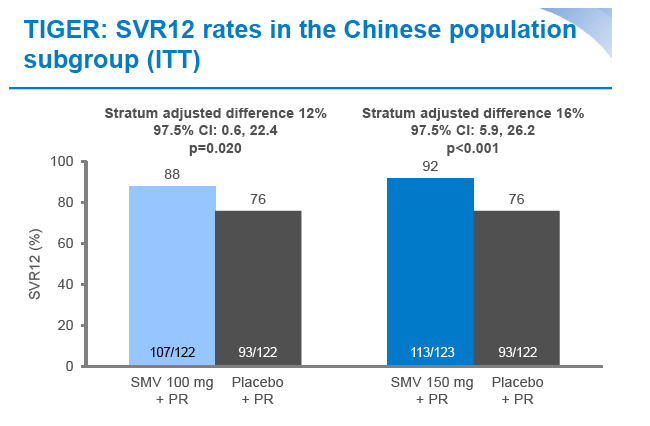
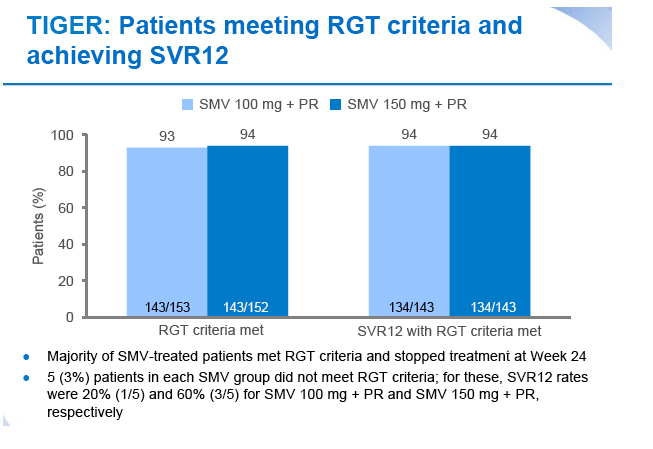
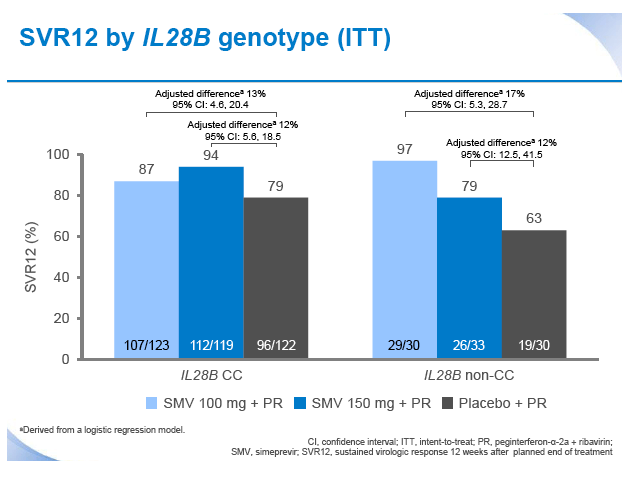
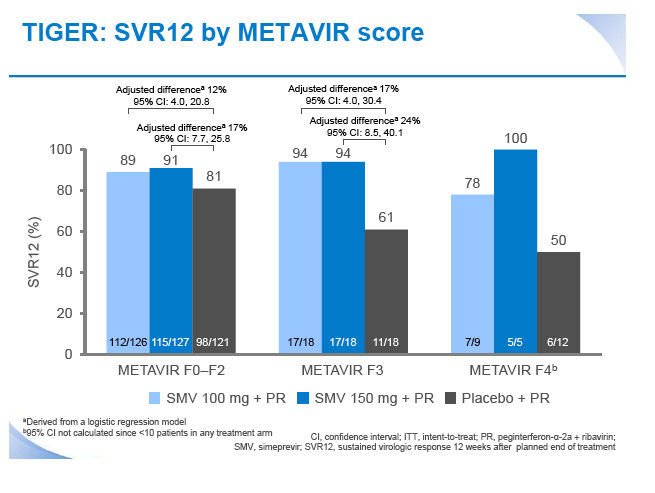
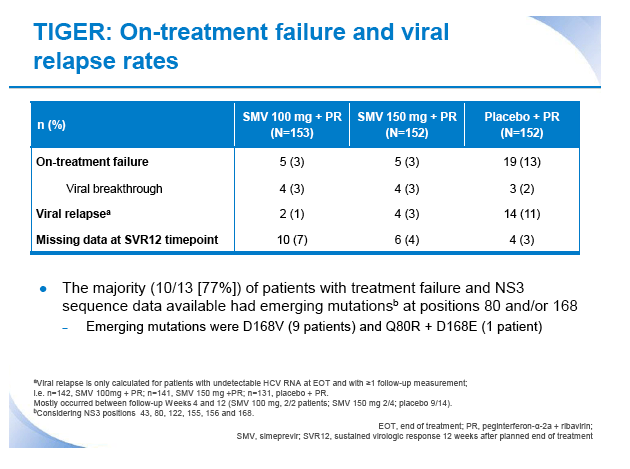
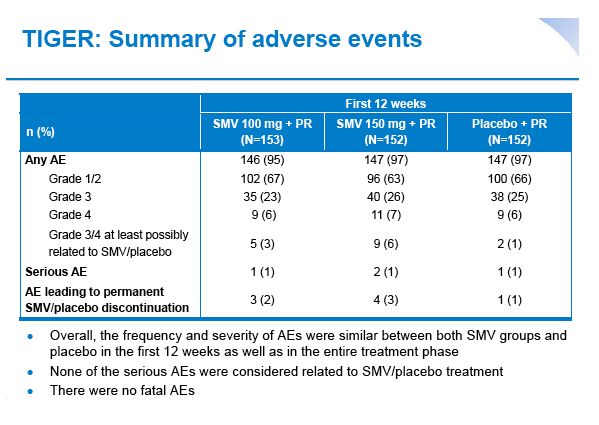
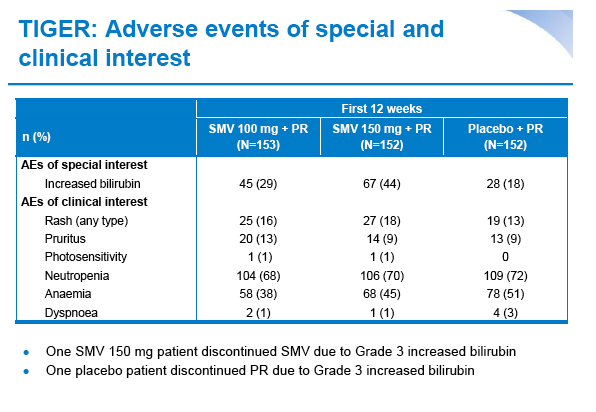
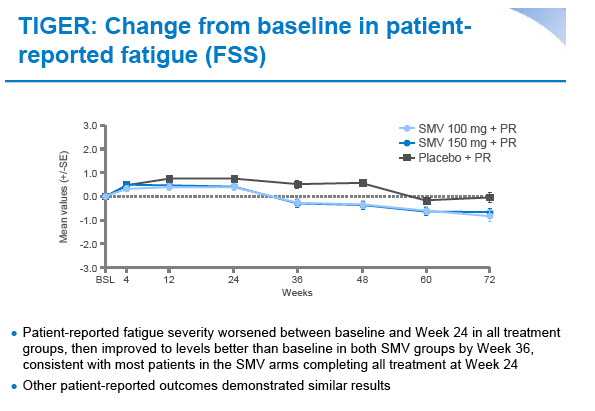
|
| |
|
 |
 |
|
|