 |
 |
 |
| |
Untimed Drug Levels & Resistance in Patients Experiencing Low-Level Viremia
|
| |
| |
CROI 2015 Feb 23-26, Seattle, WA
..... the study analyzed the association between untimed plasma drug levels at the first low-level viremia episode & the subsequent viral failure & to explore whether retrospective untamed plasma drug levels together with resistance data gives a better prediction of outcome than either measure alone & an association between retrospective untamed drug levels & the accumulation of resistant mutations.
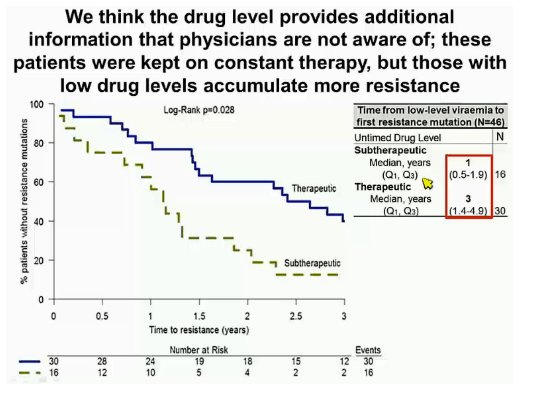
AUTHORS CONCLUDE - a single untamed plasma drug level independently predicts subsequent viral failure; low drug levels are more common and a better predictor than resistance data at low-level viremia; together, untamed drug plasma levels & resistance data can explain a higher proportion of treatment failures than either measure alone; resistance accumulates faster in patients with sub therapeutic drug levels; although retrospective these results could justify the potential investigation if untamed drug plasma levels in prospective management of low-level viremia episodes
Webcast: http://www.croiwebcasts.org/console/player/25759?mediaType=slideVideo&

program abstract
Background: HIV resistance testing in HAART patients experiencing low-level viremia (LLV, 50-999 HIV-RNA copies/mL) predicts the risk of subsequent virologic failure (VF, ≥1000 copies/ml). Suboptimal plasma drug levels can arise as a result of poor adherence and/or pharmacokinetic issues. Here, our aim was to evaluate whether retrospective analysis of plasma levels also provides insight into patient outcomes after experiencing LLV.
Methods: The first documented LLV episode of 2176 patients was analyzed. A total of 328 consenting patients with drug levels, genotypic resistance data and with follow-up clinical data while on constant therapy available were eligible. Untimed plasma drug levels (UDL) of PIs and NNRTIs in the sample corresponding to the first LLV episode were measured by HPLC-tandem MS. Drug levels were categorized as 'therapeutic' or 'suboptimal' based on target trough concentrations from DHHS guidelines. Resistance was assessed using the Stanford algorithm (GSS, corresponding to the number of 'active' drugs prescribed). Time to VF after LLV was evaluated by Kaplan-Meier analysis and Cox proportional hazards regression.
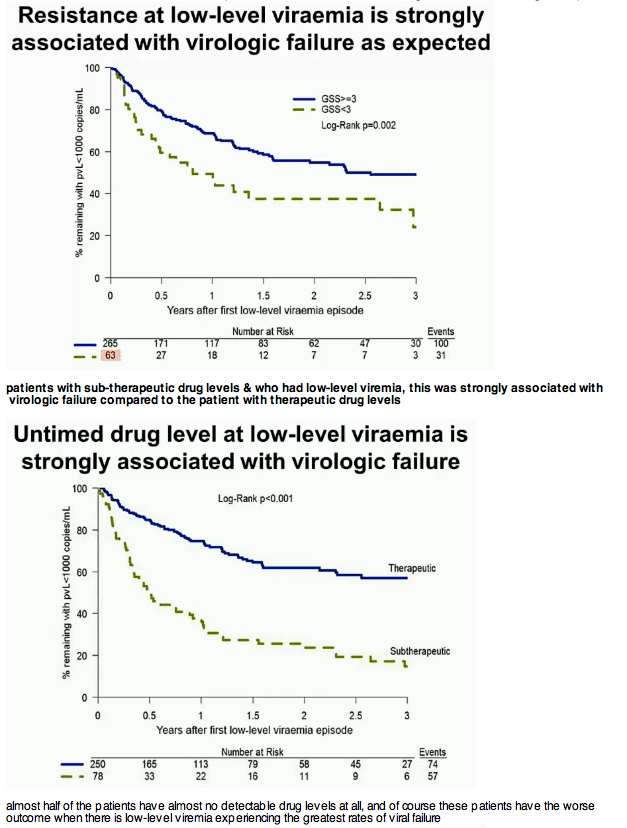
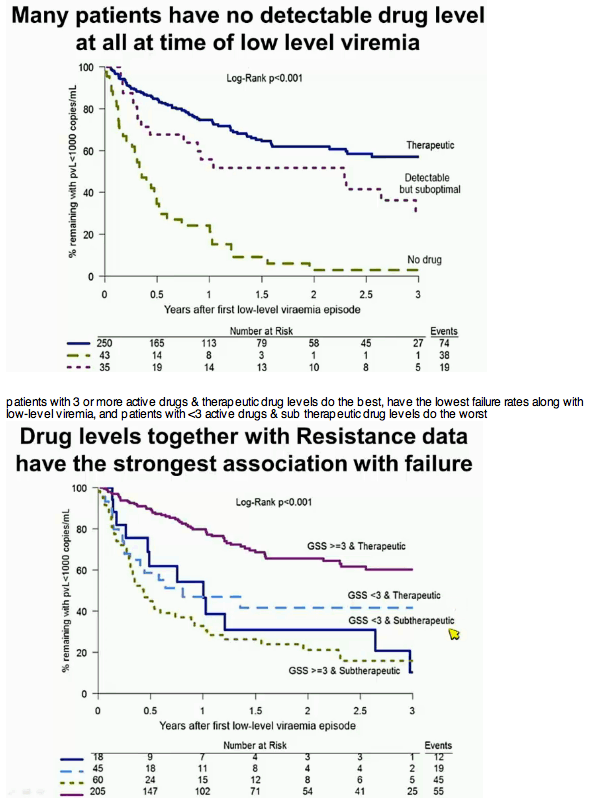
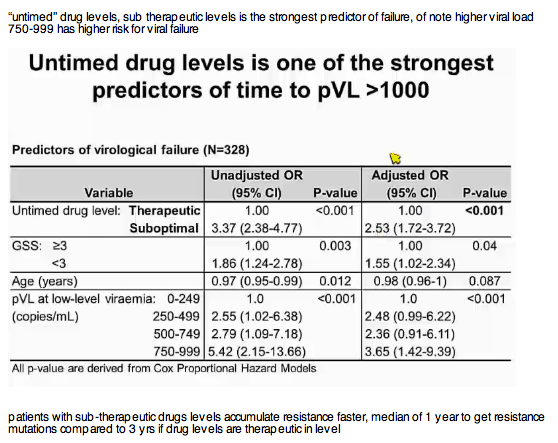
Results: 78 of 328 patients (24%) had suboptimal drug levels at LLV, compared with 63 (19%) having GSS1000. Within a year, 56/78 (72%) patients with suboptimal UDL had failed, compared to 45/63 (71%) with GSS In the adjusted multivariable model, variables associated with VF were suboptimal UDL (Adjusted Hazard Ratio, AHR 2.9, 95%CI 2.0-4.2, p
Conclusions: A single untimed drug level (UDL) and GSS independently predict subsequent VF, with suboptimal UDL having a greater hazard than GSS
|
| |
|
 |
 |
|
|