 |
 |
 |
| |
Fat and lean mass changes similar with raltegravir and PIs in ACTG analysis
|
| |
| |
CROI 2015, February 23-26, 2015, Seattle, Washington
webcast: http://www.croiwebcasts.org/console/player/25794?mediaType=slideVideo&
Mark Mascolini
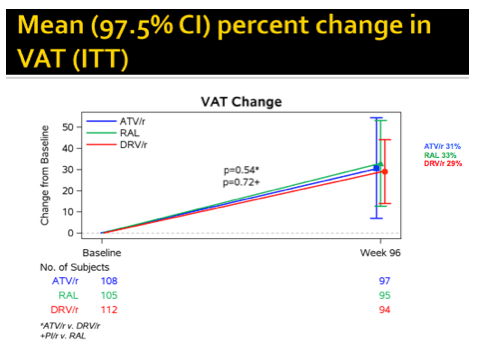
Changes in lean mass and fat gains did not differ through 96 weeks of treatment with raltegravir versus atazanavir or darunavir in AIDS Clinical Trials Group (ACTG) study A5260s [1]. People in all three study arms had significant 96-week gains in limb fat, subcutaneous adipose tissue (SAT), visceral adipose tissue (VAT), trunk fat, and lean mass.
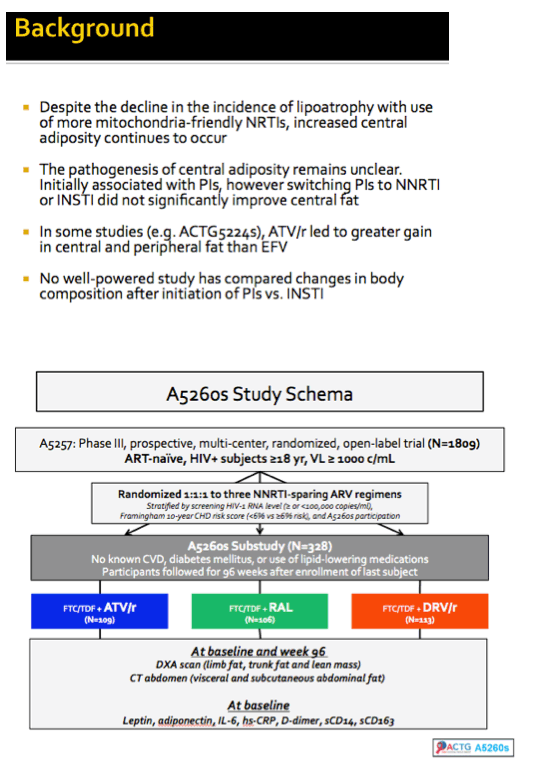
Although lipoatrophy has become uncommon with current antiretroviral regimens, the ACTG team noted, fat accumulation remains a problem. Whether today's protease inhibitor (PI) regimens lead to fat build-ups remains uncertain. Because no well-powered studies have compared changes in body composition between people starting therapy with a PI versus raltegravir, Grace McComsey and ACTG colleagues conducted this substudy of ACTG A5257 [2].
The substudy involved 328 antiretroviral-naive people without cardiovascular disease or diabetes. No one was taking lipid-lowering drugs. There was 106 people in the raltegravir arm, 109 in the atazanavir arm, and 113 in the darunavir arm. Before treatment began and at week 96, everyone had a DXA scan for limb fat, trunk fat, and lean mass, and everyone had abdominal CT for visceral and subcutaneous abdominal fat. The researchers also measured various markers of inflammation, coagulation, and immune activation at baseline and again at week 96.
Across the three study groups, median age stood at 36, 90% were men, and 44% were white. Median pretreatment CD4 count stood at 349 and median viral load at 4.55 log (about 35,000 copies). Median pretreatment body mass index measured about 25 kg/m2, the lower end of the overweight range.
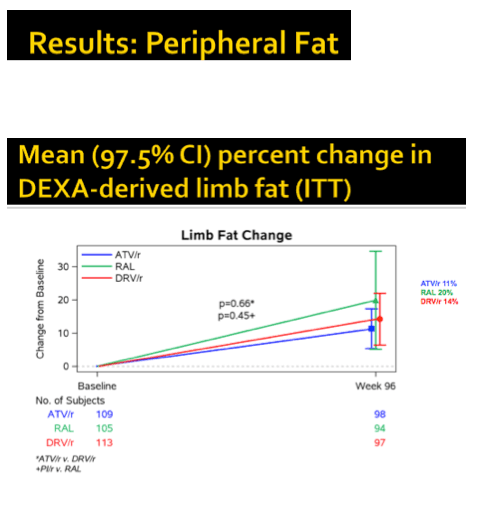
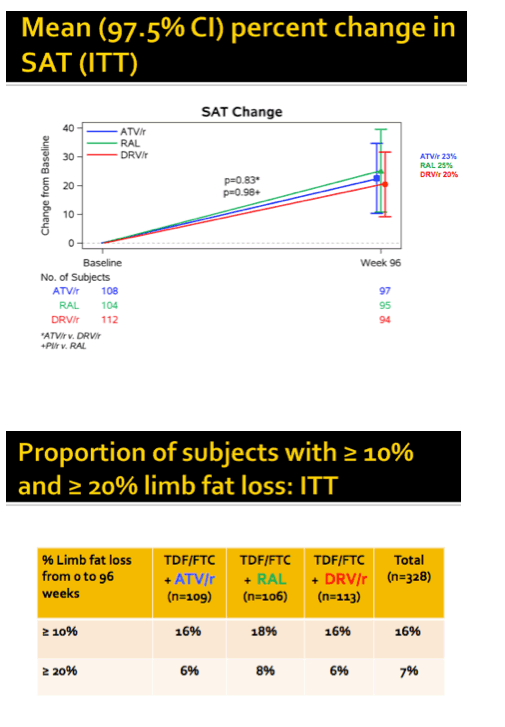
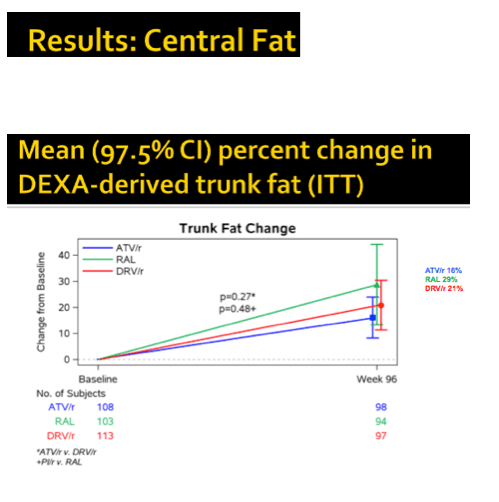
At study week 96, median limb fat, SAT, VAT, trunk fat, and lean mass increased significantly in all study arms (8.2%, 10.9%, 13.9%, 11.4%, and 1.3%, P < 0.001). No fat changes differed significantly between the combined PI arms and the raltegravir arm. Similar proportions of study participants lost 10% or more limb fat through 96 weeks--18% on raltegravir and 16% in each of the PI arms. Only 6% to 8% of participants lost more than 20% of limb fat through 96 weeks, with no big differences between study arms.
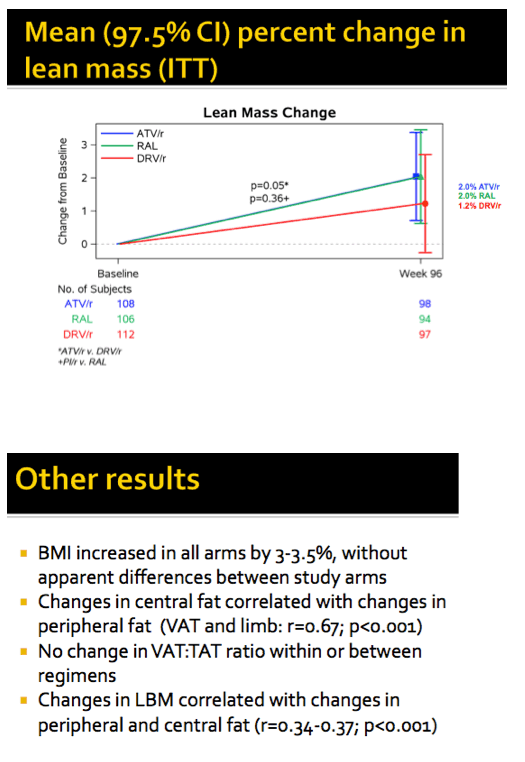
Lean mass rose more with atazanavir than with darunavir, 2.0% versus 1.2% (P = 0.05 but significance set at 0.025 in this study). People randomized to raltegravir also averaged a 2.0% gain in lean mass. The pooled PI group and the raltegravir group had similar 96-week changes in lean mass.
Body mass index climbed by 3% to 3.5% across study arms through 96 weeks, with no differences between arms. Changes in central fat correlated with changes in peripheral fat (r = 0.67, P < 0.001). Changes in lean body mass correlated with changes in peripheral and central fat (r = 0.34 to 0.37, P < 0.001).
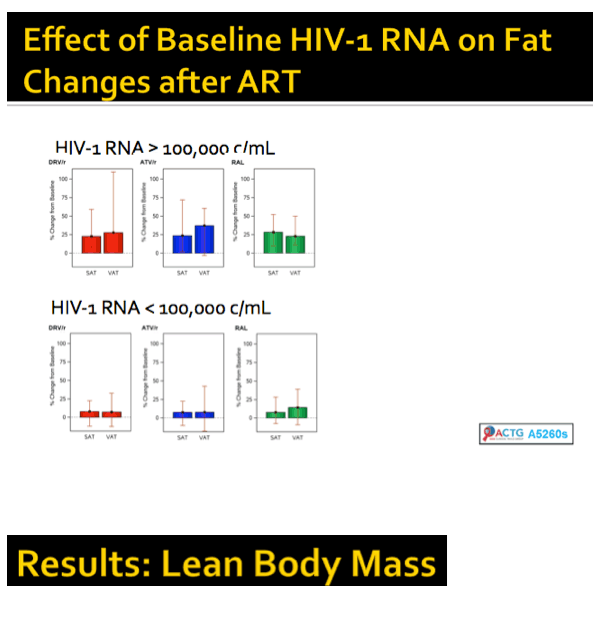
Higher levels of pretreatment inflammatory markers were linked to increased peripheral fat and lean body mass, but not central fat.
References
1. McComsey GA, Carlee Moser C, Currier JS, et al. Body composition changes after initiation of raltegravir or protease inhibitors. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 140.
2. Lennox JL, Landovitz RJ, Ribaudo HJ, et al. Efficacy and tolerability of 3 nonnucleoside reverse transcriptase inhibitor-sparing antiretroviral regimens for treatment-naive volunteers infected with HIV-1: a randomized, controlled equivalence trial. Ann Intern Med. 2014;161:461-471. http://www.ncbi.nlm.nih.gov/pubmed/25285539



|
| |
|
 |
 |
|
|