 |
 |
 |
| |
Real-World Assessment of Renal and Bone Safety Among Patients Exposed to Tenofovir Disoproxil Fumarate-Containing Single Tablet Regimens
|
| |
| |
Reported by Jules Levin
IDSA October 7-11, 2015, San Diego, CA, USA
Edwin DeJesus, MD, FIDSA;1 Joel Gallant, MD, MPH;2 Peter Shalit, MD, PhD;3 Huyen Cao, MD;4 Ya-Pei Liu, PhD;4 Joel Myers, MPharm;5 Lisa Rosenblatt, MD, MPH;5 Lingfeng Yang, PhD, MPH;6 Javier Szwarcberg, MD, MPH4
1Orlando Immunology Center, Orlando, FL, USA; 2Southwest CARE Center, Santa Fe, NM, USA; 3Tribal Med, Seattle, WA, USA; 4Gilead Sciences, Inc., Foster City, CA, USA; 5Bristol-Myers Squibb, Plainsboro, NJ, USA; 6Bristol-Myers Squibb, Hopewell, NJ, USA
-----------------------
from Jules: although TAF is about to be FDA approved & become available & this changes the discussion, this study presents interesting information & notions.
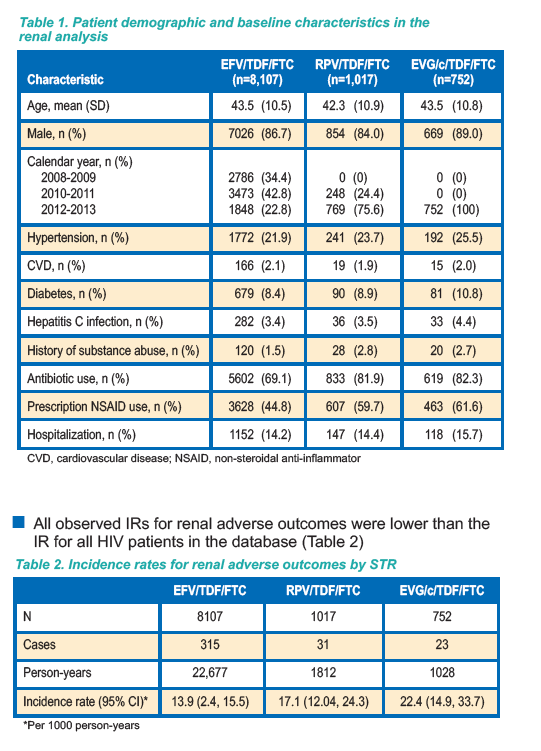
First, take a look in the database adverse events outcomes for ALL with HIV. "All observed IRs for renal adverse outcomes were lower than the
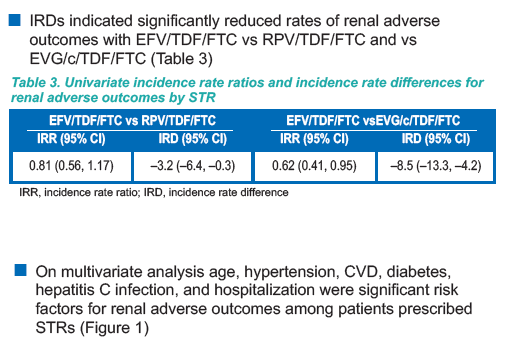
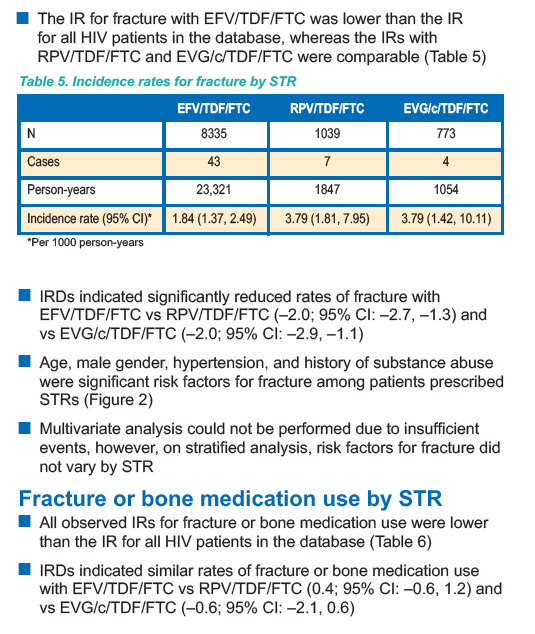
IR for all HIV patients in the database (Table 2)" reflecting that adverse renal outcomes were higher before ART. Fracture IR (incidence rates) was lower for EFV after ART started (Table 5) 1.84 vs 3.2, but IR was higher after ART started for RPV & EVG/c.
Renal and bone adverse outcomes for all HIV
patients in the database
- Renal adverse outcomes: 6964 cases/296,116 person-years,
IR=23.5 per 1000 person-years (95% CI: 23.0, 24.1)
- Fracture: 1209 cases/378,212 person-years, IR=3.2 per 1000-
person years (95% CI: 3.0, 3.4)
- Fracture or bone medication use: 2076 cases/356,508 personyears,
IR=5.8 per 1000 person-years (95% CI: 5.6, 6.1)
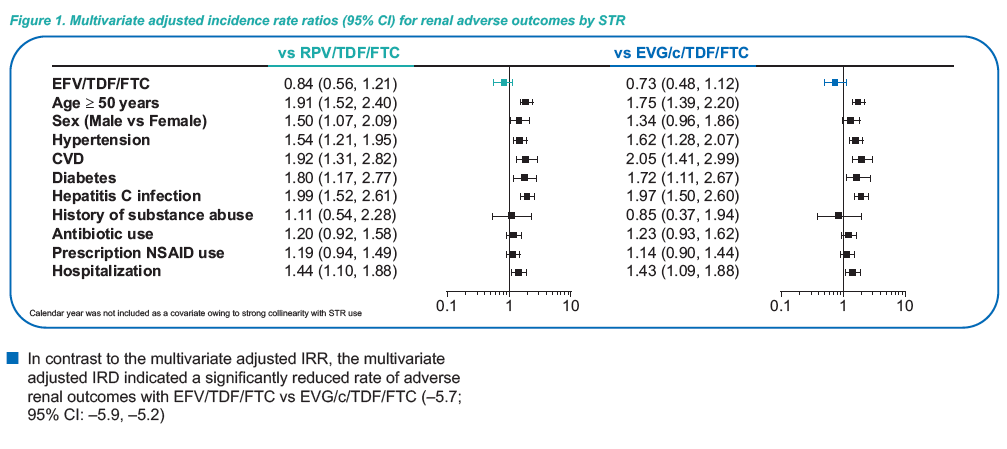
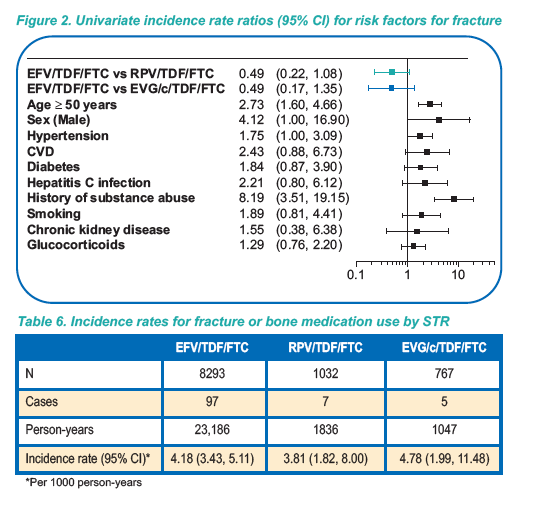
ALSO of note: On multivariate analysis age, hypertension, CVD, diabetes,
hepatitis C infection, and hospitalization were significant risk
factors for renal adverse outcomes among patients prescribed
STRs (Figure 1)
AND
Age, male gender, hypertension, and history of substance abuse
were significant risk factors for fracture among patients prescribed
STRs (Figure 2)
----------------






|
| |
|
 |
 |
|
|