 |
 |
 |
| |
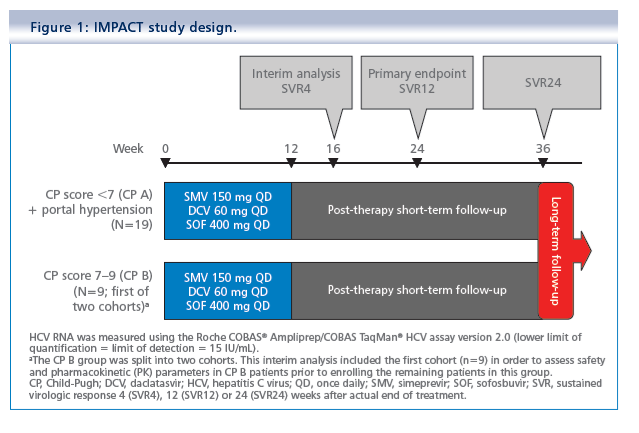
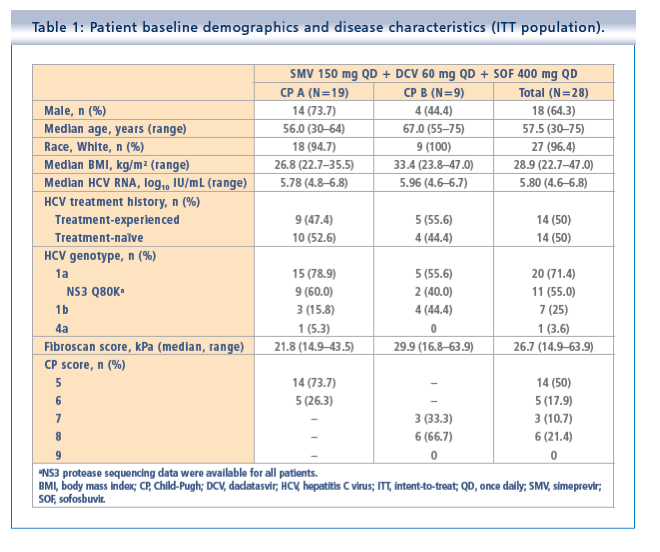
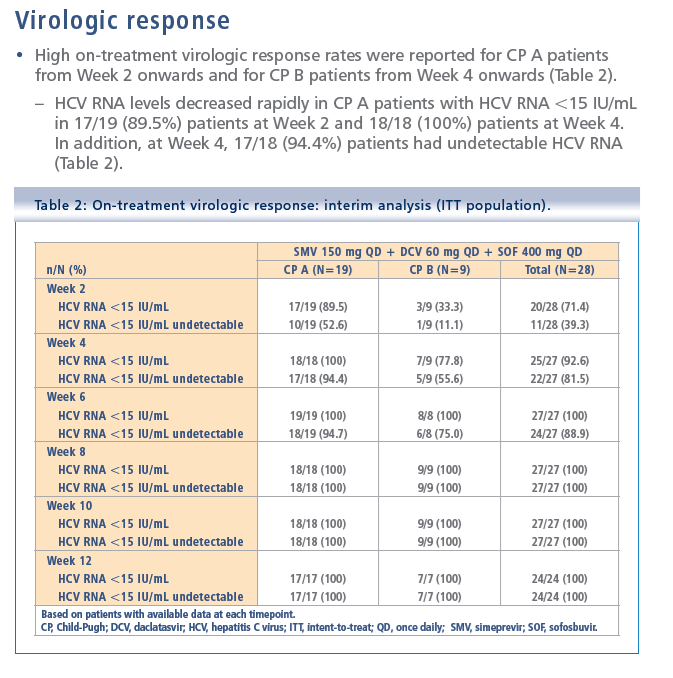
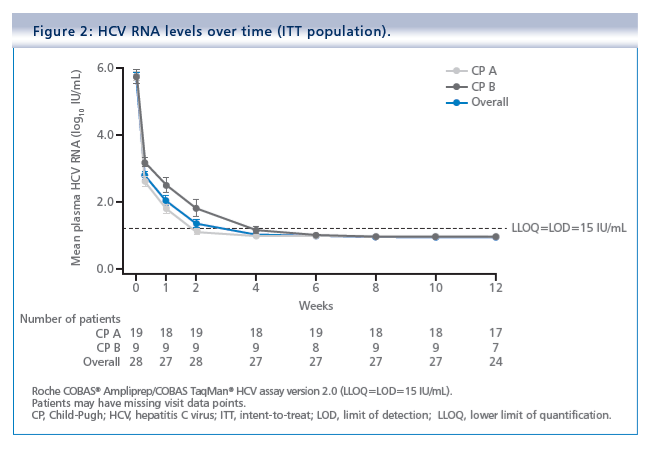
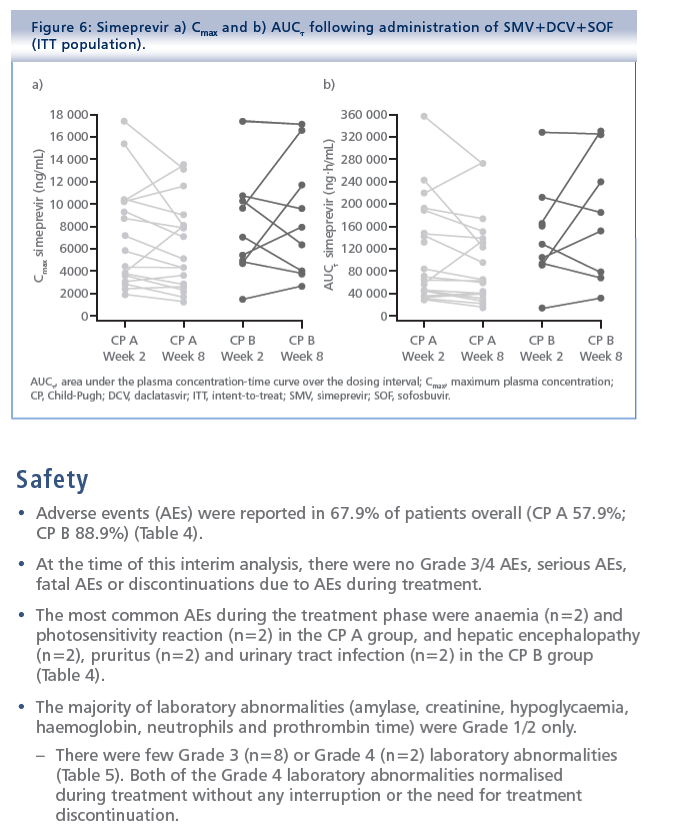
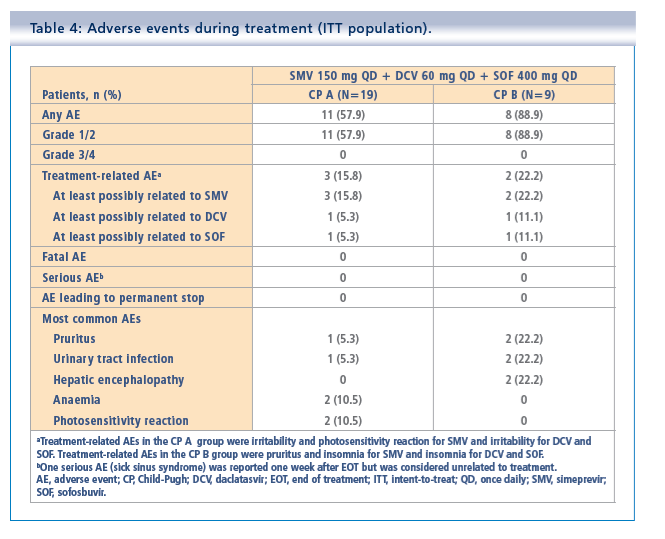
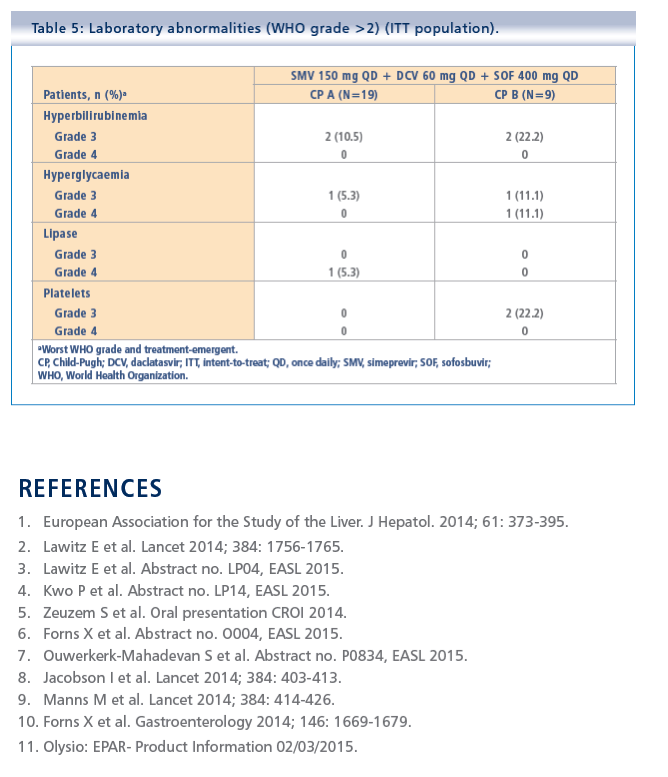
Simeprevir plus daclatasvir and sofosbuvir in treatment-naïve and treatment-experienced patients with chronic hepatitis C virus genotype 1 or 4 infection and decompensated liver disease: interim results from the Phase II IMPACT study
|
| |
| |
Reported by Jules Levin
EASL 2015 April 22-26 Vienna Austria
Lawitz E,1 Poordad F,1 Gutierrez J,1 Kakuda T,2 Picchio G,3 De La Rosa G,4 Beets G,5 Vandevoorde A,5 Van Remoortere P,2 Jacquemyn B,5 Quinn G,5 Luo D,2 Ouwerkerk-Mahadevan S,6 Vijgen L,5 Van Eygen V,5 Beumont M6
1Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, USA; 2Janssen Research & Development LLC, Titusville, NJ, USA; 3Infectious Diseases, Janssen Research & Development, San Francisco, CA, USA; 4Janssen Global Services, LLC, Titusville, NJ, USA; 5Janssen Infectious Diseases BVBA, Beerse, Belgium; 6Janssen Research & Development, Beerse, Belgium






|
| |
|
 |
 |
|
|