 |
 |
 |
| |
Daclatasvir Plus Sofosbuvir for Treatment of HCV Genotypes
1-4 in HIV-HCV Coinfection: The ALLY-2 Study
|
| |
| |
Reported by Jules Levin
EASL 2015 April 22-26 Vienna Austria
Wyles DL,1 Ruane P,2 Sulkowski MS,3 Dieterich D,4 Luetkemeyer AF,5 Morgan TR,6 Sherman KE,7 Liu Z,8 Noviello S,8 Ackerman P8
1University of California, San Diego, CA, USA; 2Ruane Medical and Liver Health Institute, Los Angeles, CA, USA; 3Johns Hopkins University School of Medicine, Baltimore, MD, USA; 4Icahn School of Medicine at Mount Sinai, New York, NY, USA; 5University of California and San Francisco General Hospital, San Francisco, CA, USA; 6VA Long Beach Healthcare System, Long Beach, CA, USA; 7University of Cincinnati College of Medicine, Cincinnati, OH, USA; 8Bristol-Myers Squibb, Princeton, NJ, USA.



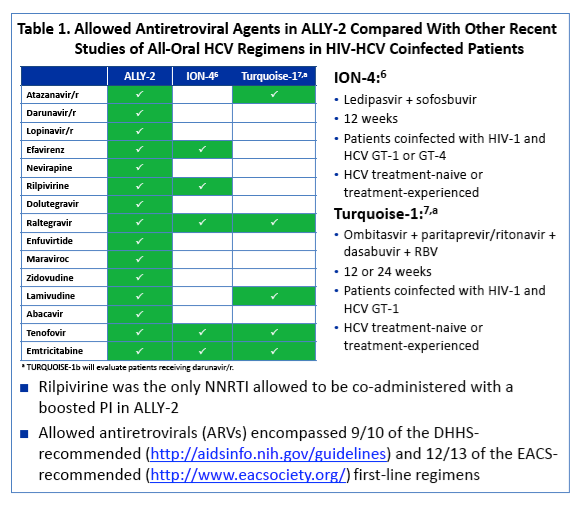
Allowed antiretrovirals (ARVs) encompassed 9/10 of the DHHS-recommended (http://aidsinfo.nih.gov/guidelines) and 12/13 of the
EACS-recommended (http://www.eacsociety.org/) first-line regimens
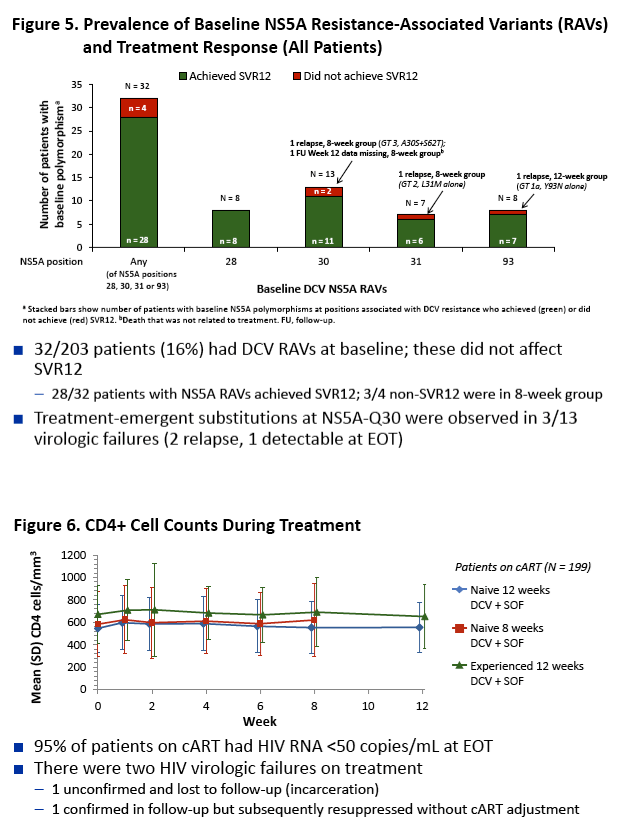
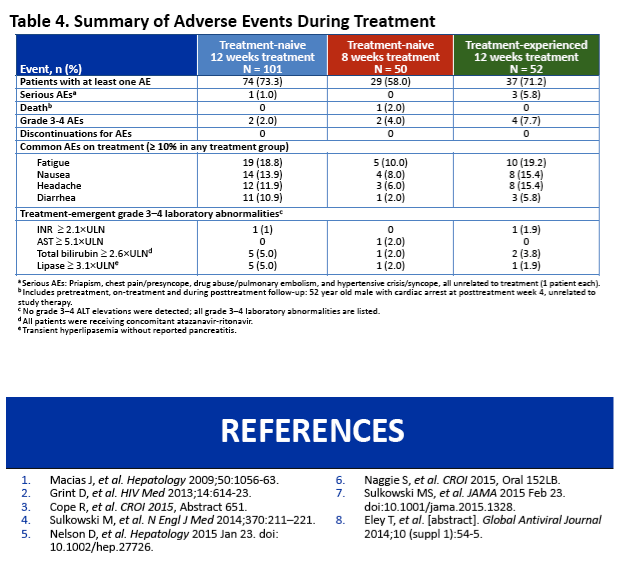
|
| |
|
 |
 |
|
|