 |
 |
 |
| |
VAC-3S, an Immunoprotective HIV Vaccine Directed to the 3S Motif of gp41, in Patients Receiving ART: Safety, Dose & Immunization Schedule Assessment
|
| |
| |
Download the PDF here
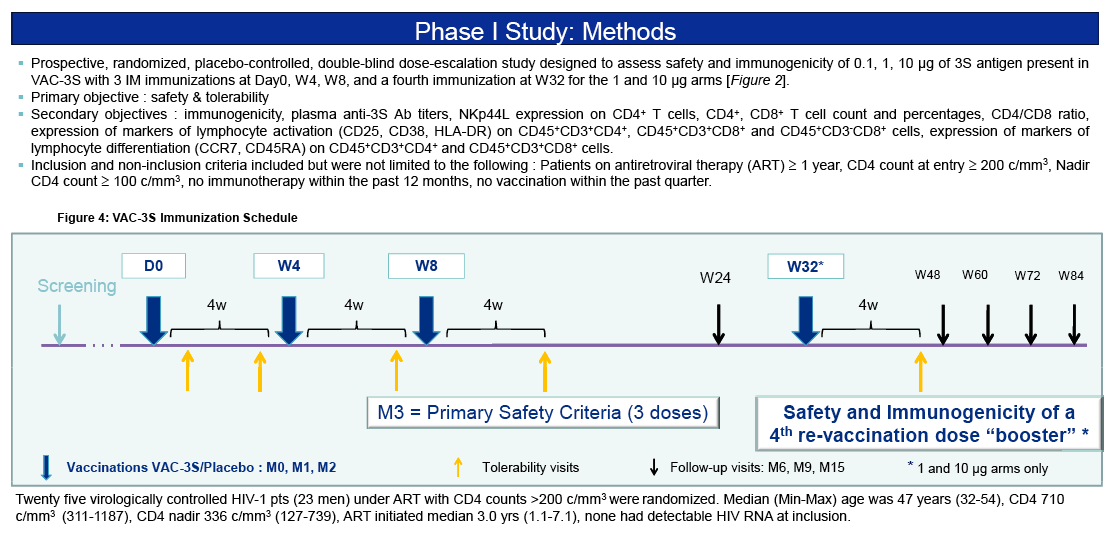
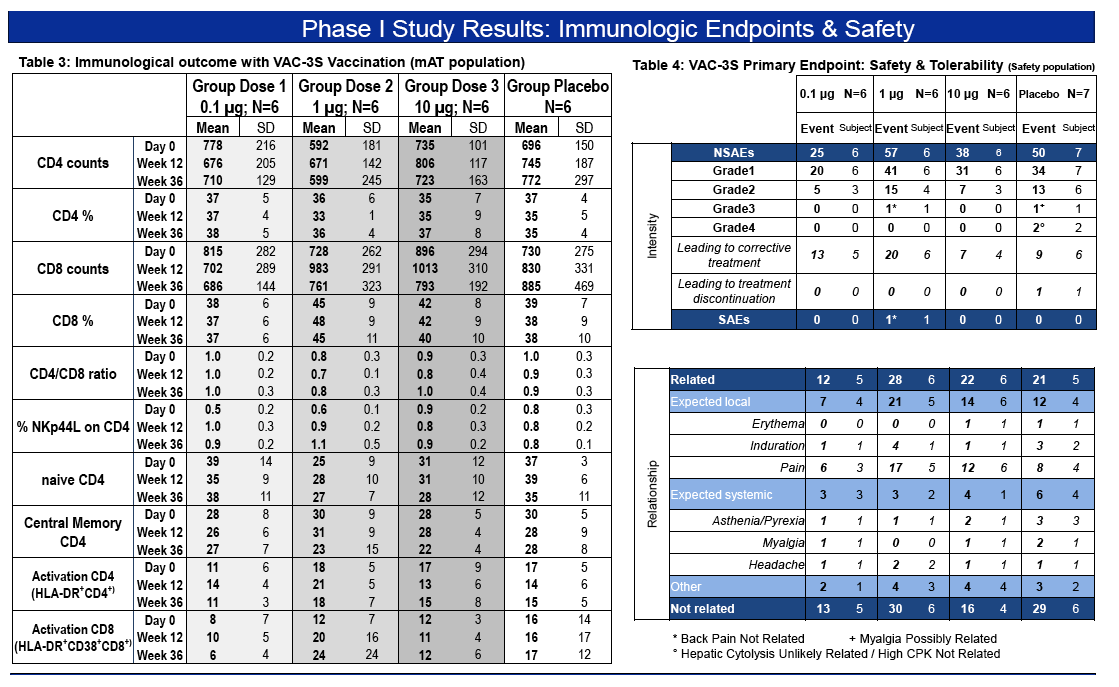
Primary objective : safety & tolerability
Twenty five virologically controlled HIV-1 pts (23 men) under ART with CD4 counts >200 c/mm3were randomized. Median (Min-Max) age was 47 years (32-54), CD4 710 c/mm3 (311-1187), CD4 nadir 336 c/mm3 (127-739), ART initiated median 3.0 yrs (1.1-7.1), none had detectable HIV RNA at inclusion.
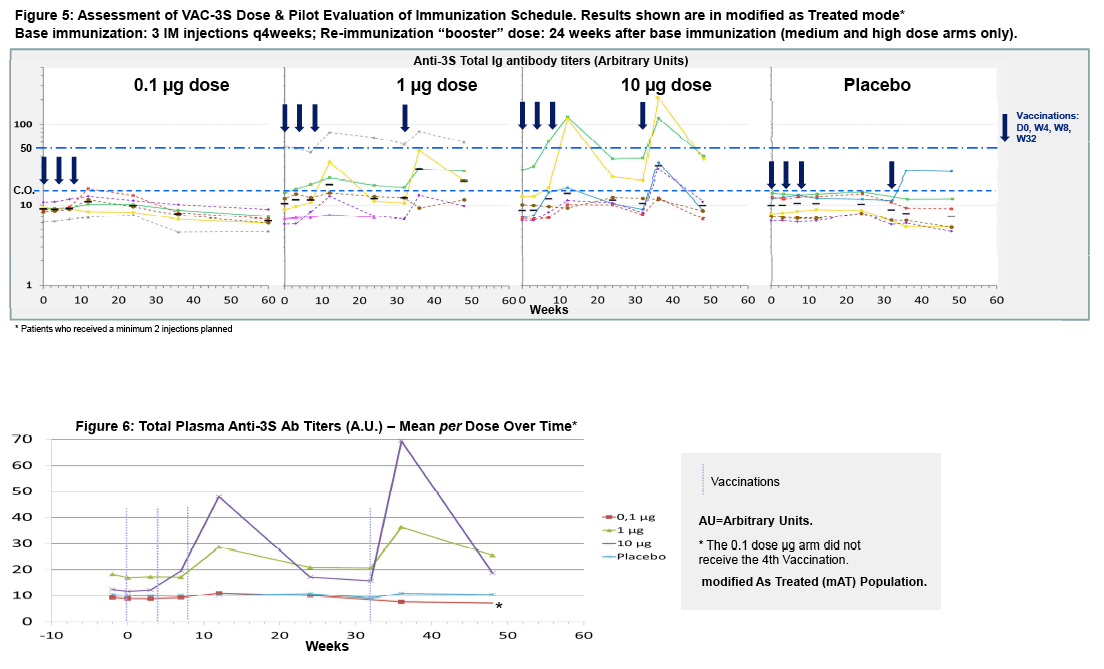
Prospective, randomized, placebo-controlled, double-blind dose-escalation study designed to assess safety and immunogenicity of 0.1, 1, 10 μg of 3S antigen present in VAC-3S with 3 IM immunizations at Day0, W4, W8, and a fourth immunization at W32 for the 1 and 10 μg arms [Figure 2].
- Primary objective : safety & tolerability
- Secondary objectives : immunogenicity, plasma anti-3S Ab titers, NKp44L
expression on CD4+ T cells, CD4+, CD8+ T cell count and percentages, CD4/CD8 ratio, expression of markers of lymphocyte activation (CD25, CD38, HLA-DR) on CD45+CD3+CD4+, CD45+CD3+CD8+ and CD45+CD3-CD8+ cells, expression of markers of lymphocyte differentiation (CCR7, CD45RA) on CD45+CD3+CD4+ and CD45+CD3+CD8+ cells.
Reported by Jules Levin

S Gharakhanian1ab, Ch Katlama2,5, O Launay3, H Bodilis3, R Calin2, R Ho Tsong Fang1a, M Marcu1a, B Autran4-5, V Vieillard4-5, J Crouzet1a, P Debre4-5
1aInnaVirVax, Genopole, Evry, France, 1bCambridge Innovation Center, Cambridge MA, USA, 2Inserm U943, AP-HP Pitie Salpetriere Hospital, 3AP-HP Cochin Hospital & Inserm CIC BT505,
4 Inserm UMR-S 945, 5UPMC: Universite Pierre et Marie Curie, Paris, France.


|
| |
|
 |
 |
|
|