 |
 |
 |
| |
Characteristics and Oral PrEP Adherence in the
TDF2 Open-Label Extension in Botswana
|
| |
| |
Reported by Jules Levin
IAS 2015 Vancouver
Henderson FL1, Taylor AW1, Chirwa LI2,Williams TS1,3, Borkowf CB1, Kasonde M2, Mutanhaurwa R2, Matlhaba O2, Hageman K1, Casillas P2, Samandari T1
1Centers for Disease Control and Prevention, Atlanta, GA
2Centers for Disease Control and Prevention, Gaborone and Francistown, Botswana
3ICF Macro, Atlanta, GA
Webcast: https://www.youtube.com/watch?v=JjLzaovB7Pc

·One potential consideration in the interpretation of these findings is that the TDF2 OLE study population participated in previous clinical trial where they given adherence and risk reduction counselling which could have affected their responses and behavior during the OLE
·Since this represents a self-selected highly-motivated population, our findings may not be representative of the general population
·And we have not yet completed drug level testing for all of our study participants but....

·Our next steps include performing intracellular drug level testing using dried blood spots to give us a better look at consistency of adherence over time.
·Additional next steps include an analysis of a qualitative substudy that was conducted at the end of OLE that will help us to understand barriers and facilitators of adherence and visit attendance.
·And we will continue to investigate the differences in adherence between men and women.

·The TDF2 study was the predecessor for the open label extension and was a randomized, double-blind, placebo-controlled clinical trial that enrolled 1219 HIV-negative heterosexual young adults in Botswana.
·Participants were randomized to a once-daily combination pill containing tenofovir and emtricitibine or placebo.
·Since 62% protective efficacy was demonstrated in TDF2, we provided open label drug to TDF2 participants for 12 months in order to continue to assess safety, effectiveness, behavior risk compensation and adherence to PrEP.

·OLE screening started in February 2013 and ended in May 2013.
·After eligibility was determined and participants started drug, they attended monthly follow-up visits.
·At each visit, participants were dispensed a 30-day supply of drug and adherence counselling was provided.
·Dual rapid HIV testing was conducted with an antigen/antibody combo test, pregnancy test was performed, and condoms were dispensed.
·Quarterly visits included additional HIV testing with an HIV antigen/antibody combo Elisa along with safety labs. Risk reduction counselling was performed.
·Our retention strategies during the study included SMS text messaging and phone calls to remind participants of their appointments.
·No reimbursement was provided at any point in order to simulate more of a real-world setting.
Reference as needed:
·Study inclusion criteria included former TDF2 participants, ability to provide informed consent, normal serum creatinine/phosphorus levels and if female, willing to use hormonal contraceptive.
·Exclusion criteria included HIV infection, pregnant or breastfeeding and any other clinical conditions that made the participant unsuitable for enrollment.

·After the study was complete, we measured Tenofovir levels using dried blood spots which were collected monthly.
·We selected 120 participants and classified them to 4 groups with different time points in order to create a longitudinal view of adherence over the length of the study.
·This table shows the study month grouping.
·Group 1,2, and 3 contain participants who attended study visits throughout the length of the study.
·For Group 4, we selected participants who had 1 or more missed visits and test blood spots around the time of any missed visits.
Reference notes if needed:
Linear range for this assay was 0.01ng/ml to 2000ng/ml.
The limit of quantification (LOQ) was 15 ng/ml.
461 time points were tested of the 500. 39 samples were not found/completed.

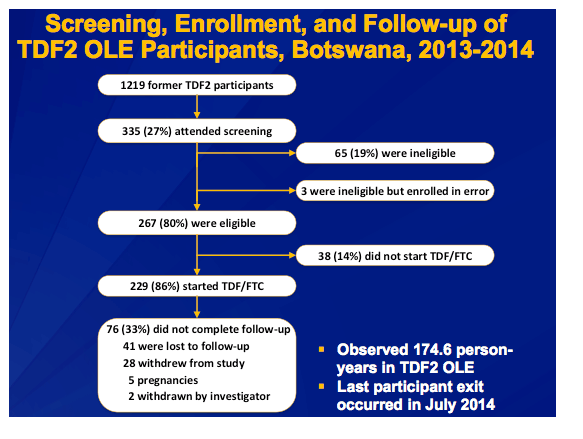
·Of the former 1219 TDF2 participants, 27% came to the clinics and attended screening. This was experienced because Botswana has a highly mobile population, and up to 4 years had elapsed between many participants� exit from TDF2 and recruitment into OLE. We found that many former participants� contact information were no longer current but we ran newspaper and radio ads to attempt to address this issue.
·Once participants returned and were assessed for eligibility, 86% of those identified as eligible started TDF/FTC.
·33% did not complete follow-up and the majority of study participants reported the reason as being due to work commitments. We suspect that the monthly study visits contributed to decreased study visit adherence since approx. 1/2 day was taken away from a work day each month to come to the clinic.
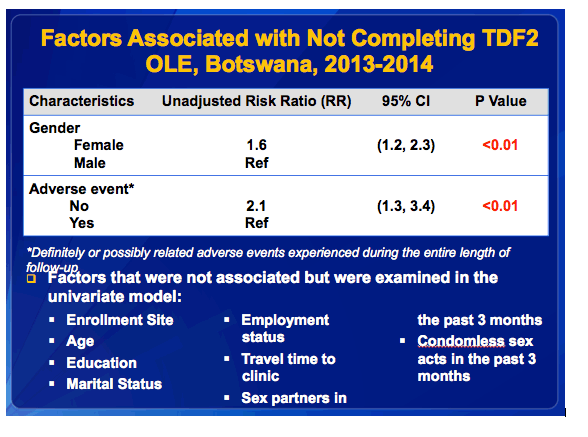
·Given this attrition experienced, we looked at factors that could be associated with not completing the study.
·In an univariate analysis, we looked at baseline characteristics except for adverse events which was defined as a definitely or possibility related AE experienced at any point during the length of the study.
·We saw that gender and adverse events were associated with not completing OLE.
·Female were 60% more likely than males to not complete the study.
·If participants did not experience an AE, then they were 2x more likely to not complete the study than those who did experience an AE. It�s possible that this observed difference could be related to more intensive follow-up efforts aimed at ensuring participant safety among those with an AE.
·The factors listed below were not associated with completing OLE but were explored in the univariate models.
·The next couple of slides are going to look at behavioral risk reported by participants over the course of the study.
·A total of 10 severe adverse events involving 8 participants were reported during the study which were judged to be at least possibly related to study drug. All resolved. One grade 1 hypercreatininemia. Only 2 / 229 were permanently discontinued due to lab. 2 participants discontinued drug due to recurrent grade 3 hypophosphatemia.
·Clarify that this is only for definitely and possibly related AEs (n=64 participants, 144 obs).
·Though we don�t yet have a definitive explanation for this observation, we can hypothesize that perhaps
those who stayed in the study for longer might have had more time on drug which is associated with the possibility of experiencing an AE.
·For the following 2 models, we used a Poisson regression and accounted for repeated measures within each subject.
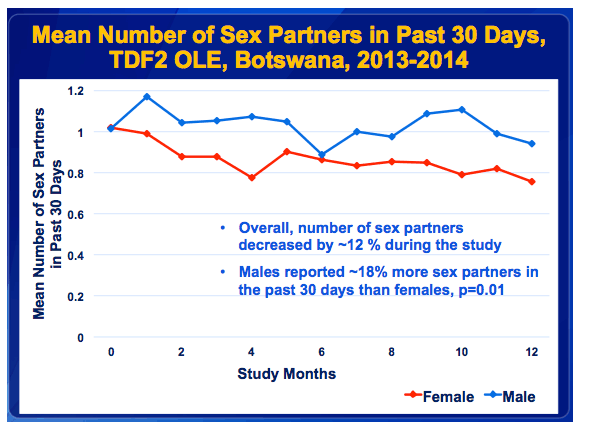
·This graph shows the mean number of sex partners reported in the past 30 days over the length of the study, by gender.
·We observed that the number of sex partners decreased by approximately 12% during the study and males reported approximately 18% more sex partners in the past 30 days than females.
Number of participants who had 0 partners during the entire study: 12
Number of participants who reported 0 partners at any point during the study: 103
·We modeled our outcome variables as count data using Poisson regression.
·We used generalized estimating equations (GEE) to account for correlations among responses (i.e., repeated measures within subject).
·This method allows us to model both the change in behavior over time as well as compare the reported behavior between males and females.
·The outcome measure is the estimated risk ratio (RR) which estimates the risk of reporting the behavior in one group (or at a given time) relative to another group (or another time).
Number of sexual partners:
Mean sex partners in past 30 days: 1.5 acts
Female: 0.87 partners
Male: 1.03 partners
Decreased ~12% over the course of the study: (RR=0.88; 95% CI: 0.78, .99; .0358)
Males report ~18% more partners in the past 30 days than females (RR=1.18; 95% CI: 1.04, 1.34; p=.0123)
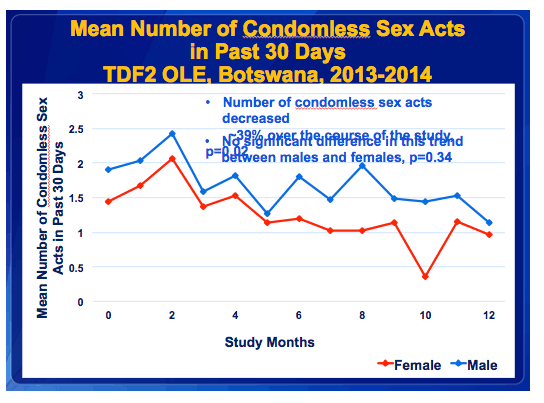
·This graph shows the mean number of condomless sex acts reported in the past 30 days over time.
·The overall number of condomless sex acts decreased ~39% over the course of the study.
·There was no significance in this trend between males and females.
·Our next slides explore drug adherence, using drug levels and self reported adherence.
Condomless sex acts in the past 30 days:
Female: Mean 1.3 acts
Male: Mean 1.7 acts
Number of condomless sex acts decreased ~39% over the course of the study
RR=0.61; 95% CI: 0.40, 0.92
p=.0177
No significant difference between males and females
RR=1.3; 95% CI: 0.74, 2.4
p=.3433
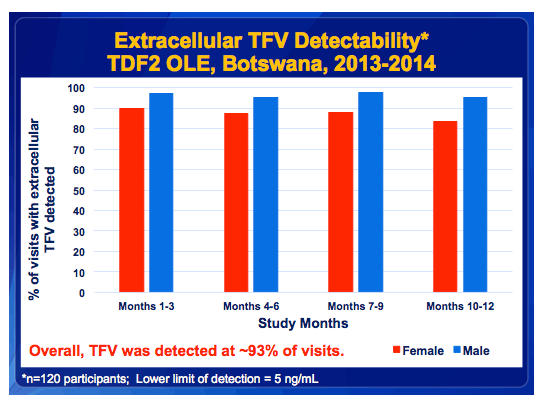
·As discussed in the methods, we sampled 120 participants at various time points in the study.
·Using dried blood spots collected at monthly visits, we measured the extracellular tenofovir levels.
·We found that, overall, participants had detectable tenofovir at approximately 93% of visits.
·Looking at detectability by gender: 87% of women had detectable drug over time and 96% of men had detectable drug over time.
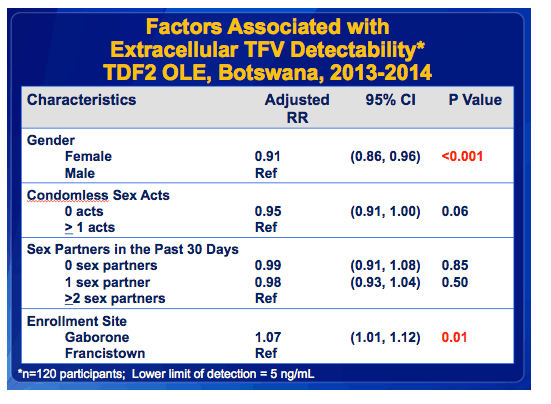
·In a multivariable model accounting for repeated measures for each time point, women were slightly less likely to have detectable drug compared to men
·In addition, participants in Gaborone were slightly more likely to have detectable drug than those in Francistown.
·Reported condomless sex acts and number of partners were not associated with drug detectability in this model.
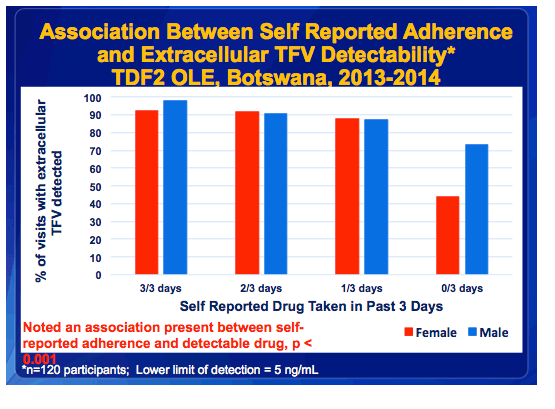
·Next, we looked at drug detectability compared to the participants 3-day self report.
·We observed an association between 3-day self reported adherence and drug detectability.
·At least 85% detectability was observed when at least 1 dose was reported in the last 3 days.
·Among those who reported taking no doses in the last 3 days, 60% had detectable drug.
·P value from GENMOD procedures.
·Among women reporting having taken 3, 2, 1 or 0 doses in the past 3 days, 92%, 92%, 88%, and 44% had detectable drug, respectively.
·Among men reporting having taken 3, 2, 1, or 0 doses in the past 3 days, 98%, 91%, 87%, and 73% had detectable drug, respectively.
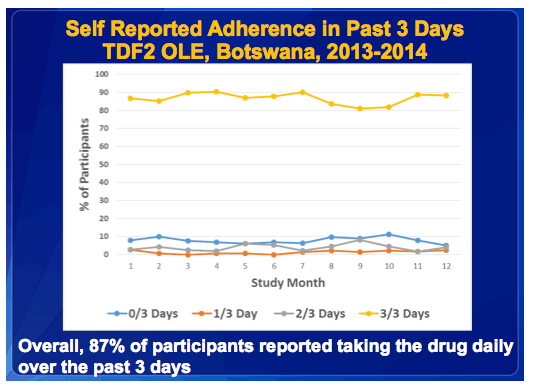
·And now, looking at the entire population of OLE participants, this slide shows the self-reported adherence and overall, 87% of participants reported taking the drug daily over the past 3 days.

And finally, we observed no HIV infections among participants after initiation of TDF/FTC.
Based on the incidence experienced during the TDF2 clinical trial reported in the methods, we would have expected 5-6 cases of HIV infection to occur in OLE.
·During the TDF2 study, the incidence of HIV was estimated to be 3.1 cases per 100 person years in the placebo group.

|
| |
|
 |
 |
|
|