 |
 |
 |
| |
Sofosbuvir-Based Regimens for Patients with Chronic Hepatitis C Genotype 3 Infection:
Integrated Analysis of Five Clinical Studies
|
| |
| |
Reported by Jules Levin
AASLD 2016 Nov 11-15 Boston, MA
GR Foster1, S Zeuzem2, E Gane3, C Stedman4, JJ Feld5, A Mangia6, K Agarwal7, M Swain8, HM Mir9, PJF Troke10, J Llewellyn11, M Natha11, B Kreter11, J Zhang11, J McNally11, DM Brainard11, S Strasser12, S Pianko13
1Queen Mary University, London, United Kingdom; 2Johann Wolfgang Goethe University Medical Center, Frankfurt Germany; 3Auckland Clinical Studies, Auckland, New Zealand; 4Christchurch Hospital, Christchurch, New Zealand;
5Toronto Western Hospital, Toronto, Canada; 6Casa Sollievo della Sofferenza Hospital, San Giovanni Rotondo, Italy; 7Institute of Liver Studies, King's College Hospital, London, United Kingdom; 8University of Calgary, Calgary, Canada; 9Gilead Sciences, Melbourne, Australia;
10Gilead Sciences, London, United Kingdom; 11Gilead Sciences, Foster City, United States; 12Royal Prince Alfred Hospital, Sydney, Australia; 13Monash Health, Melbourne, Australia
AASLD: A Randomized, Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks and Sofosbuvir/Velpatasvir for 12 Weeks for Patients with Genotype 3 HCV Infection and Cirrhosis: The POLARIS-3 Study - (11/15/16)
AASLD: A Randomized Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks Compared to Sofosbuvir/Velpatasvir for 12 Weeks in DAA-Naïve Genotype 1-6 HCV Infected Patients: The POLARIS-2 Study - (11/15/16)
AASLD: Sofosbuvir/Velpatasvir/Voxilaprevir for 12 Weeks as a Salvage Regimen in NS5A Inhibitor-Experienced Patients With Genotype 1-6 Infection: The Phase 3 POLARIS-1 Study - (11/15/16)
AASLD: A Randomized, Controlled, Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir or Sofosbuvir/Velpatasvir for 12 Weeks in Direct-Acting Antiviral-Experienced Patients With Genotype 1-6 HCV Infection: The POLARIS-4 Study - (11/14/16)
AASLD: Integrated Resistance Analyses of HCV-Infected Patients Treated With Sofosbuvir, Velpatasvir, and Voxilaprevir for 8 and 12 Weeks From Phase 2 Studies - (11/14/16)



References
1. AASLD/IDSA/IAS-USA. Recommendations for testing, managing, and treating hepatitis C. www.hcvguidelines.org. Accessed on 20 March 2016.
2. EASL Recommendations on Treatment of Hepatitis C 2015. Journal of Hepatology. 2015;63:199-236.
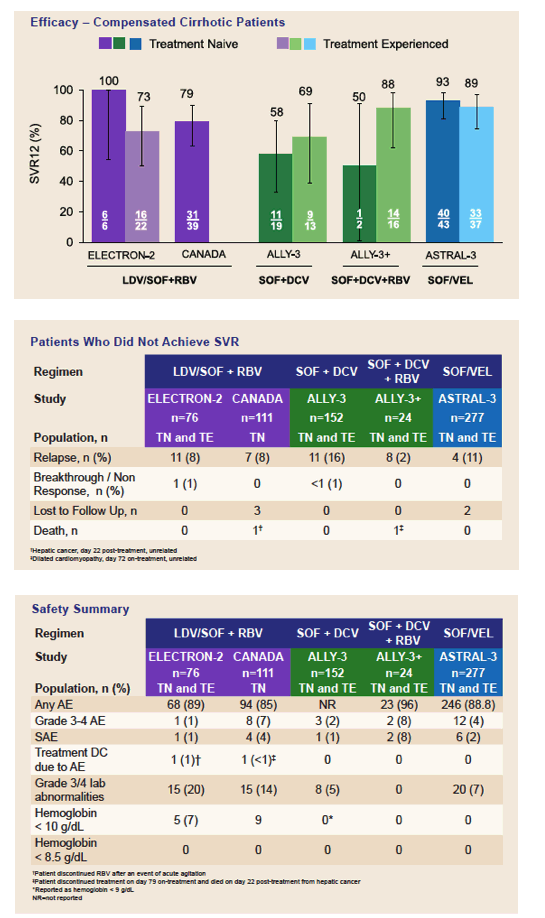
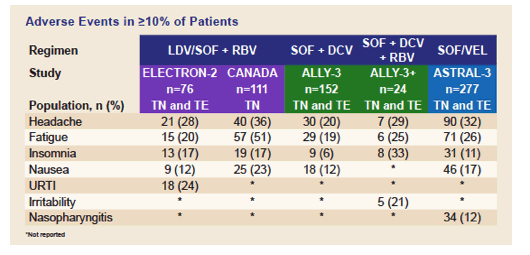
3. Gane et al. Efficacy of Ledipasvir and Sofosbuvir, With or Without Ribavirin, for 12 Weeks in Patients With HCV Genotype 3 or 6 Infection. Gastroenterology 2015;149:1454-1461.
4. Feld et al. Ledipasvir/Sofosbuvir with Ribavirin for 12 Weeks Is Effective and Safe in Treatment-Naïve Genotype-3 HCV-Infected Patients in Canada. European Association for the Study of the Liver. 2016, Barcelona, Spain. Poster SAT-183.
5. Nelson et al. All-Oral 12-Week Treatment With Daclatasvir Plus Sofosbuvir in Patients With Hepatitis C Virus Genotype 3 Infection: ALLY-3 Phase III Study. Hepatology 2015; 61:1127-1135.
6. Leroy et al. Daclatasvir, Sofosbuvir, and Ribavirin for Hepatitis C Virus Genotype 3 and Advanced Liver Disease: A Randomized Phase III Study (ALLY-3+). Hepatology 2016;63:1430-1441.
7. Foster et al. Sofosbuvir and Velpatasvir for HCV Genotype 2 and 3 Infection. New England Journal of Medicine 2015;373(27):2608-17.
|
| |
|
 |
 |
|
|