 |
 |
 |
| |
Clinical Pharmacology at CROI 2016: TAF, PrEP- TDF-Bone, TAF (vaginal, rectal), Maraviroc, Dapivirine Vaginal Ring, Long-Acting ARTs, Peds
|
| |
| |
....TAF...
TDF & PrEP-Bone....
- Long-Acting Cabotegravir/rilpivirine LATTE2)....
- Long-Acting new NRTI MK8591.....
PrEP:
- dapivirine (DPV) vaginal ring, Maraviroc.....
- TAF in rectal tissues and female genital tract tissues......
PEDS -
- Dolutegravir/children 6-12 years of age.....
- EVG/c/FTC and TAF adolescents aged 12-17.....
- NVP dosing for very early ARV therapy in newborns
Courtney V. Fletcher, Pharm.D.
Dean and Professor
College of Pharmacy
University of Nebraska Medical Center
986000 Nebraska Medical Center
Omaha, NE 68198
The 2016 (23rd) Conference on Retroviruses and Opportunistic Infections (CROI) was held in Boston, MA, from February 23-25, 2016. CROI is the premier HIV-focused scientific meeting. In this report I will highlight abstracts focused on pharmacologic issues that are of broad interest or might benefit from some expert clarification. Abstracts will be discussed in the categories of: (i) the therapy of HIV infection with existing and new drugs, (ii) PrEP and PK in tissues, and (iii) drug development in pediatrics. You can find more information on these abstracts on the CROI website and many are covered in depth elsewhere on the NATAP http://natap.org website.
I. The Pharmacotherapy of HIV Infection
a. A switch from TDF/FTC to TAF/FTC maintains HIV suppression and is associated with fewer adverse renal and bone effects (abstract 29).
Tenofovir alafenamide (TAF) is approved by the FDA in combination tablet with FTC, EVG and COBI, and with FTC and RPV. A TAF/FTC combination tablet has been submitted for FDA review and a decision is expected quite soon. 663 virologically suppressed persons receiving TDF/FTC (and a third agent) were randomized to remain on that regimen or to switch to TAF with FTC and the same third agent. At 48 weeks, the proportion of participants with HIV-RNA < 50 copies/ml was 93% for TDF/FTC and was 94.3% for TAF/FTC.
Important differences were seen in rates of tenofovir (TFV)-associated adverse effects. In the TAF recipients but not in those who continued on TDF: (1) measures of proteinuria (urine protein, albumin, beta-2-microglobulin) improved; (2) estimated glomerular filtration rate (eGFR) improved by 8.4 mL/min (vs 2.8 mL/min with TDF); (3) bone mineral density (BMD) at the hip and spine improved; and (4) fasting lipid measures (total cholesterol, LDL, HDL and triglycerides) all increased with the switch to TAF.
These virologic and safety measures are consistent with differences in the pharmacokinetic characteristics of TAF and TDF. The ability of TAF to maintain HIV suppression is entirely consistent with the ability of TAF to produce about 4-fold higher concentrations of TFV-diphosphate, the virologically active moiety, in PBMCs compared with TDF. Conversely, plasma concentrations of TFV with TAF are about 11-fold (91%) lower than those with TDF. The documented associations of TFV plasma concentrations with adverse renal and bone effects, and with the lipid lowering effect of TFV explain the improvement in renal and bone measures and the loss of some of the lipid lowering effects.
This study supports the role of TAF in antiretroviral therapy and illustrates some safety benefits of TAF over TDF. Additionally, it nicely reinforces pharmacologic principles of relationships among concentrations and response - both desired and adverse.
CROI: Switching to F/TAF (Tenofovir Alafenamide) from F/TDF (Tenofovir DF) based Regimen Study 311-1089: 48-Week Data
b. TDF is an independent risk factor for fractures in a large European cohort (abstract 46) and BMD recovers after stopping TDF/FTC for PrEP (abstract 48LB).
Illustrating the clinical opportunities for a safer alternative to TDF are abstracts 46 and 48LB (among others at CROI 2016, including a very nice presentation by Dr. Paddy Mallon on what ARVs do to bone, see session S-6, #126, http://www.croiwebcasts.org/console/player/29685?mediaType=slideVideo&).
Abstract 46 assessed fractures and osteonecrosis during 86118 person-years of follow-up among 11820 EuroSIDA participants. They reported that TDF use, either ever or current use, but not other ARVs, was an independent risk factor for fractures. They did not find an association between the use of any of the ARVs and the risk of osteonecrosis.
CROI:Antiretrovirals, fractures and osteonecrosis in a large European HIV cohort - EuroSIDA
Abstract 48LB evaluated the recovery of BMD after stopping TDF/FTC among participants of the iPrEx study. BMD decreased during the 24 week IPrEx study, and stratifying participants as having TFV-DP concentrations either < 16 or ≥ 16 fmol/106 cells, the decrease was greater in participants who had higher TFV-DP concentrations. The TFV-DP concentration of 16 fmol/106 cells was associated with a 90% reduction in HIV incidence, and this finding would provide a basis for that same concentration as associated with a loss of BMD in the hip and spine. Six months after TDF/FTC was stopped, average BMD had completely recovered in the spine and hip in young adults (< 25 years). In older adults, BMD completely recovered in the spine, although not in the hip, at 6 months after stopping TDF/FTC PrEP. Complete recovery of hip BMD had occurred by the time the iPrEx open label extension study started.
These findings provide a clear illustration that the loss of BMD is a TFV exposure-related response, and that when the drug is stopped the adverse effect goes away.
It is always reassuring when pharmacologic principles apply in the real world.
CROI:Recovery of bone mineral density after stopping oral HIV pre-exposure prophylaxis
c. Intramuscular (IM) injection of long-acting cabotegravir (CAB-LA) and rilpivirine (RPV-LA) maintains suppression of HIV (LATTE2; abstract 31LB).
In this trial of maintenance therapy, 309 HIV-infected individuals were randomized to receive CAB-LA and RPV-LA either every 4-weeks or every 8-weeks, or to remain on the oral induction regimen. All participants had HIV-RNA < 50 copies/mL with an induction regimen of oral CAB (30 mg once daily) given with ABC+3TC. The every 4-week CAB-LA and RPV-LA doses were 400 mg and 600 mg, respectively; the every 8-week doses were 600 mg and 900 mg, respectively. The CAB-LA formulation is 200 mg/mL and the RPV-LA is 300 mg/mL; injection volumes, therefore, for the every 8-week regimen were 3 mLs for CAB-LA and 3mLs for RPV-LA. The primary endpoint was the percent of subjects with HIV-RNA < 50 copies/mL and safety at week 32. At week 32, the proportions of participants with HIV-RNA < 50 copies/mL were: oral CAB/ABC/3TC, 91%; every 4-week CAB-LA and RPV-LA, 94%; and every 8-week CAB-LA and RPV-LA, 95%. The most common adverse event was injection site reactions ISRs), which were reported from 53% of the injections. The majority of these were grade 1; the number of subjects reporting ISRs decreased over time, and only 2/230 (1%) withdrew from the study because of ISRs. CAB concentrations with LA dosing were less than the trough value achieved with 30 mg orally once daily; they averaged at about the midpoint of trough concentrations between a 10 mg and 30 mg oral dose of CAB and were quite stable over time. There was a clear upward trend over time in RPV plasma concentrations with LA administration; those during the first 16 weeks were lower than those from 16-32 weeks. The authors' commented that a strategy to increase RPV concentrations during treatment was being investigated.
My interpretation of these results is that they provide clear evidence that CAB and RPV LA formulations can maintain suppression of HIV. While ISRs were frequently reported, participant withdrawal for these adverse events was rare. Whether this holds up with longer durations of administration, and outside of the setting of a controlled clinical trial, remains to be learned. This study may well mark the beginning of an era of long-acting parenteral therapy for HIV infection.
CROI: Cabotegravir + Rilpivirine as Long-Acting Maintenance Therapy: LATTE-2 Week 32 Results
d. MK-8591 (4-ethynyl-2-fluoro-2'-deoxyadenosine, EFdA) is an investigational, potent, long acting NRTI with potential for both oral and parenteral administration (abstracts 98 and 437LB).
EFdA, in vitro, has very potent activity against HIV: the IC50 is approximately 0.2 nM, which would make it approximately 8400 times more potent than tenofovir (see Antimicrob Agents Chemother 2015;59:4190. doi: 10.1128/AAC.05036-14). In macaques, preliminary PK studies indicated an estimated plasma half-life of 7 hours, and an intracellular half-life of EFdA-triphosphate (EFdA-TP) of > 72 hours.
Weekly doses of EFdA were evaluated in macaques in Abstract 98. Viral load declines of approximately 2logs were observed, and an intracellular concentration of EFdA-TP of > 0.5 pmol106 cells was associated with weekly efficacy.
Six HIV-infected, ARV-naïve males received a single oral dose of 10mg of MK-8519. A rapid reduction in HIV-RNA was observed, with a mean reduction of 1.67 log10 at day 7. The mean plasma half-life of MK-8591 was 60.3 hours, and the intracellular half-life of EFdA-TP was 108 hours. Mean EFdA-TP concentration at day 7 was 1.01 pmol/106 cells, above the target of 0.5 pmol/106. Short-term safety and tolerance was good.
Collectively, the macaque and human studies indicate that MK-8591 is a potent NRTI, with a long plasma and intracellular half-life. These characteristics may allow weekly oral dosing, and even more prolonged, perhaps every 6-month parenteral dosing. NRTIs have a well-established record of safety and efficacy in the treatment and prevention of HIV-infection. It will be exciting to watch the development of this drug, and in particular to see whether a long-acting parenteral formulation is feasible as that would extend further the choices of ARVs that could be used in long-acting parenteral regimens.
CROI: Long-Acting Oral and Parenteral Dosing of MK-8591 for HIV Treatment or Prophylaxis - (02/24/16)
CROI: A Single Monotherapy Dose of MK-8591,a Novel NRTI, Suppresses HIV for Ten Days - (02/24/16)
II. Pre-Exposure Prophylaxis (PrEP)
a. Two phase III trials demonstrate monthly use of a dapivirine (DPV) vaginal ring reduces the risk of HIV-1 infection (abstracts 109LB and 110LB).
The results of two randomized, double blind, placebo-controlled trials of the DPV vaginal ring were presented. Both had the same design: healthy, sexually-active, women 18-45 years of age, not infected with HIV, were randomized to use a vaginal ring monthly that contained either DPV or placebo. The results of both trials were generally similar: overall, the incidence of HIV-infection in the DPV ring group was approximately 30% lower than the placebo group, and in post-hoc analyses, there was an age effect showing no efficacy in women 18-21 years of age. In both studies, greater adherence to use of the DPV ring was associated with greater efficacy. The rates of adverse events were similar between the DPV ring and placebo groups.
Both studies illustrate the challenges of adherence, even with a product that only needs to be taken (i.e. removed and a new ring inserted) once monthly. But, as noted by the ASPIRE investigators in their presentation (abstract 109) and publication (Baeten JM, NEJM, February 22, 2016; DOI: 10.1056/NEJMoa1506110... http://www.nejm.org/doi/full/10.1056/NEJMoa1506110) adherence may not explain all of the age-related difference in efficacy; other biologic factors may be at play. I agree, and from the pharmacologic point-of-view, here are some significant unknowns: (1) are there age-related differences in DPV PK in the vaginal compartment; (2) as DPV is a substrate for CYP metabolism, and metabolism in vaginal tissues has been shown (see: Bumpus N, Biochem Pharmacol 2013: http://www.ncbi.nlm.nih.gov/pubmed/23965226) could concomitant medications/natural products that are inducers of drug metabolism affect DPV efficacy; (3) does the use of douches or lubricants affect the efficacy of the DPV-ring; and finally (4) would a higher dose of DPV in the ring be more effective? On this latter point, only a 25 mg containing DPV vaginal ring has been studied, whereas the standard approach is to conduct a dose-ranging study at some point in drug development. In my opinion, the lack of a dose-finding study creates a major limitation in our knowledge of the efficacy of this product. That said, these are important results and would advance (if the DPV ring gains regulatory approval) choices available to women for HIV prevention.
CROI: Dapivirine Vaginal Ring Cuts HIV Risk 27% to 56% in Two Trials in African Women - written by Mark Mascolini - (02/23/16)
CROI: A Phase III Trial of the Dapivirine Vaginal Ring for HIV-1 Prevention in Women (ASPIRE)
CROI: 2 Dapivirine Vaginal Ring Studies
b. Maraviroc (MVC) containing regimens for PrEP were comparably as safe as TDF/FTC and observed efficacy supports evaluation in larger clinical efficacy trials (abstract 103).
HPTN 069 / ACTG 5305 is a randomized, double blind, study of 4 regimens for PrEP: (1) MVC alone; (2) MVC + FTC; (3) MVC + TDF; and (4) TDF/FTC. This study enrolled 406 adult men and was powered for a comparison of safety and tolerance, not efficacy. Plasma concentrations of MVC were equivalent whether given alone or with TDF or FTC. The safety and tolerance of the MVC regimens were comparable to TDF+FTC. 5 HIV seroconversions occurred: 1 in the MVC + TDF arm and the other 4 in the MVC alone arm. In 4 of these cases, no or very low concentrations of MVC were measurable at the time of seroconversion. These data provide a safety and efficacy basis for further evaluation of MVC for PrEP, and once again show the value of measured concentrations as an indicator of medication adherence.
CROI: Maraviroc induces HIV production in a RCT and in vitro, potentially via the NF-kB pathway
CROI: Three Maraviroc PrEP Regimens Largely Protective in US MSM Study - Mark Mascolini
CROI: HPTN 069 / ACTG A5305 Phase II Study of Maraviroc (MVC)-Containing Regimens for HIV PrEP in Men Who Have Sex With Men (MSM)
c. Tenofovir (TFV) and tenofovir-diphosphate (TFV-DP) concentrations in rectal tissues and female genital tract tissues were substantially lower following a single dose of TAF compared with TDF (abstract 102LB).
As discussed above, TAF achieves higher intracellular concentrations in PBMCs and lower plasma concentrations than does TDF. There has not been any information on concentrations in mucosal tissues, such as rectal and the female genital tract. Abstract 102LB described a PK study in eight healthy women not infected with HIV. TFV and TFV-DP concentrations were measured in plasma, PBMCs, and mucosal tissues following a single dose of 25mg of TAF. These concentrations were compared with those from a previous study of a single dose of TDF. Plasma and PBMC concentrations were consistent with expectations. This was not true of cervicovaginal fluid (CVF) and mucosal tissue concentrations.
⋅ TFV concentrations in CVF were 11 fold lower following TAF;
⋅ TFV concentrations in mucosal tissues were 2-10 fold lower following TAF;
⋅ TFV-DP concentrations in genital tract tissues were 1-folder lower following TAF; and
⋅ TFV-DP concentrations in rectal tissue were 13-fold lower following TAF.
Lower TFV concentrations in CVF and mucosal tissues were expected, given that TAF produces lower plasma concentrations than does TDF. Lower concentrations of TFV-DP in genital tract and rectal tissues following TAF were a surprise. Based on animal studies, and human data showing higher PBMC concentrations of TFV-DP following TAF, the rational expectation was that TFV-DP concentrations should be higher in genital tract and rectal tissues. This was a well-done study by an established group of investigators, and at this time it is not clear why concentrations of TFV-DP in these tissues would be lower. The authors concluded that TAF, because of low TFV-DP concentrations in mucosal tissues, should not be used for PrEP until further clinical studies are completed. I agree, even though a study of oral TAF + FTC in macaques (abstract 107) did provide protection against rectal SHIV infection. It is clear that in human we have some gaps in our understanding of the tissue pharmacology of TAF, and those should be filled in, prior to any recommendations to use TAF for prevention.
CROI: Chemoprophylaxis with Oral FTC/TAF Protects Macaques from Rectal SHIV Infection - (02/29/16)
CROI: Concentrations of TFV and TFVdp in Female Mucosal Tissues After a Single Dose of TAF
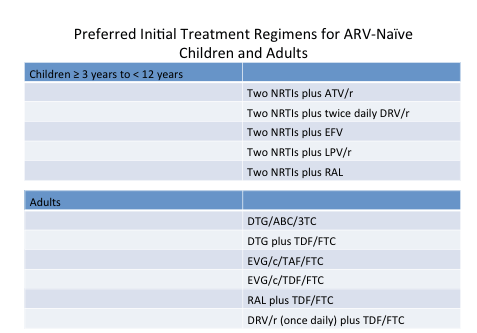
III. Drug Development for Children
Children are often therapeutic-orphans, meaning that drug development for them lags well behind that of adults. There were several abstracts and two themed discussion sessions (TD1 and TD10) at CROI 2016 that dealt with new drugs for children. To make the point, the following slide is modified from David Burger's presentation in TD1 (see http://www.croiwebcasts.org/console/player/29496?mediaType=slideVideo&
It shows the most current DHHS recommended regimens for initial therapy in ARV-naïve children and adults. Several differences in the regimens are apparent, which are driven by the lack of fixed dose formulations and pediatric formulations. There was some progress in meeting the therapeutic needs of children, and I will highlight some of those abstracts below.
a. Dolutegravir (DTG) in treatment-experienced, INSTI-naïve, children 6-12 years of age was safe and well tolerated, and 74% of children achieved HIV-RNA < 50 copies/mL at week 48 (abstract 816).
23 children were enrolled in this study and received weight-based doses (target of 1 mg/kg) of DTG plus an optimized background regimen. At 48 weeks, 74% of children had HIV-RNA < 50 copies/mL. DTG was safe and well tolerated: there were no discontinuations due to adverse events and no DTG-related adverse events. The mean plasma AUC24 and trough concentration were: 50.5 μg*h/mL and 0.92 μg/mL, respectively (see also CROI 2014 poster 901). These mean values in children compare well with mean values of 53.6 μg*h/mL and 1.1 μg/mL for AUC and trough in adults taking 50 mg once daily. However, variability in trough concentrations in children was 89%, which is approximately twice that in adults of 46%. This likely reflects age-related differences in children, and indicates a potential for some children to have subtherapeutic concentrations. I think some further refinement in the dosing approach for children may be needed. This work does indicate progress in bringing much needed new drugs to treatment-experienced children.
CROI: IMPAACT 1093: Dolutegravir in 6-12 Year Old HIV Infected Children: 48-Week Results
b. 92% of treatment-naïve adolescents who received EVG/c/FTC and TAF achieved HIV-RNA < 50 copies/mL at week 48 (abstract 817).
50 ARV-naïve adolescents, 12-17 years of age, weighing > 35 kg, received EVG/c/FTC/TAF for 48 weeks. At week 48, 92% had HIV-RNA < 50 copies/mL. The mean increase in CD4 cells was 224 cells/μL. No adolescent discontinued therapy due to an adverse event; there were no cases of renal tubulopathy. The mean change in spine BMD was +3.3% and total body BMD was +0.9%. While PK data were collected in this study, none, unfortunately, were presented in this poster. The safety and virologic response observed to EVG/c/FTC/TAF in these adolescents provide support for further study in younger children, and a study in children 6 to <12 is currently enrolling.
c. NVP dosing for very early ARV therapy in newborns (abstracts 440 and 815).
Very early ARV therapy (not prophylaxis) in newborns has gained attention because of the possibility such therapy may limit the size of HIV reservoirs and maintain immune responses. NVP has a well-established role in prophylaxis; it is approved for use in children ≥ 15 days, an oral suspension is available and well absorbed, and extensive PK and safety data are available. However, the appropriate treatment dose for newborns <15 days is not known.
Abstract 440 described the use of population PK modeling to evaluate NVP dosing over the first month of life, with the objective of maintaining NVP trough concentrations ≥ 3 μg/mL, which is the target suggested from treatment studies in adults. This modeling study estimated that a dose of 6 mg/kg twice daily in a term infant, and a dose of 4 mg/kg twice daily for 1 week followed by 6 mg/kg twice daily in a late preterm infant, would achieve the NVP target.
Abstract 815 described the use of very early ART, age 2.8 ± 1.7 days of life, in 6 newborns. These newborns received NVP, 6 mg/kg twice daily, in addition to 3TC and ZDV. The average NVP concentrations (approximately 12 hours post dose) at weeks 1 and 2 were 5.5 ± 3.5 μg/mL and 3.9 ± 2.5 μg/mL, respectively. These data indicate that on average, therapeutic NVP concentrations were achieved, but there was considerable variability. No infant had any adverse event related to ARV therapy. These data provide support for additional study of this NVP treatment dose in newborns. These findings are consistent with a recent report of 3 case studies of newborns who also all received the 6 mg/kg twice-daily treatment dose of NVP (see Bolaris M, J Pediatric Infectious Diseases Society, http://www.ncbi.nlm.nih.gov/pubmed/26803329 ).
Finding the right dose for newborns, children and adolescents is complex for any drug, but particularly for those metabolized in the liver like NVP, because of the effects of age, growth and development on drug metabolism. These abstracts are a good illustration of how to learn all we can by modeling and simulation (see also abstract 441 for a modeling study predicting ARV concentrations in infant from exposure to maternal drugs) and then confirming (hopefully) those predictions in patients. I believe this is clearly a strategy by which we can speed up drug development for HIV-infected children.
All in all, CROI 2016 was an outstanding meeting, and progress on several fronts was reported. CROI 2017 will be in Seattle, WA, February 13-16, 2017 and then back to Boston for CROI 2018 on March 4-7, 2018.
Abbreviations
%CV, percent coefficient of variation
ABC, abacavir
ACTG, adult AIDS clinical trials group
APV, amprenavir
ARV, antiretroviral drug
ART, antiretroviral drug therapy
AUC, area under the concentration-time curve
ATV, atazanavir
BID, twice daily
C12, drug concentration at 12 hours post dose
Cmax, maximum drug concentration
Cmin, minimum drug concentration
CVC, cenicriviroc
CNS, central nervous system
c or COBI, cobicistat
CSF, cerebrospinal fluid
CVF, Cervicovaginal fluid
Ctrough, concentration immediately before the next dose
CYP, cytochrome P450 drug metabolizing enzymes
DBS, dried blood spot
DCV, daclatasvir
DHHS, Department of Health and Human Services
DSMB, data safety monitoring board
DTG, dolutegravir
DRV, darunavir
ddI, didanosine
EFV, efavirenz
EVG, elvitegravir
FDV, faldaprevir
FTC, emtricitabine
ETR, etravirine
fAPV, fosamprenavir
GMR, geometric means ratio
HAND, HIV-associated neurocognitive disorders
HDAc, histone deacetylase
IC50, concentration of drug required to inhibit viral replication in vitro by 50%
IC90, concentration of drug required to inhibit viral replication in vitro by 90%
IDV, indinavir
IM, intramuscular
IQ, inhibitory quotient
IVR, intra-vaginal ring
3TC, lamivudine
LDV, ledipasvir
LPV, lopinavir
MVC, maraviroc
MPA, medroxyprogesterone acetate
NVP, nevirapine
NRTI, nucleoside reverse transcriptase inhibitor
NNRTI, non-nucleoside reverse transcriptase inhibitor
PACTG, pediatric AIDS clinical trials group
PBMCs, peripheral blood mononuclear cells
PD, pharmacodynamic
PEG-IFN, pegylated Interferon
PG, pharmacogenetics/pharmacogenomics
PK, pharmacokinetic
PI, inhibitor of HIV protease
PrEP, pre-exposure prophylaxis
QD, once daily
r or RTV, ritonavir
RAL, raltegravir
RBT, rifabutin
RBV, ribavirin
RPT, rifapentine
RIF, rifampin
RPV, rilpivirine
SQV, saquinavir
SC, subcutaneous
SOF, sofosbuvir
TAF, tenofovir alafenamide
TDF, tenofovir disoproxil fumarate
TFV, tenofovir
TDM, therapeutic drug monitoring
TPV, tipranavir
TB, tuberculosis
ZDV, zidovudine
|
| |
|
 |
 |
|
|