 |
 |
 |
| |
Switching tenofovir DF to tenofovir alafenamide in virologically suppressed adults....TAF/FTC Noninferior to TDF/FTC, and Safer, in Switch Study
|
| |
| |
Conference on Retroviruses and Opportunistic Infections (CROI), February 22-25, 2016, Boston
Mark Mascolini
WEBCAST link: http://www.croiwebcasts.org/console/player/29457?mediaType=audio&
Tenofovir alafenamide coformulated with emtricitabine (TAF/FTC) plus a third antiretroviral proved virologically noninferior to tenofovir disoproxil fumarate (TDF) coformulated with FTC plus a third antiretroviral for 48 weeks in a randomized switch study involving virologically suppressed adults [1]. TAF/FTC significantly improved signals of kidney or bone toxicity in people switching from TDF/FTC.
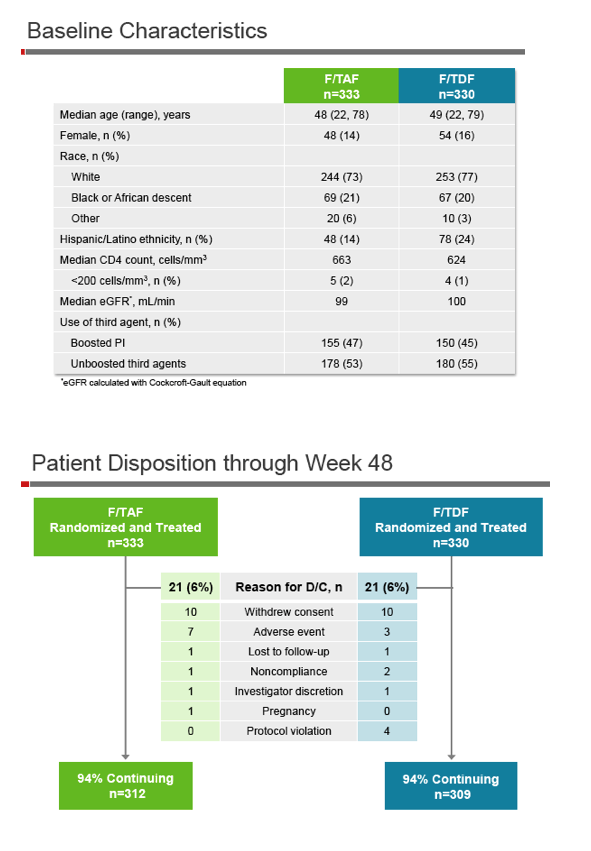
TAF, already licensed in combination with elvitegravir, cobicistat, and FTC, has 91% lower plasma tenofovir exposure than TDF and thus should send less tenofovir to off-target cells and organs. This trial randomized 663 adults with a viral load below 50 copies while taking TDF/FTC plus a third antiretroviral to continue TDF/FTC (plus TAF/FTC placebo) or to switch to TAF/FTC (plus TDF/FTC placebo). Everyone had an estimated glomerular filtration rate (eGFR) at or above 50 mL/min. TAF/FTC is dosed at 10/200 mg with ritonavir-boosted protease inhibitors and at 25/200 mg with unboosted third agents.
Median age of the study group stood at 48 in the TAF group and 49 in the TDF group. About 15% of participants were women and about three quarters white. Median eGFR was 99 or 100 mL/min in the two groups.
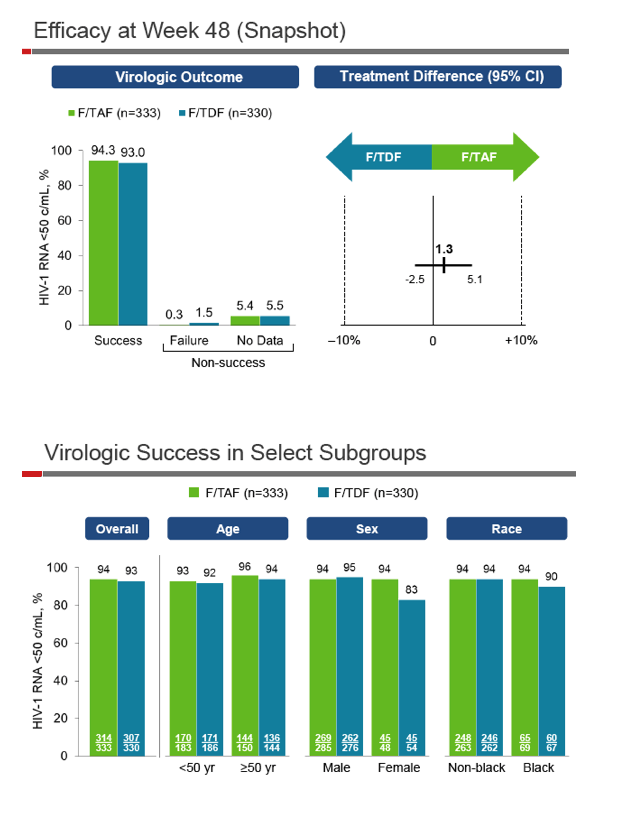
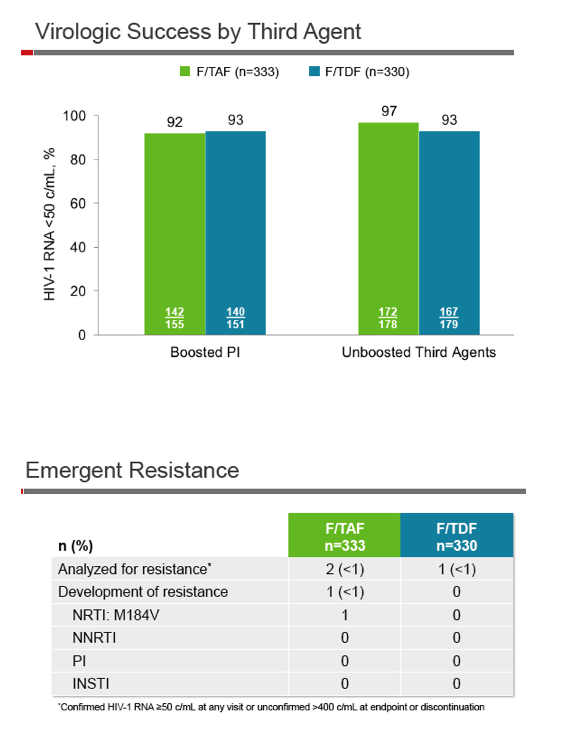
After 48 weeks a snapshot analysis determined that 94.3% randomized to TAF/FTC and 93.0% randomized to continued TDF/FTC maintained a viral load below 50 copies. The resulting treatment difference (1.3, 95% confidence interval -2.5 to 5.1) indicated that TAF/FTC was noninferior to TDF/FTC in this population. Virologic suppression results with the two regimens were similar in people younger or older than 50, in blacks and nonblacks, and in men. A higher proportion of women maintained suppression with TAF/FTC than with TDF/FTC (94% versus 83%).
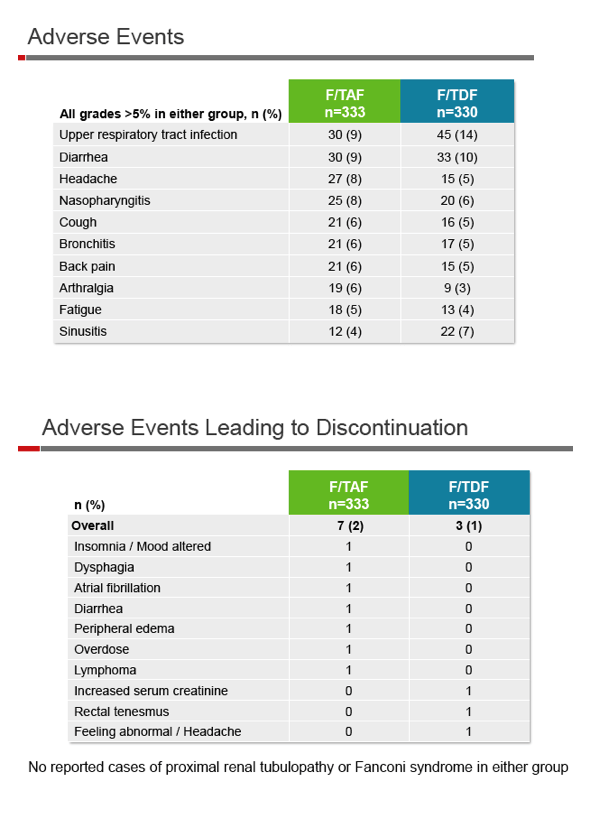
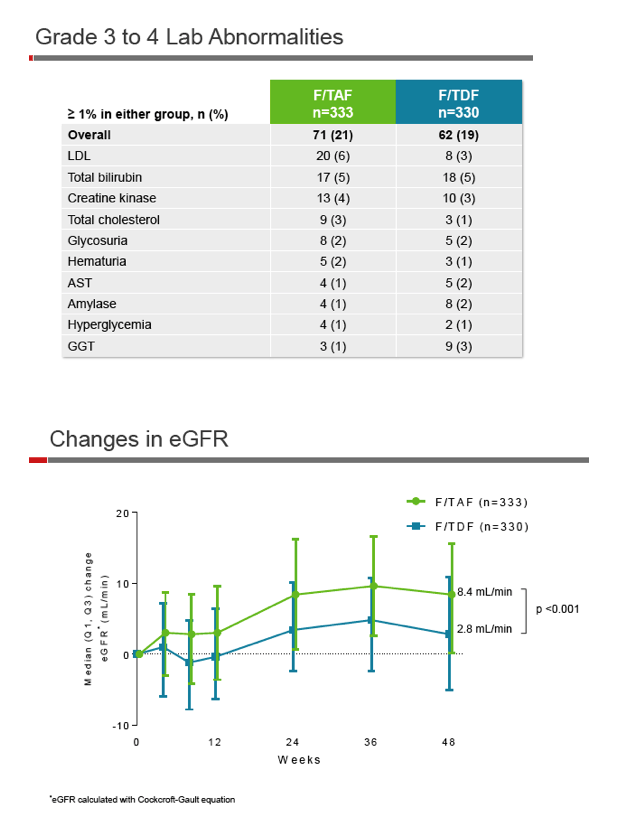
The same proportion of trial participants, 6%, discontinued treatment in each study arm, 7 people (2%) because of adverse events in the TAF group and 3 (1%) for that reason in the TDF group. Similar proportions taking TAF and TDF (21% and 19%) had a grade 3 or 4 lab abnormality. High low-density lipoprotein (LDL) cholesterol was the most frequent abnormality in the TAF group, affecting 20 people (6%).
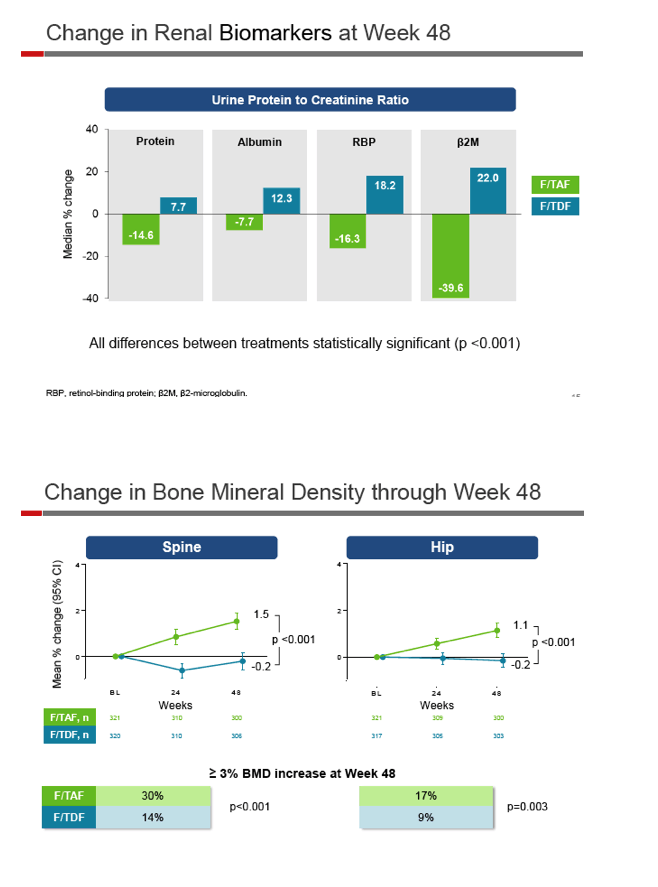
Through 48 weeks eGFR improved significantly more with TAF than with TDF (8.4 versus 2.8 mL/min, P < 0.001). Four renal biomarkers improved with TAF while worsening with TDF (P < 0.001 for all four differences). Spine and hip bone mineral density (BMD) improved with TAF while staying flat with TDF (P < 0.001 for both differences). After 48 weeks 30.3% and 16.7% assigned to TAF had at least a 3% BMD increase in the spine and hip, compared with 13.7% and 8.6% assigned to TDF (P < 0.001 and P = 0.003).
Median LDL cholesterol rose significantly more with TAF (from 187 to 200 mg/dL) than with TDF (from 182 to 183 mg/dL) (P < 0.001), and the total-to-HDL cholesterol ratio worsened marginally with TAF (from 3.6:1 to 3.7:1) while remaining the same with TDF (at 3.6:1) (P = 0.069). Investigator Joel Gallant, who presented the findings, noted that TDF has intrinsic lipid-lowering properties, so a slight jump in lipids upon switching to TAF can be expected. Gallant characterized the clinical significance of lipid changes with TAF in this trial as "questionable."
Regulatory agencies are reviewing coformulated TAF/FTC. Single-tablet combinations of TAF/FTC with rilpivirine, darunavir/cobicistat, and GS-9883 (a nonboosted integrase inhibitor) are in development.
Reference
1. Gallant JE, Daar E, Raffi F, et al. Switching tenofovir DF to tenofovir alafenamide in virologically suppressed adults. Conference on Retroviruses and Opportunistic Infections (CROI), February 22-25, 2016, Boston. Abstract 29.
---------------------



|
| |
|
 |
 |
|
|