 |
 |
 |
| |
Longer-Term Safety of Tenofovir Alafenamide in Renal Impairment
|
| |
| |
Reported by Jules Levin
CROI 2016 Feb 22-24 Boston
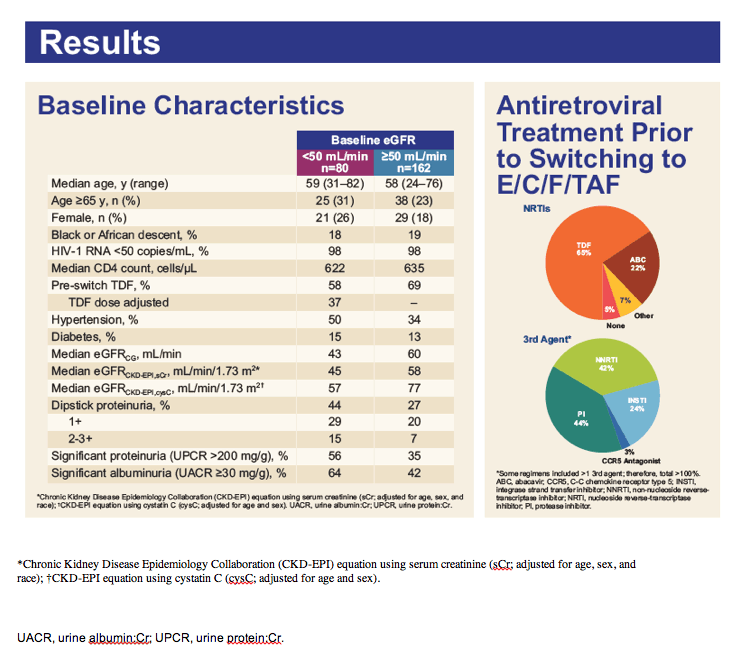
Frank A. Post,1 Pablo Tebas,2 Amanda Clarke,3 Laurent Cotte,4 William Short,2 Michael E. Abram,5 Shuping Jiang,5 Andrew Cheng,5 Moupali Das,5 Marshall W. Fordyce5
1King's College Hospital NHS Foundation Trust, London, UK; 2University of Pennsylvania, Philadelphia, PA; 3Brighton and Sussex University Hospitals NHS Trust, Brighton, UK; 4Croix-Rousse Hospital, Hospices Civils de Lyon, France; 5Gilead Sciences, Inc., Foster City, CA
Program Abstract
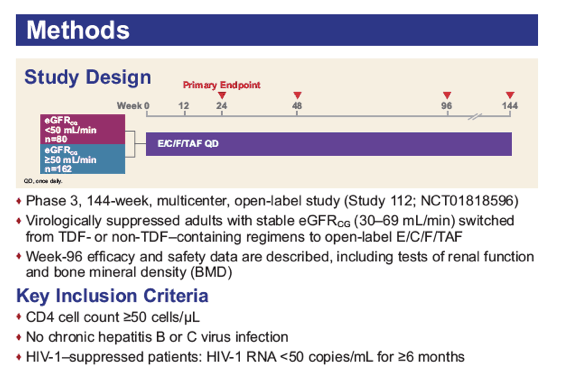
Tenofovir alafenamide (TAF) is a novel prodrug of tenofovir (TFV) that results in 91% lower plasma TFV levels compared to TDF. Switch to a once-daily single tablet regimen of elvitegravir, cobicistat, emtricitabine, and TAF (E/C/F/TAF) in HIV-1 infected patients with eGFRCG (Cockcroft-Gault) 30 to 69 mL/min was shown to be effective and safe through 48 weeks. Here, we report longer term results.
Virologically suppressed adults with stable eGFRCG of 30 to 69 mL/min had their treatment switched to open-label E/C/F/TAF. The primary endpoint was the change from baseline in glomerular filtration rate estimated using various formulae at 24 weeks. Longer term efficacy and safety data are described, including tests of renal function and bone mineral density (BMD).
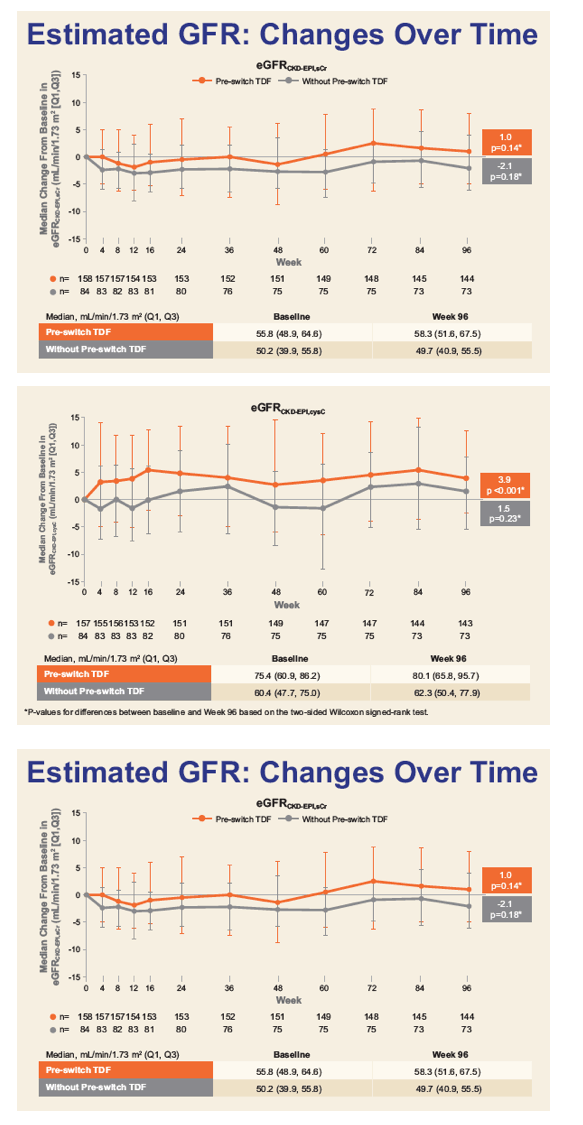
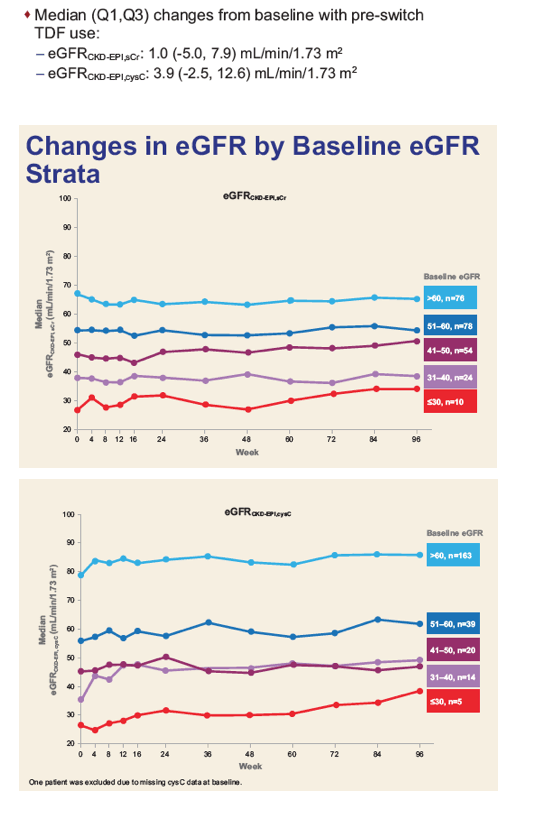
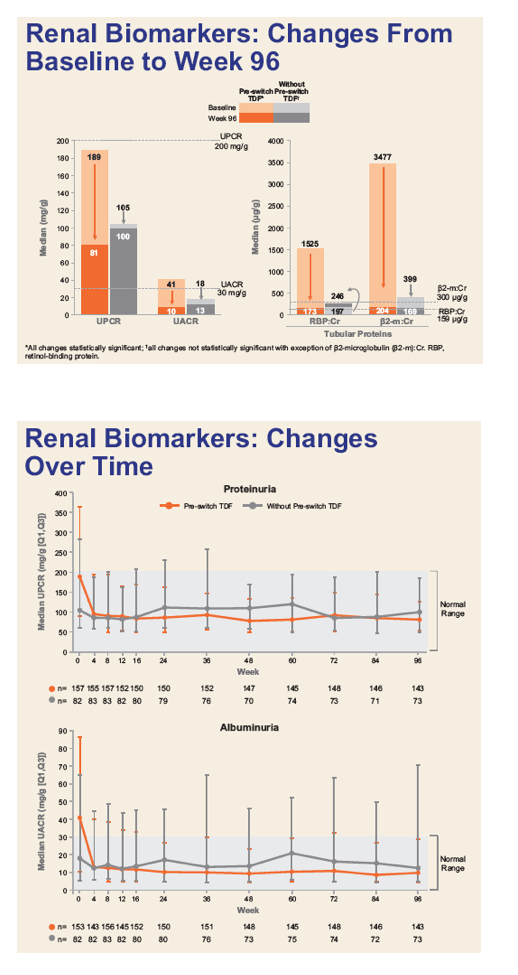
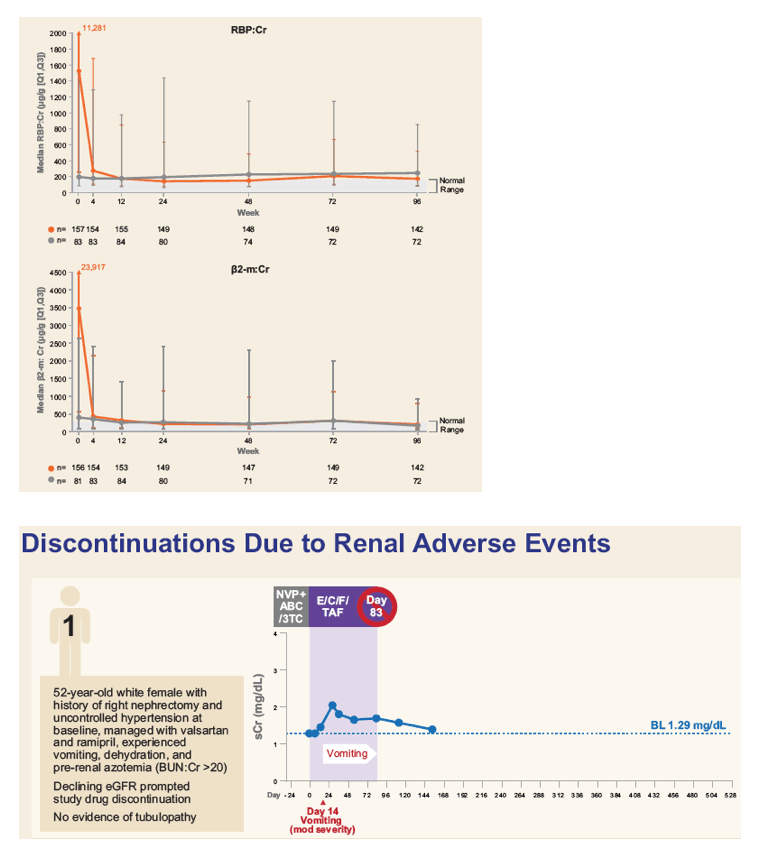
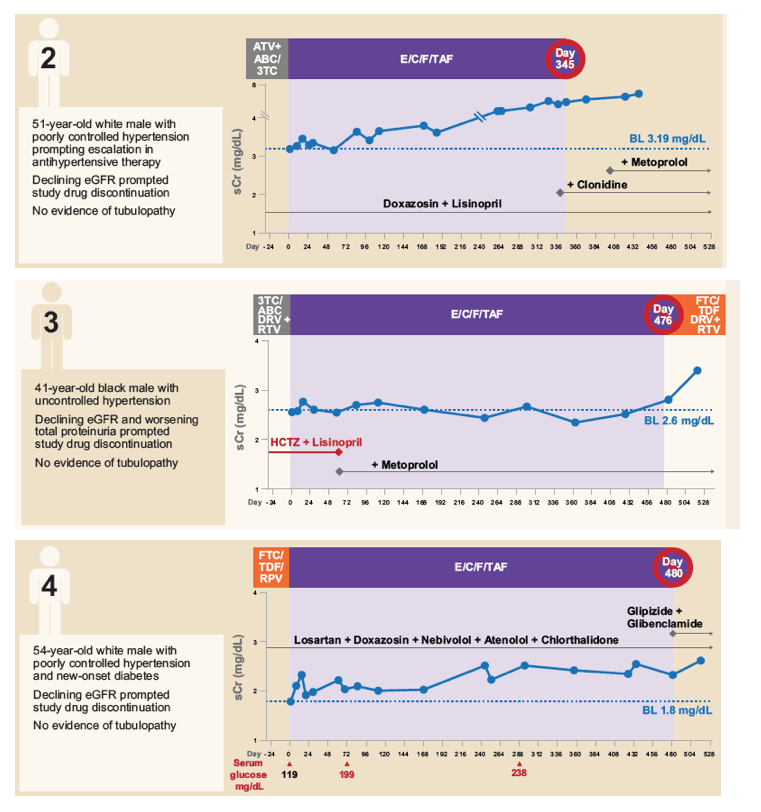
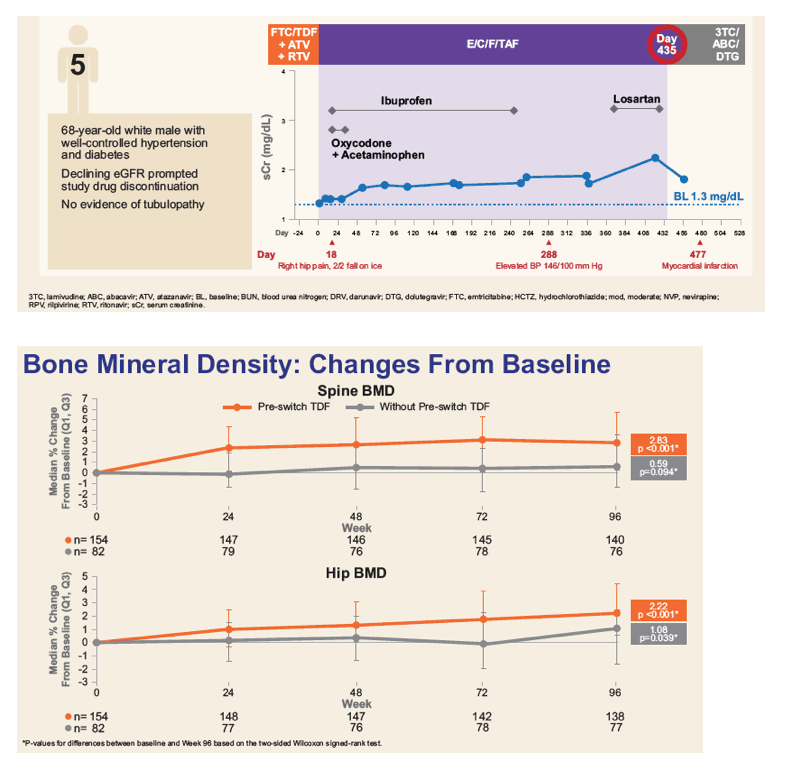
Of 242 subjects enrolled, mean age was 58 years (range: 24 - 82), 18% Black, 39% hypertension, 14% diabetes, and 65% were taking TDF-containing regimens prior to switch. Through Week 72, minimal change in eGFRCG was observed. Five patients (2.0%) with baseline eGFR <50 mL/min discontinued study drug for decreased creatinine clearance, none had evidence of proximal renal tubulopathy and all had risk factors for renal disease progression (diabetes and poorly controlled hypertension). Subjects who received TDF at baseline had significant improvements in proteinuria and albuminuria to levels seen with non-TDF regimens (Figure 1). The prevalence of significant proteinuria (UPCR > 200 mg/g) and albuminuria (UACR ≥ 30 mg/g) decreased from 42% to 18% and 49% to 28%, respectively. Hip and spine BMD increased significantly (mean % changes from baseline +1.50 and +1.91, respectively, p<0.001). 93% maintained HIV-1 RNA <50 copies/mL based on Missing = Failure analysis.
Through 72 weeks, switch to E/C/F/TAF was associated with minimal change in eGFRCG. Proteinuria, albuminuria and bone mineral density significantly improved. These data support the efficacy and safety of once daily E/C/F/TAF in HIV+ patients with eGFR 30-69 mL/min without dose adjustment.

|
| |
|
 |
 |
|
|