 |
 |
 |
| |
Cost per Sustained Virological Response 4 (SVR 4) in Genotype 1 Patients Treated With Sofosbuvir and Ledipasvir With and Without Ribavirin. A Detailed Cost-Mapping Study
|
| |
| |
Reported by Jules Levin
DDW 2016 May 22-24 San Diego, CA
Lisa M. Nyberg1, Kevin M. Chiang3, Santiago Lindsay4, T. Craig Cheetham5, Ekaterina Sadikova5, Jiaxiao M. Shi5, Zobair M. Younossi2, Anders H. Nyberg1
1 Kaiser Permanente, San Diego, California, United States; 2 Medicine, Inova Fairfax Hospital, Falls Church, Virginia, United States; 3 Pharmacy Analytical Services, Kaiser Permanente, Downey, California, United States; 4 Finance Analytics, Decision Support, Kaiser Permanente, Pasadena, California, United States; 5 Research and Evaluation, Kaiser Permanente, Pasadena, California, United States



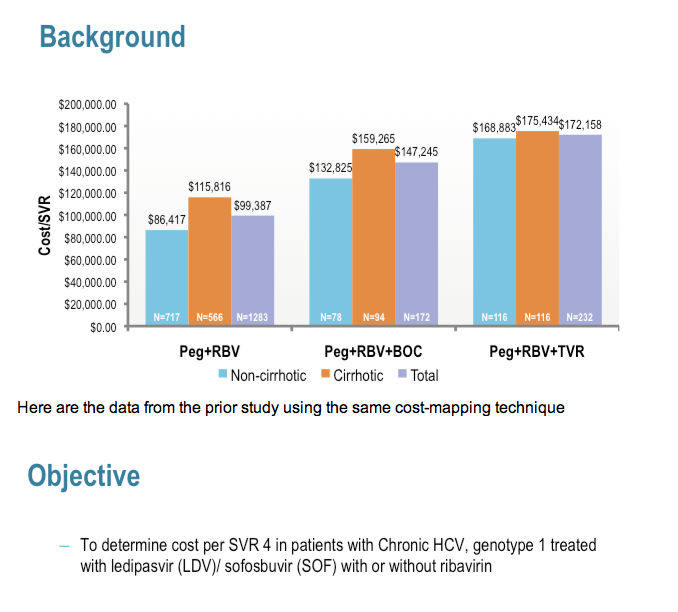
Costs of telaprevir-based triple therapy for hepatitis C: $189,000 per sustained virological response......
http://www.natap.org/2014/HCV/092914_01.htm
AASLD/Costs of Telaprevir-based Triple Therapy Including Adverse Event Management at the Mount Sinai Medical Center, NY $189,000 per
SVR.......
http://www.natap.org/2013/AASLD/AASLD_103.htm


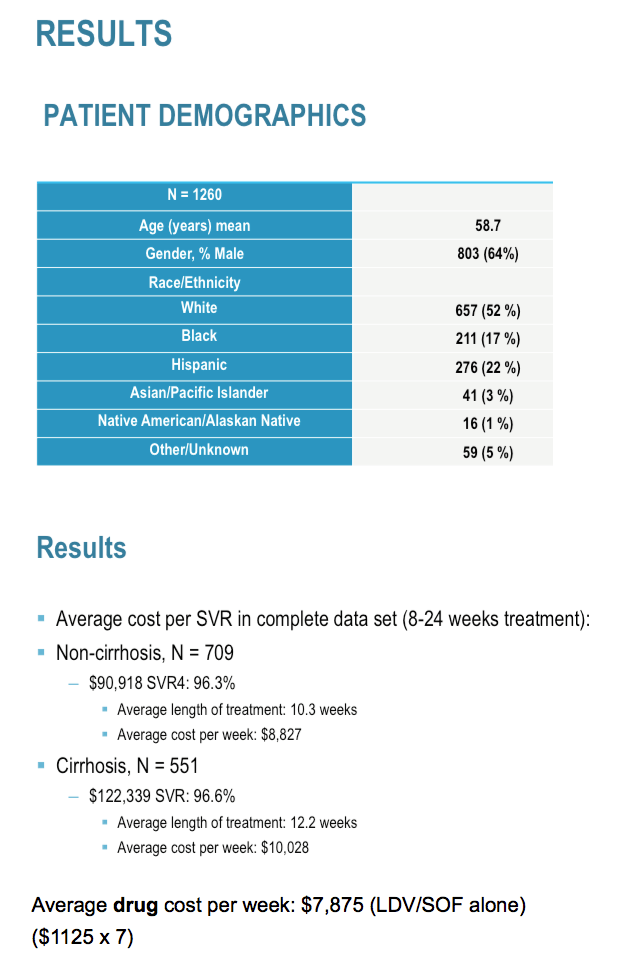
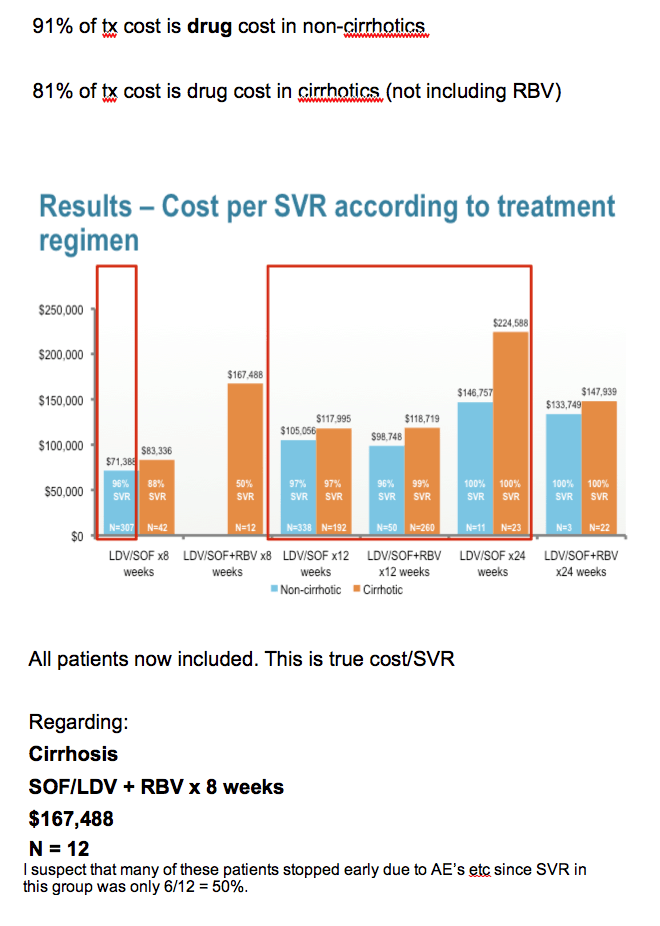
In the 8 week "cirrhotic" grp tx with Led/Sof, 60% were actually not cirrhotic - they were defined as cirrhotic by APRI score alone. The grp of cirrhotic tx w 8 w led/Sof + riba were really sick and several stopped early and needed OLT eval. A couple rolled over from receiving Sof+ riba after led/Sof was approved. That's why they received < 12 w tx with led/Sof. Finally, in the grp of Led/Sof + riba x 24 w Again, very sick with several hospitalizations and txp evals thus the high cost. In the standard 12 w Rx grps, excellent SVR and cost/SVR for Rx is 90% drug for non-cirrhotics and 81% for cirrhotics. Remember, it is also by WAC.
|
| |
|
 |
 |
|
|