 |
 |
 |
| |
In Vitro and In Vivo Antiviral Activities of AB-423 a
Potent Small Molecule Inhibitor of Hepatitis B Virus Capsid Assembly
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Nagraj Mani* 1, Andrew Cole2, Janet Phelps3, Andrzej Ardzinski1, Kyle Cobarrubias3, Andrea Cuconati1, Bruce Dorsey2, Ellen Evangelista4, Troy Harasym4, Salam Kadhim4, Amy Lee3, Hongyan Liang5, Alice Li3, Kevin McClintock4, Sara Majeski4, Stephen Reid3, Rene Rijnbrand6, Sunny Tang4, Xiaohe Wang1, Michael Sofia2, 6
1Virology, 2Chemistry, ARBUTUS BIOPHARMA, Doylestown, United States, 3In Vivo Pharmacology and Macro-Molecular Discovery, 4Drug Metabolism and Pharmacokinetics, ARBUTUS BIOPHARMA, Burnaby, Canada, 5BARUCH S. BLUMBERG INSTITUTE, 6Biology, ARBUTUS BIOPHARMA, Doylestown, United States

Program abstract
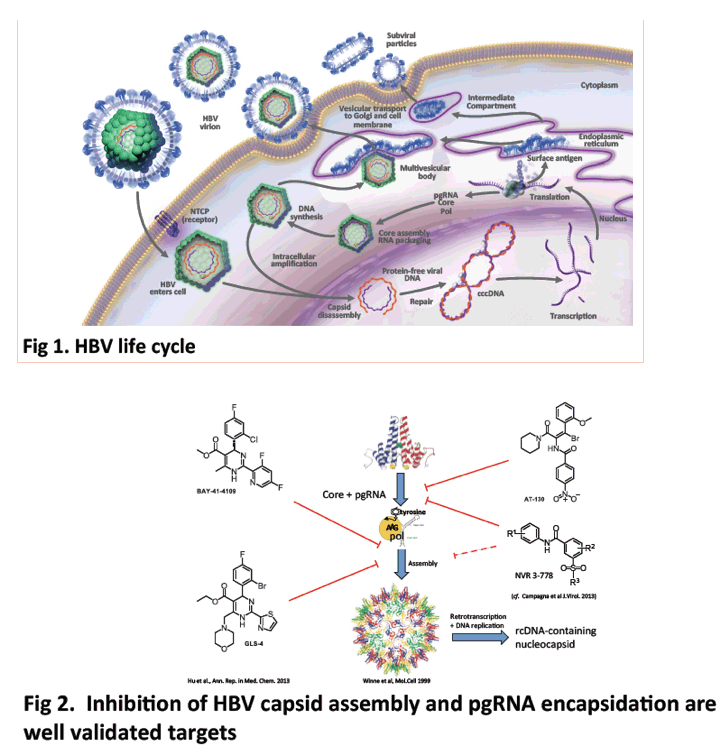
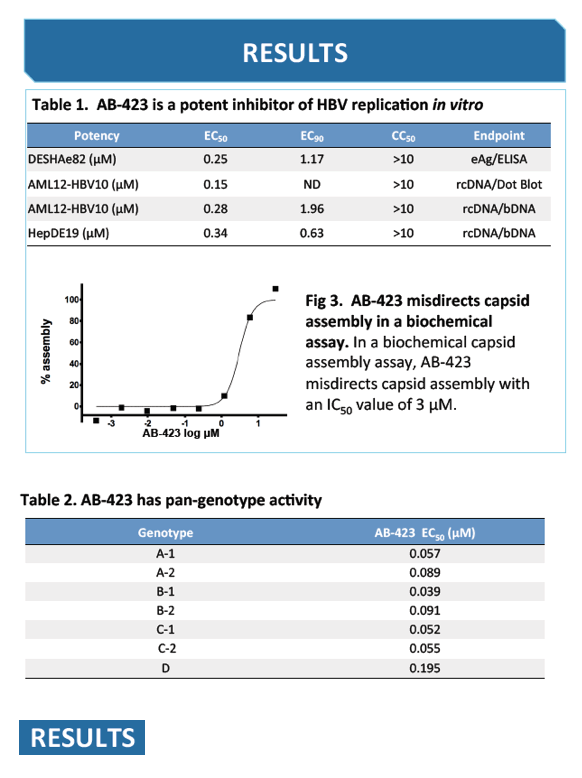
Background and Aims: Hepatitis B virus replication is strictly dependent upon capsid assembly around pregenomic RNA, which is essential for viral genome (rcDNA) synthesis, infectious virion production and maintenance of the nuclear cccDNA pool. The capsid assembly process thus represents a bona fide antiviral target, and constitutes a novel mechanism that is distinct from the nucleos(t)ide analogues currently available for clinical use. Here we characterised the in vitro and in vivo antiviral activities of AB-423, a potent small molecule inhibitor of capsid assembly.
Methods: Antiviral potency, cytotoxicity and in vitro combination studies of AB-423 were performed using HBV cell culture models and quantified using dot blot, branched DNA or quantitative PCR assays. The in vivo antiviral activity of AB-423 alone and in combination with entecavir and TKM-HBV, an RNAi treatment under development for chronic hepatitis B, was assessed in a hydrodynamic injection HBV mouse model.
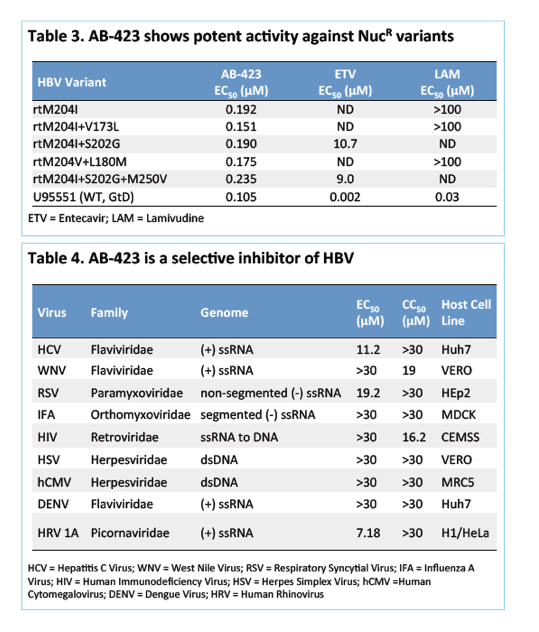
Results: AB-423 showed potent inhibition of HBV rcDNA production in a mouse cell line with an average EC50 of 275 nM and CC50 of >10 μM. In a human hepatoma cell line in which inducible cccDNA formation is dependent upon capsid assembly and rcDNA synthesis, similar potency was observed in the inhibition of cccDNA formation. In primary human hepatocytes isolated from uPA/SCID chimeric mice and infected in vitro, AB-423 at 1.0 μM concentration exhibited a ∼1-log reduction in the extracellular HBV DNA pool. AB-423 inhibited HBV capsid assembly in a biochemical assay confirming its mechanism of action. In vitro combination studies of AB-423 with entecavir in the cccDNA formation cell culture system showed additive/synergistic effects.
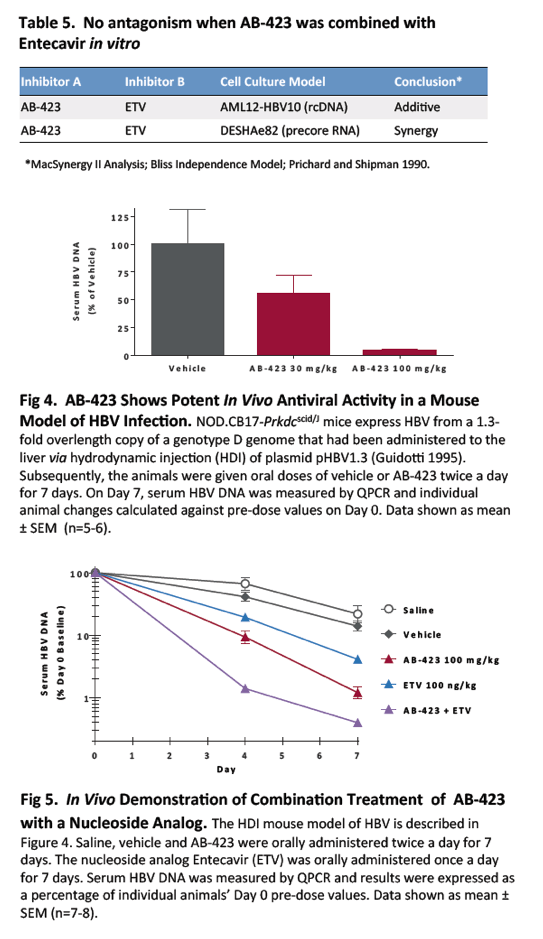
AB-423 showed favourable pharmacokinetic properties when dosed orally in mice and was tested for efficacy in SCID mice that were hydrodynamically injected with HBV plasmid DNA. AB-423 showed rapid and strong dose-dependent reduction in serum HBV DNA with a maximum reduction of ∼2 logs at 200 mg/kg on day 4 that was maintained through day 7 of the daily b.i.d. dosing period. When tested in combination with entecavir or TKM-HBV, no antagonism was detected
Conclusions: AB-423 is a promising antiviral agent that exhibited potent inhibition of HBV replication in cell culture systems and in an HBV mouse model both singly as well as in combination with entecavir or with TKM-HBV. AB-423 is being evaluated for advancement into clinical development.



|
| |
|
 |
 |
|
|