 |
 |
 |
| |
Summary from EASL 2016 for Hepatitis C
Highly efficacious pan-genotypic DAA combinations on their way: the last gaps in difficult-to-treat patient populations are going to be closed soon.
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Introduction
The International Liver Congress™ (ILC) (the annual meeting of the European Association for the Study of the Liver (EASL)) was held in April from 13-17th in in Barcelona, Spain. After broad introduction of direct acting antivirals (DAAs) for treatment of hepatitis C has occurred in North America and Europe, and DAAs have become the main pillar of HCV treatment in the various international HCV guidelines, multiple presentations at the EASL conference focused on HCV cohort and registry analysis providing feedback on efficacy and safety of these HCV regimens in real-life settings. Most reassuringly, overall sustained virological response rates (SVR) are equally high and above 90% in numerous patient cohorts which are much more enriched for more elderly and cirrhotic patients than usually seen in randomized clinical trials (1-3). Interestingly, new data from the TRIO network suggested that SVR rates are not decreased in patients on ledipasvir/sofosbuvir and concomitant proton pump inhibitor (PPI) therapy, somewhat in contrast to previously reported cohort analysis (4). Only bid intake of PPIs was associated with a lower cure rate for hepatitis therapy. Hence, PPI therapy if indicated and needed throughout HCV therapy with ledipasvir can be co-administered if given at the same time as the HCV medication (preferably with low doses of 20mg), bid dosing however, should be avoided. Moreover, new data was presented for HCV therapy in special patient populations. Shortened treatment with ledipasvir/sofosbuvir for 6 weeks in patients with acute HCV was associated with a 100% SVR rate (5). Patients with inherited blood disorders (sickle cell anemia, thalassemia, hemophilia, von Willebrand disease) who often were difficult to treat in the past, responded favorably to 12 weeks of grazoprevir/elbasvir (SVR rate 93.5%) (6) as well as kidney transplant recipients, yet another group which was extremely difficult to treat in the interferon/ribavirin era, which was cured to 100% after 12 weeks of ledipasvir/sofosbuvir therapy (7). Many presentations also covered new data on resistance associated variants (RAVs) which either may already be present at baseline and subsequently impact success of HCV therapy in patients with additional unfavorable host factors at baseline (cirrhosis, genotype 1a, previous treatment failure) or have developed after experiencing relapse after stopping HCV therapy (8,9). Clearly, guidelines are starting to recommend genotypic resistance testing in HCV patients developing virological failure under DAA-based therapy. This allows ensuring that subsequent treatment approaches include at least two fully active DAAs. Finally many new dual and triple HCV DAA combinations were presented suggesting that more broad pan-genotypic DAA combinations will become available soon which will not only allow retreatment in prior DAA failures but also close some of the remaining treatment challenges such as the cirrhotic GT3 patient with prior treatment failure (10,11). The advantage of these new combinations is that they can further simplify HCV therapy as they can be used for all genotypes and various patient populations. Some also allow shortening of HCV therapy to 8 weeks independent of HCV viral load. In conclusion, the HCV treatment success story continues with improved treatment strategies for many to come.
DAA Therapy in Acute Hepatitis C
At EASL a first pilot trial was presented which examined the efficacy and safety of shorter treatment of a 6 week therapy with Ledipasvir/Sofosbuvir in patients with acute HCV mono-infection (5). The study design is shown below in figure 1.
Figure 1: Study design

The study included patients with symptomatic acute HCV infection with genotype 1 infection. Acute HCV was defined by the following criteria: known or suspected exposure to HCV within the preceding four months, documented seroconversion to positivity for antibodies against HCV and /or ALT level of more than 10 times upper the limit of the normal rangeHCV RNA-positive and negative anti-HCV antibody/HCV RNA test within last 6 months or elevated ALT/AST >2.5 x ULN in past 6 months with normal LFTs in past year, and other causes excluded. Overall 20 subjects were enrolled. The baseline characteristics are shown below in Table 1:
Table 1: Baseline characteristics of the acute genotype 1 HCV mono-infection study
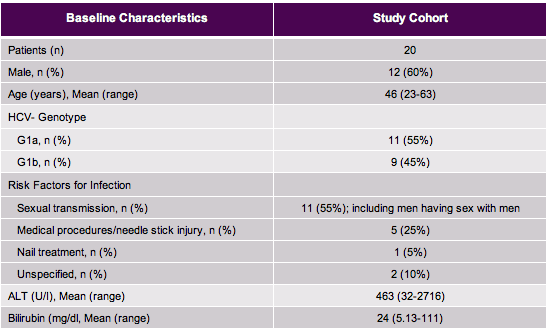
Most interestingly, sexual transmission accounted for most acute HCV infections including men having sex with men underling that the initially observed and still ongoing outbreak of acute HCV in HIV seropositive men who have sex with men (MSM) has now also reached the HIV-negative MSM population. Liver enzymes were quite high and in accordance with symptomatic acute HCV. As spontaneous clearance in symptomatic acute HCV in immunocompetent patients is high with 50% and treatment was started early within this trial it has to be discussed whether a considerate proportion of patients would have cleared HCV infection even without receiving DAA therapy. Unfortunately IL28b data at baseline was not presented but it can be assumed that there will be an over representation of the favorable IL28b CC genotype. The IL28b CC genotype has been associated with higher spontaneous HCV clearance rates. Of note all patients responded very well to the 6 weeks therapy and successfully cleared HCV infection. Figure 2 summarizes the main virological outcomes.
Figure 2: Week 2,4 and end-of-treatment (EOT) and Sustained virological response (SVR) 12 weeks after stopping HCV therapy
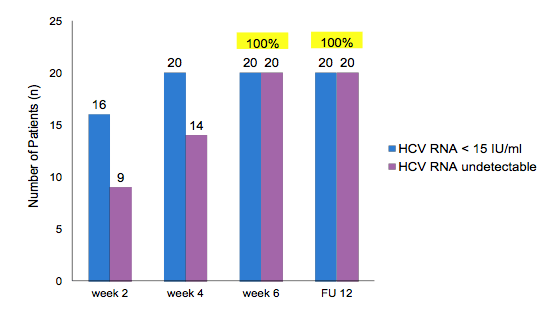
Of note all patients responded very fast with all patients having a HCV viral load below the detection limit at week 4 and all being completely undetectable at EOT and SVR12. Figure 3 shows the early virological response for each patient with regard to their respective baseline HCV viral load. Most strikingly, patients with higher baseline HCV RNA levels were more likely to have a week 4 HCV RNA <15 IU/ml but still detectable (versus completely undetectable for the lower HCV viral load at baseline patients), suggesting that baseline viral load may be a key determinant of length of therapy required for sustained viral clearance, even in the setting of acute/early HCV. At CROI data from the same treatment regimen, namely Ledipasvir/sofosbuvir again for 6 weeks was presented for treatment of acute HCV in HIV/HCV coinfected subjects (12). In this trial SVR12 was only 77% with 3 relapses, one reinfection and two patients lost to follow-up after achieving SVR4. Of note, the three relapsers all had pre-treatment HCV viral loads above 6.9 log10 IU/mL suggesting that all oral DAA-associated HCV elimination may take longer in patients with a high HCV burden. In the mono-infection acute HCV trial less patients had a high viral load which indeed may be a result of the HIV-associated perturbation of innate and adaptive immune responses.
Figure 3: Baseline HCV viral load and early viral load response under DAA therapy
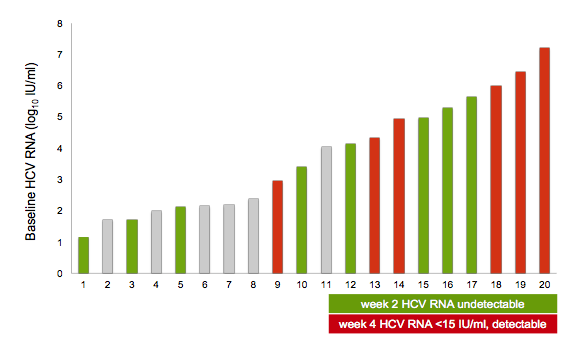
Altogether these results however, are exciting as they suggest that shorter treatment durations may be feasible in acute HCV which is in contrast to chronic HCV where even potent 3 DAA combinations were not successful in all patients receiving 6 weeks of therapy. The possibility to treat early is particularly important in order to prevent the further spread of HCV in corresponding risk populations. Shorter treatment durations could potentially make this very cost effective.
EASL: Six weeks of sofosbuvir/ledipasvir (SOF/LDV) are sufficient to treat acute hepatitis C virus genotype 1 monoinfection: The HepNet Acute HCV IV Study - (05/27/16)
EASL: EFFECTIVENESS AND SAFETY OF DAA COMBINATION THERAPIES FOR TREATMENT OF HCV IN ELDERLY PATIENTS (>70 YRS.): RESULTS FROM THE GERMAN HEPATITIS C-REGISTRY (DHC-R) - (05/31/16)
EASL: A NOVEL TASK SHIFTING MODEL TO EXPAND THE HCV CARE CONTINUUM: THE ASCEND INVESTIGATION - (05/31/16)
One of the main issues after successful treatment of acute hepatitis C obviously is the high risk for reinfections in case the transmission risk for acute HCV remains unchanged. In this context very interesting data was presented from the PROBE-C cohort (NEAT-ID funded European database on all HIV positive patients with episodes of acute HCV (13). Aim of the corresponding study was to calculate reinfection incidence among the European AIDS Treatment Network (NEAT) consortium centres in Western Europe (UK, Germany, Austria and France) and to look for factors associated with reinfection and spontaneous clearance (SC) of reinfection. All HIV-positive MSM cured of HCV with HCV PCR for follow-up were included. For treated patients HCV PCR had to be negative 12 weeks after end of treatment and for spontaneously cleared patients the HCV PCR was negative 24 weeks after first negative PCR following infection. Overall, 606 individuals with an incident acute HCV infection and were cured were included into the analysis. The overall outcome is shown in table 2.
Table 2: Results overall study population of the PROB-C acute HCV cohort

From the 606 patients included, 149 people developed a reinfection at a median time of 1.8 years of follow up. That's 24.6% of people reinfected. In line with the first HCV infection the majority of HCV infections were genotype 1 and 4 infections. 50% were infected with a different genotype or subtype. It appears that HCV reinfection affects mostly people in their late thirties and early forties with a long history of HIV. Most had a suppressed HIV VL. From the reinfections that had a reported outcome, the spontaneous clearance proportion was 15.6%. 113 of the reinfections received treatment achieving an SVR rate of 78% consistent with treatment of AHC. Many individuals went on to have a second, third and even fourth reinfection. Most interestingly, 69 individuals under follow up had a second reinfection (42%). In multivariate analysis spontaneous clearance was associated with reduced risk of reinfection (HR 0.55) and increased chance of spontaneous clearance if reinfected (OR 12.7).
In summary, the substantial HCV reinfection risk among HIV/HCV coinfected MSM with 25% reinfected at 3 years underlines the urgent need for developing new prevention strategies in this particular patient group where the outbreak of acute HCV still continues.
EASL: Hepatitis C virus reinfection incidence and outcomes among HIV-positive MSM in Western Europe - (04/15/16)
Feedback from real-life patient cohorts
The presentation of HCV cure rates and safety of all oral DAA therapy in real-life cohorts and from national treatment programs is of utmost interest for two reasons. First these cohorts are enriched with more difficult to treat patients (more advanced fibrosis stages, more previous treatment failure, older age) and give a good idea in how far clinical trials in selected patient groups translate into the larger clinic reality and secondly, it shows how different governments or health care officials tackle the respective HCV epidemic in their country. Among others an ambitious HCV elimination program has started in Portugal. At EASL first outcome results from this national web registry were reported in the first 1000 patients receiving DAA treatment (either Ledipasvir/Sofosbuvir or sofosbuvir + ribavirin). The cohort overall, consists of 8,856 patients with treatment authorization, of which 5,449 patients have initiated their treatment. This first analysis is then restricted to the 1,069 patients with SVR assessment at the cut-off date of 31th of January 2016. SVR was defined as undetectable HCV RNA at 12 (SVR12) or 24 (SVR24) weeks after treatment completion. Of the first 1069 HCV patients, 94% were treated with ledipasvir/sofosbuvir. The average age of patients was 51.6 years-old and more than half have advanced fibrosis F3/F4, 54.4% (582/1,069). The overall SVR rate was 96.3%. The corresponding figure 4 summarizes the main SVR data.
Figure 4: SVR rates according to HCV genotype and METAVIR stages
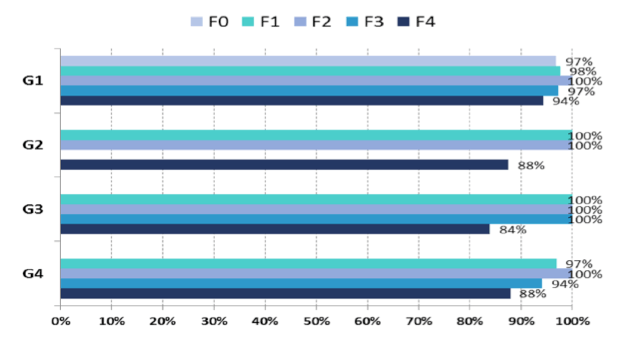
Clearly, this first data shows impressive real world SVR from the Portuguese ledipasvir/ sofosbuvir and sofosbuvir universal coverage program to eradicate HCV. Regardless of baseline METAVIR score HCV cure rates were on average way above 90%. Only exception, were GT 2 or 3 patients with cirrhosis receiving sofosbuvir and ribavirin therapy which corresponds nicely with similar numbers from clinical trials in these more difficult to treat patient groups.
EASL: Evidence of impressive real world SVR from the Portuguese ledipasvir/sofosbuvir and sofosbuvir universal coverage program to erradicate (eliminate) hepatitis C - (04/19/16)
Another interesting presentation was a first feedback on the real-world safety and effectiveness of OBV/PTV/r with DSV and/or RBV in the German HCV Registry (1). So far much real-life data has been generated with other DAA combinations but this was the first for the Abbvie 3D drug regimen. The main patient characteristics (GT, cirrhosis) and the respective DAA regimen used are shown below in table 3. Of note 76-89% of patients took the label-recommended regimen for GT 1 treatment and 92-94% took the label-recommended regimen for GT4 therapy.
Table 3: Patient disposition for OBV/PTV/r +/-DSV therapy from the German HCV registry.
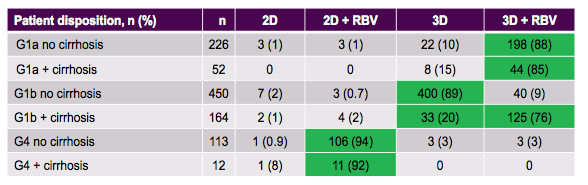
A substantial number of GT1a patients received the 3-D regimen without ribavirin most likely because of RBV contraindications or favorable baseline host factors. The main treatment outcome results for the various analyses by genotype, presence of cirrhosis, pretreatment and concomitant renal insufficiency are summarized in figure 5.
Figure 5: Efficacy by genotype, presence of cirrhosis, pretreatment and concomitant renal insufficiency for OBV/PTV/r with DSV and/or RBV
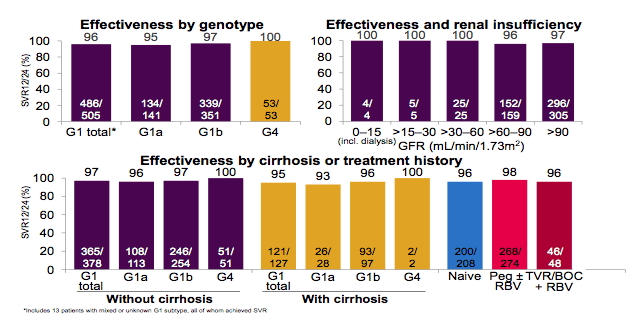
Overall, excellent SVR rates were obtained for all patient groups suggesting that the efficacy and safety of the 3-D regime in real-life cohorts parallels the findings of randomized clinical trials.
EASL: REAL-WORLD SAFETY AND EFFECTIVENESS OF OMBITASVIR/PARITAPREVIR/R ± DASABUVIR ± RIBAVIRIN IN THE GERMAN HEPATITIS C REGISTRY (clear version) - (04/15/16)
Another cohort study from Canada analyzed the efficacy and safety of ledipasvir/sofosbuvir + ribavirin for 12 weeks in treatment-naïve genotype 3 patients. Although ledipasvir does not show good GT3 activity in vitro some pilot study results have claimed good antiviral efficacy for this combination. Also in some places such as Europe initially the drug label for ledipasvir/sofosbuvir included the possibility of treating GT3 infection. In the meantime less promising data has been presented particularly in more advanced disease stages and precious treatment failure. As other regimens have demonstrated better GT3 activity such as daclatasvir/sofosbuvir guidelines no longer preferably recommend ledipasvir/sofosbuvir for GT therapy. Table 4 summarizes the main baseline characteristics of this Canadian HCV patient population.
Table 4: Demographic and baseline characteristics
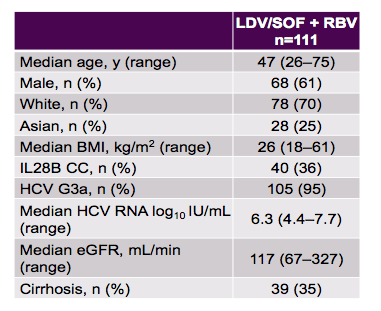
The SVR rates are shown below in figure 6.
Figure 6: SVR 12 rates in GT3 patients receiving ledipasvir/sofosbuvir + ribavirin
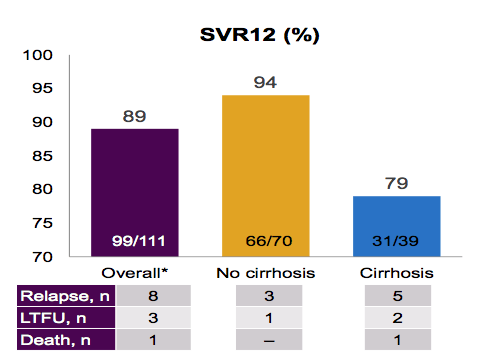
Overall, surprisingly high SVR 12 rates were obtained in non-cirrhotic patients but clearly if negative host factors are added such as cirrhosis, cure rates dramatically drop off underling the somewhat low GT3 activity of this regimen.
EASL: Ledipasvir/Sofosbuvir With Ribavirin for 12 Weeks Is Effective and Safe in Treatment-Naïve Genotype 3 HCV-Infected Patients in Canada - (05/02/16)
Another important cohort study was an analysis from the TRIO network which studied the effect of Proton Pump Inhibitor (PPI) use on SVR with Ledipasvir/ Sofosbuvir (LDV/SOF) therapy (4). Most interestingly, previously an incomplete report from the HCV TARGET network suggested that PPI use at baseline was associated with a 5% reduction in the SVR12 rate (15). The reason for this was seen in a lower AUC of ledipasvir following PH changes through PPI use. Data however, on which PPI, duration of PPI therapy as well as dosage were not available. The aim of this study was therefore to evaluate the impact of type of PPI, dose and duration of PPI on SVR12 following ledipasvir/sofosbuvir therapy. Figure 7 summarizes the SVR findings based on concomitant PPI use.
Figure 7: TRIO network: Effect of PPI on SVR: SVR12 Rates by Duration of PPI
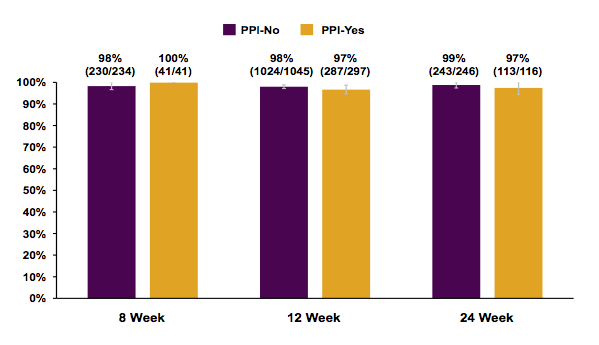
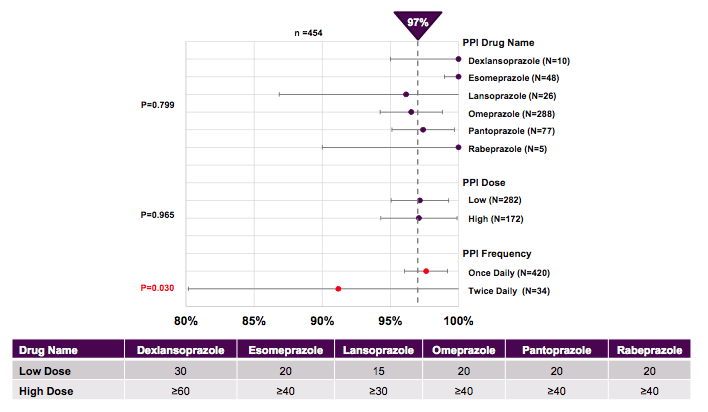
Overall, PPI use was not predictive of SVR in this large real-life population. Matched propensity analysis confirmed no effect of PPI use on SVR in 8 week, 12 week or 24 week regimens of ledipasvir/sofosbuvir. Further analysis looked at impact of type, dose and duration of PPI (see figure 9). Interestingly, the type of PPI, PPI dose and PPI duration had no effect on SVR. Twice daily PPI however, had an effect with reduced SVR12 of 91.7% in univariate analysis but not multivariate analysis. Despite small numbers and wide confidence intervals the authors suggested to be cautious when using twice daily PPI. In general in our practice the first question is whether the patient really needs his or her PPI and then if necessary try to give only one dose together with the HCV drugs.
Figure 8: TRIO - Effect of PPI on SVR: Predictors of Response By PPI Type and Dose
EASL: No Effect of Proton Pump Inhibitor (PPI) Use on SVR with Ledipasvir/ Sofosbuvir(LDV/SOF): Real-World Data from 2034 Genotype 1 Patients in the TRIO Network - (04/21/16)
Special patient populations
HIV/HCV coinfection
Although HIV/HCV coinfected patients are no longer regarded as a special patient population because of similar SVR12 rates coming out of large phase III HCV trials with various all oral DAA combinations this topic has been readdressed by a large cohort analysis from Seville claiming HIV coinfection to impair responses to DAA-based HCV therapy (16). This prospective multi-cohort study enrolled all patients who consecutively attended 33 Infectious Diseases Units throughout Spain and who initiated DAA-based therapy against chronic hepatitis C have been included since October 2011. Overall, (n=404) HIV (-) and (n=423) HIV (+) patients receiving IFN free DAA therapy were enrolled as well as (n=276) HIV (-) and (n=173) HIV (+) subjects which received DAA based IFN containing HCV therapy. The main baseline characteristics for the four patient groups are shown in table 5. The numbers of subjects receiving different IFN-free DAA combinations were: 260 (31.4%) for SOF/LED, 207 (25%) for SOF/SMV, 191 (23.1%) for 3D, 139 (16.8%) for SOF/DCV and 30 (3.6) for 2D. A DAA in combination with Peg-IFN/RBV was administered to 449 (35.2%) subjects: TVR (62.6%), BOC (19.6%), SMV (10.7%) and SOF (7.1%). In the group receiving IFN-free therapy, RBV was applied in 179 (42.3%) HIV/HCV-coinfected patients and in 177 (43.8%) of the HCV-monoinfected subjects (p=0.664). In this subpopulation, 33 (7.8%) HIV/HCV-coinfected patients versus 31 (7.7%) of the HCV-monoinfected individuals were DAA-experienced (p=0.945).
Table 5: Baseline characteristics of the study population according to treatment regimen and
HIV status.
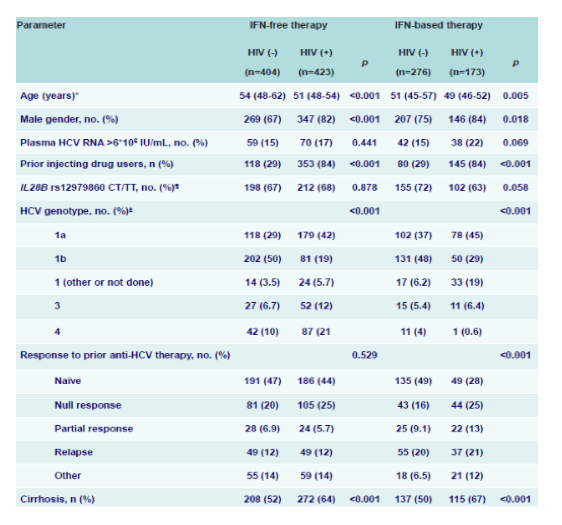
Of note, HIV+ HCV patients were more likely to be male and younger and much more likely to have a history of prior IVDU. Potentially adherence levels could have been different between the two groups. Also the coinfected subjects were more likely to be cirrhotic and have a GT1a infection. Obviously, with all the significant differences found between the various groups the question arises how confident one can be with comparative analyses. In particular the higher cirrhosis rate may impact SVR rates. Also the regimens used where different also potentially accounting for differences in SVR12 rates. The overall SVR rates for all groups are shown in figure 9.
Figure 9: Treatment outcome in an on-treatment analysis. (A) IFN-free therapy; (B) IFN-based therapy.
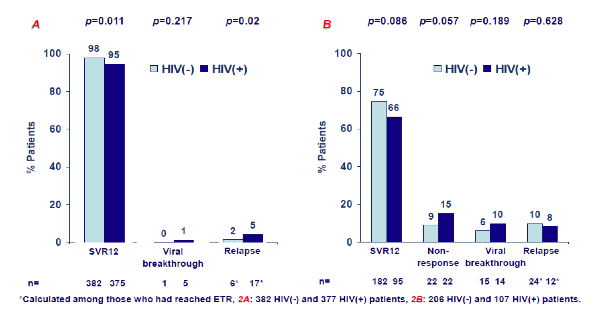
The following factors were not (p>0.2) associated with SVR12 in the univariate analysis and were thus not included in the multivariate model: use of of ribavirin in the subgroup of IFN-free therapy, treatment duration, DAA experience, IL28B genotype, baseline HCV RNA levels, previous injecting drug use and response to previous therapy against chronic HCV infection with Peg-IFN plus RBV. Overall, the authors hypothesize that HIV/HCV coinfected subjects respond worse to HCV therapy.
EASL: HIV Coinfection Impairs the Response to DAA-based HCV Therapy - (05/09/16)
Another study of great interest in the context of HIV/HCV coinfection was the ASTRAL-5 study, which looked at the efficacy and safety of the new DAA fix-dose combination of sofosbuvir/velpatasvir for 12 weeks in HIV/HCV coinfected individuals (17). Overall, 106 patients were included, the study design is shown in figure 10 below. Patients had to be on stable ART for ≥8 weeks with inclusion of non-nucleoside reverse-transcriptase inhibitor (NNRTI), integrase inhibitor, and protease inhibitor (PI) regimens with TDF/FTC or ABC/3TC.
Figure 10:Study design of the ASTRAL-5 study
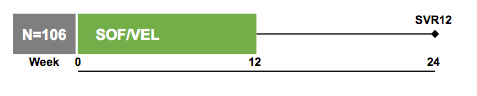
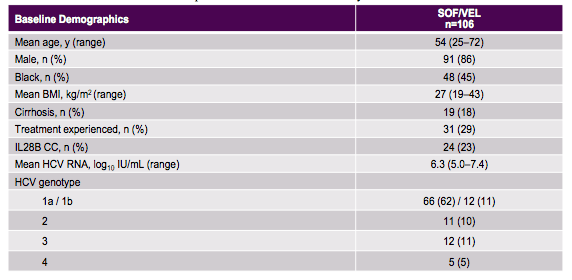
The baseline characteristics of the study population are summarized in table 6. 18% had cirrhosis allowing also a look at the efficacy of this regimen in more difficult to treat patients. Broad genotype inclusion was possible but the majority of treated patients had a GT1 infection following the general distribution of HCV genotypes in the coinfected patient population.
Table 6: Baseline characteristics of the patients from the ASTRAL-5 study
The SVR12 rates by genotype and presence of cirrhosis at baseline are shown below in figure 11.
Figure 11: Results of the ASTRAL-5 study
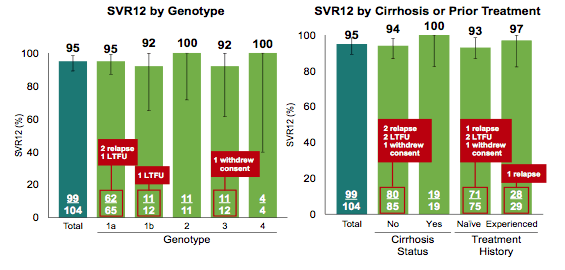
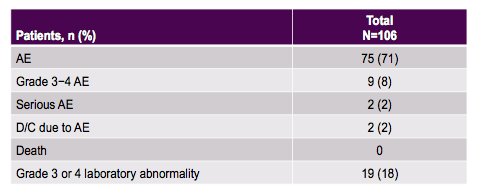
Overall, treatment success rates were extremely high. Please note that because of small numbers one patient missing or withdrawing consent can already have a huge impact on SVR12 rate. Only two relapses were noted in the entire study, both patients had GT1a infection, one was treatment naïve one was treatment experienced. The tolerability of the regimen was excellent with very few discontinuations because of adverse events (see table 7). Two SAEs were recorded; one was an acute radial nerve palsy and the other a left toe infection/sepsis/UTI. Most common laboratory abnormality was elevated bilirubin in patients receiving atazanavir/ritonavir. No patient with confirmed on-treatment HIV virologic rebound was recorded. In summary, this new combination was highly efficacious and safe, providing a simple, safe, and highly effective treatment for patients coinfected with HIV-1 and HCV.
Table 7: ASTRAL-5 study: Overall safety reports
EASL: Sofosbuvir/Velpatasvir for 12 Weeks in Patients Coinfected With HCV and HIV-1: The ASTRAL-5 Study - (04/18/16)
Patients with inherited blood disorders
Treatment of HCV infection in patients with inherited blood disorders (IBLDs) in the past has been quite complex. Historically, the use of ribavirin has been restricted due to the risk of hemolytic anemia and frequent comorbidities in many patients with IBLD have excluded them from interferon based HCV treatment approaches. Clearly, interferon- and ribavirin-free direct-acting antiviral regimens offer new treatment opportunities for patients with IBLDs and HCV infection. At EASL for the first time a large study was presented which assessed the safety and efficacy of EBR/GZR in patients with HCV infection and sickle cell anemia, β-thalassemia, or hemophilia/von Willebrand disease (6). The study design is shown in figure 12.
Figure 12: Study design of the C-EDGE study in IBLD
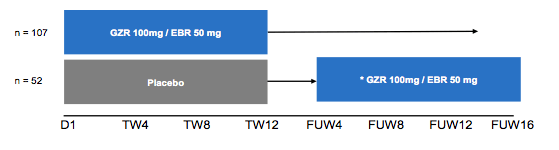
The C-EDGE study in IBLD patients was a randomized, parallel-group, multi-site, placebo-controlled trial. Patients were stratified by cirrhosis (yes/no) and disease status (sickle cell anemia vs. thalassemia vs. hemophilia/von Willebrand disease). Overall, 159 patients were randomized to immediate treatment with EBR/GZR or deferred treatment where patients received placebo for 12 weeks and then open-label EBR/GZR starting at FUW4. The baseline demographics of the study population are depicted in table 8.
Table 8: Baseline demographics of the C-EDGE study in IBLD.
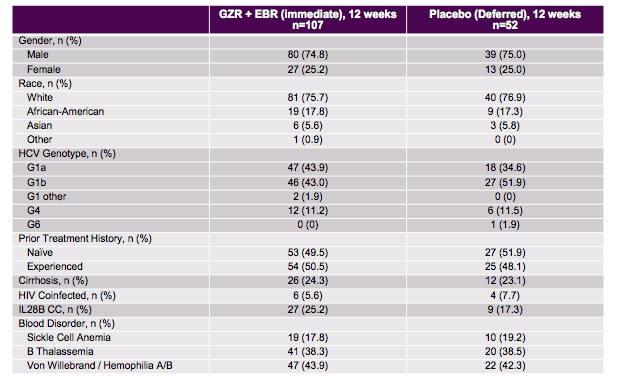
Of note, most patients were male and had a genotype 1 infection. Almost a quarter was already cirrhotic, half of the patients had already failed a prior HCV therapy. The main outcome, SVR12 rates is shown below in figure 13.
Figure 13: Primary Efficacy Analysis, Immediate Treatment Group, Full Analysis Set
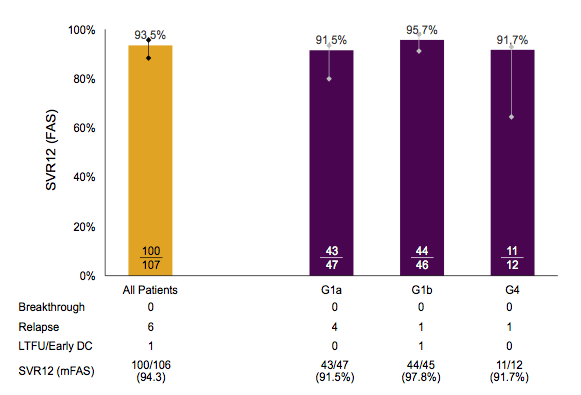
Overall, SVR12 rates were very high. Most of the relapses occurred in GT1a patients. Among patients with HCV GT1a infection, 42/43 (98%) patients without baseline NS5A resistance-associated variants (RAVs) achieved SVR12. Baseline NS3 RAVS had no impact on SVR12 in patients with HCV GT1a infection. Interestingly, baseline resistance associated variants with reduced susceptibility to elbasvir by population sequencing were associated with an increased risk for developing virological failure (see figure 14).
Figure 14: C-EDGE IBLD Study: Prevalence of NS5A at Baseline and impact on SVR12
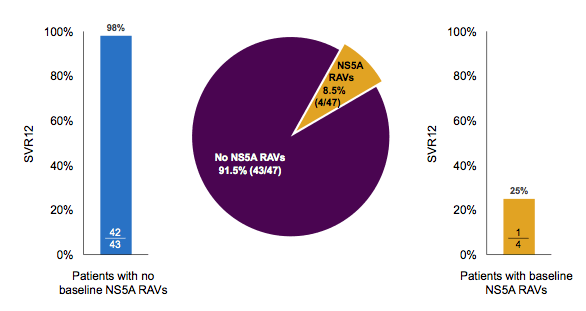
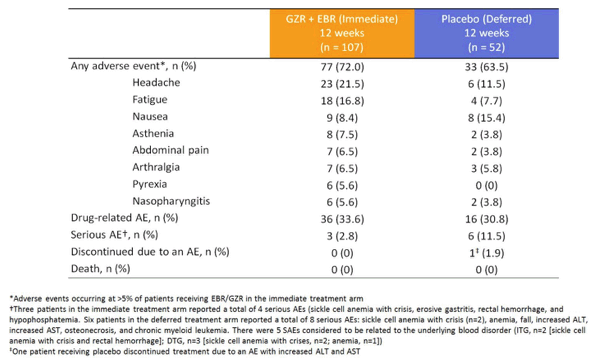
These findings are very much in line within the American label of grazoprevir/elbasvir and highlight why it can make sense to have a genotypic resistance ordered prior to commencing this therapy, even if the likelihood of having these RAVs in GT1a patients overall is low. The overall safety was excellent and no patient discontinued his HCV therapy due to adverse events in the immediate treatment arm. Table 9 summarizes the main adverse events observed in this trial.
Table 9: Adverse events
EASL: C-EDGE IBLD: Efficacy and Safety of Elbasvir/Grazoprevir in Patients With Chronic Hepatitis C Virus Infection and Inherited Blood Disorders - (04/28/16)
Another understudied patient group are patients with kidney transplantation and chronic HCV infection (7). As kidney transplantation from a HCV positive donor increases the likelihood of receiving an organ HCV therapy obviously post transplantation is of great importance, various patients do not want to be treated for HCV prior to transplantation. Hepatitis C virus infection however, has been associated with poor outcomes in kidney transplant recipients. Moreover, treatment with interferon and ribavirin in HCV patients receiving kidney transplants was complicated by risks including allograft rejection and poor tolerability. Therefore, effective and safe IFN-free treatment options are urgently needed in kidney transplant recipients. This study evaluated the safety and efficacy of LDV/SOF
for 12 or 24 weeks in kidney transplant recipients with HCV GT 1 or 4 infection. The study design is shown below in figure 15.
Figure 15: Study design Post kidney transplant study
This was a phase 2, randomized, open label multicenter study with 5 sites in Italy, France, Austria and Germany. The study enrolled kidney transplant recipients with HCV GT 1 or 4, who were treatment-naïve or -experienced, with or without cirrhosis. Cirrhosis was determined by biopsy (Metavir score =4 or Ishak score ≥5), FibroScan® > 12.5 kPa or FibroTest >0.75 + APRI >2. Key inclusion criteria were >6 months from kidney transplant, HCV RNA ≥LLOQ (15 IU/mL) at screening, hemoglobin ≥10 g/dL, platelets >50 x 103/μL, and CLcr ≥40 mL/min. The main patient characteristics are shown in table 10.
Table 10: Baseline characteristics
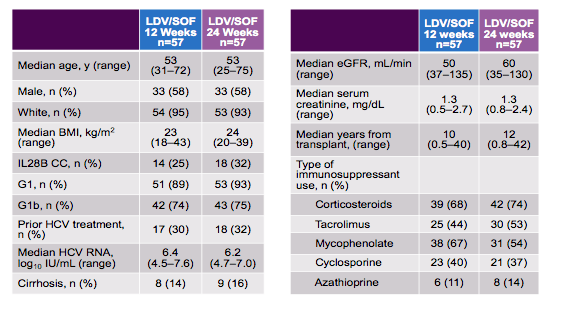
The majority of patients had a GT1b infection, a third was treatment experienced and only few patients had cirrhosis. The patients already on average had a decreased GFR and were on a mean number of three immunosuppressants. The main virological outcome findings are depicted in figure 16.
Figure 16: Results of the kidney transplant recipient study
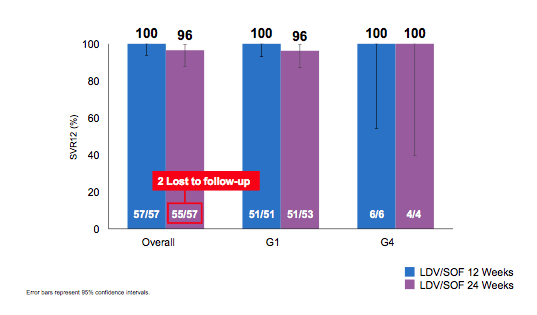
LDV/SOF for 12 weeks resulted in 100% SVR12 in HCV-infected kidney transplant recipients with or without cirrhosis and/or history of prior treatment failure. Clearly there is no need to extend therapy to 24 weeks. The safety findings are summarized in Table 11. LDV/SOF was generally safe and well tolerated in kidney transplant recipients with HCV infection. No clinically meaningful reductions in renal function were observed.
Table 11: Overall Safety: GT 1 and 4, Post-Kidney Transplant study

Overall, 4 patients had eGFR<30mL/min during therapy which however was only found to be treatment related in one patient. No episodes of rejection were reported during the study.
EASL: Ledipasvir/Sofosbuvir (LDV/SOF) for 12 or 24 Weeks Is Safe and Effective in Kidney Transplant Recipients With Chronic Genotype 1 or 4 HCV Infection - (04/19/16)
Resistance associated variants and their impact on HCV treatment outcome.
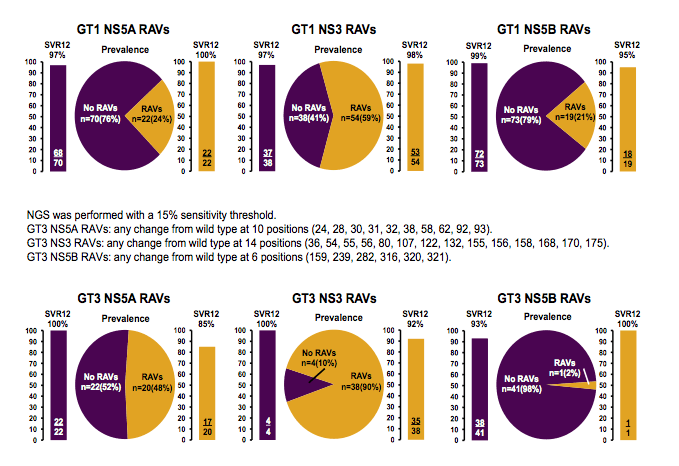
Many presentations at this year EASL dealt with the prevalence of resistance associated variants (RAVs) at baseline prior to commencing HCV therapy and what impact these RAVs could potentially have on subsequent treatment outcome (8). In addition studies assessed which RAVs were present after DAA-based treatment failure and how on the basis of documented or presumed resistance new regimens for retreatment can be potentially selected (9). A sub-study from the multi-center, prospective, observational TARGET cohort study aimed to evaluate the prevalence and impact of baseline (BL) resistance-associated variants (RAVs) on ledipasvir/sofosbuvir (LDV/SOF) ± ribavirin (RBV) or simeprevir/sofosbuvir (SMV/SOF) ± RBV regimens in patients with genotype (GT) 1 HCV infection (8). HCV resistance testing was performed on samples collected before May 12, 2015 using Monogram Biosciences assays (population sequence derived from Illumina MiSeq data with a 10% variant reporting threshold). LDV, SOF and SMV susceptibility was interpreted using Monogram's rule-based algorithm. The baseline characteristics of the tested individuals from the TARGET cohort study are shown below in Table 12.
Table 12: HCV RAVs in the HCV TARGET Cohort: Cohort Characteristics
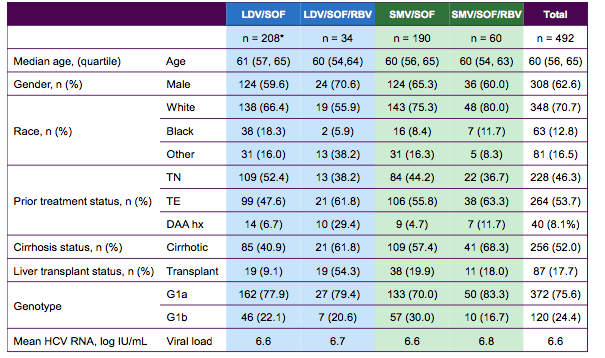
The majority of patients were infected with GT1a and around half of the patients had cirrhosis. The overall prevalence of NS5A and NS3 RAVs is shown in figure 17. The Y93H/M was the most frequent NS5A RAV observed in GT1b patients. Overall, more patients with GT1b had NS5A RAVS than GT1a patients. The most common NS3 mutation was the Q80K in GT1a patients.
Figure 17: Baseline RAVs in TARGET
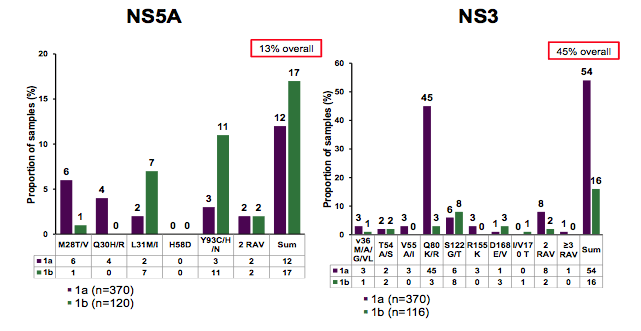
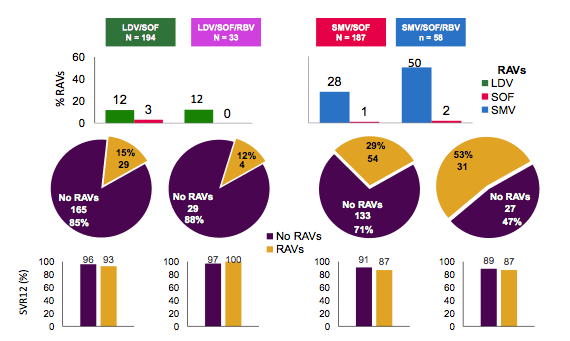
NS3 associated mutations were more common than NS5b mutations. The prevalence of RAVs
was generally comparable between treatment-naïve and treatment-experienced patients, and between cirrhotic and non-cirrhotic patients. Figure 18 shows the prevalence and impact of RAVs on subsequent SVR12 rates. Overall, it looked as if patients with and without RAVs had similar SVR12 rates.
Figure 18: Impact of RAVs on SVR in TARGET
EASL: Prevalence and Impact of Baseline Resistance-Associated Variants (RAVs) on the Efficacy of Ledipasvir/Sofosbuvir or Simeprevir/Sofosbuvir Against GT1 HCV Infection: HCV-TARGET Interim Analysis - (04/21/16)
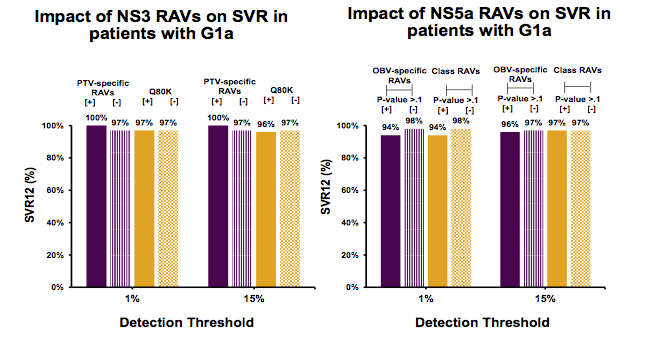
A further study on resistance assessed the prevalence of RAVs and impact on ombitasvir/paritaprevir/ritonavir and dasabuvir ± ribavirin (3D) response, using GT1a and GT1b baseline samples from Phase 3 studies with the 3D regimen (18). Next generation sequencing was conducted using Illumina MiSeq on baseline samples from naïve (PEARL-IV), treatment-experienced (SAPPHIRE-II), or cirrhotic (TURQUOISE-II; 24 week treatment arm) GT1a patients who received 3D + RBV, and treatment-experienced (PEARL-II) or cirrhotic (TURQUOISE-III) GT1b patients who received 3D without RBV. Patients who discontinued treatment early for non-virologic reasons were excluded. The prevalence and impact of baseline RAVs on SVR rates were determined using detection thresholds of 1% and 15%. The impact of baseline RAVs conferring ≥5-fold resistance to components of the 3D regimen in NS3, NS5A, and NS5B on treatment outcome was determined by comparing SVR rates in patients with or without detectable RAVs. The prevalences determined for the two detection thresholds are depicted in figure 19.
Figure 19: Prevalence of NS3A RAVS and prevalence of NS5A RAVS in GT1a Patients
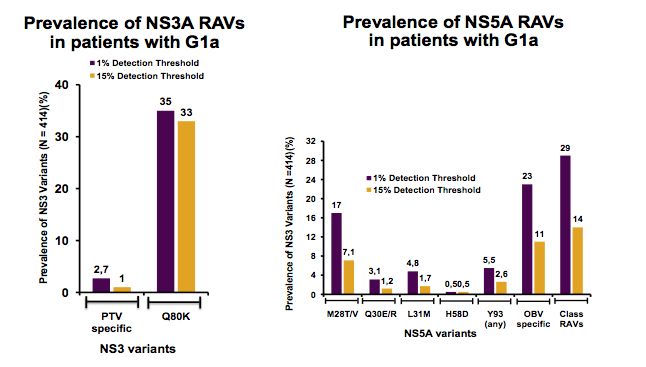
Of note, when using the 1% detection threshold the number of RAVs was much higher. Much more clinically relevant are cutoffs at 10 or 15%. In this study SVR rates were 96% in patients with GT1a and 100% in patients with GT1b. Using the 15% detection threshold, one or more NS5A RAVs were present in 11% of treatment-experienced or cirrhotic GT1a patients; whereas NS5A RAVs were found in 19% of GT1b patients. Similar SVR rates were seen in GT1a patients with or without NS5A RAVs. All GT1b patients with NS5A RAVs, including at position Y93, achieved SVR. NS3 PTV specific RAVs were uncommon in both subtypes (≤2%). NS5B RAVs were more common in GT1b than GT1a. NS3 RAVs were not seen in any of the 14 virologic failures and an NS5B RAV was seen only in 1 of 14 virologic failures. The presence of the GT1a NS3 Q80K polymorphism had no impact on SVR. Patients harboring RAVs across multiple viral targets were rare; all achieved SVR. The impact of NS3 and NS5A RAVs on SVR for GT1a patients is summarized in figure 20.
Figure 20: Impact of NS3 and NS5A RAVs on SVR rates in patients with GT1a infection for the different detection thresholds used.
EASL: Effect of Baseline Resistance-Associated Variants on SVR With the 3D Regimen Plus RBV - (04/18/16)
Finally, there was an analysis from a large European database in Frankfurt which has samples and resistance information on 3549 individuals (9). This particular sub-study focused on persons who failed DAAs outside of clinical trials (N=310) (9). Given the emergence and persistence of resistance associated variants (RAVs) retreatment in these patients remains a challenge. Only patients with guideline recommended interferon-free DAA regimens were included. Post-failure serum samples were analyzed for the presence of RAVs by direct sequencing of the NS3, NS5A and NS4B genes. The main baseline characteristics for this study population are shown in table 12.
Table 12: Baseline demographics for the various DAA-combinations given.
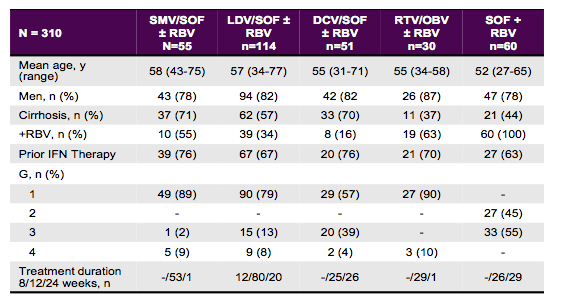
Overall, failures following five different DAA-based therapies were studied: SMV/SOF ± RBV (N=55), LDV/SOF ± RBV (n=114), DCV/SOF ± RBV (n=51), RTV/OBV ± RBV (n=30) and SOF + RBV (n=60). On average, 63-76% of patients had already failed prior IFN/RBV therapy and more than half of the patients were already cirrhotic underlining that this was a truly more difficult to treat patient population altogether. The distribution of RAVs for patients with GT1 and NS5A treatment failure (DCV or LDV + SOF) and for patients with G1 and SMV/SOF treatment failure is shown below in figure 21 a and b respectively.
Figure 21a): Distribution of RAVs for NS5A failures and b) of RAVs for SMV/SOF failures
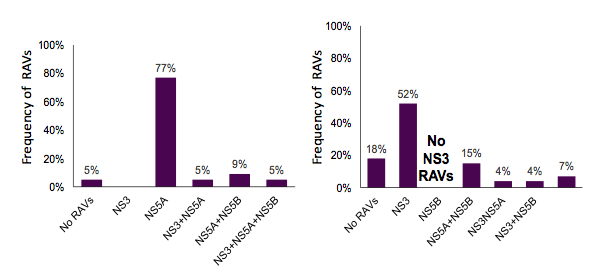
Only 5% of patients who failed a NS5A containing DAA combination showed no RAVs at all. 77% of patients who had failed an NS5A containg DAA combination showed only NS5A RAVs. 22 of 119 patients with G1 and NS5A treatment failure (DCV or LDV + SOF) were retreated with a PI containing regimen of SMV/SOF +/- RBV or 3D +/- RBV for
12 or 24 weeks. Interim analysis shows that a SVR12 so far has been achieved in 6 of 7 patients (limited data). For patients failing on a SMV/SOF based HCV treatment combination 18% showed no RAVs at all. The majority had NS3 RAVs only. 27 of 49 patients with G1 and SMV/SOF treatment failure were retreated with a NS5A containing regimen (LDV/SOF (n=23) or 3D (n=4) +/- RBV). So far 20 out of 22 patients undergoing retreatment achieved SVR12. The distribution of RAVs for patients with GT1 and 3D treatment failure and for patients with G3 and SOF/RBV treatment failure is shown below in figure 22 a and b respectively.
Figure 22a): Distribution of RAVs for 3D failures and b) of RAVs for SOF/RBV failures
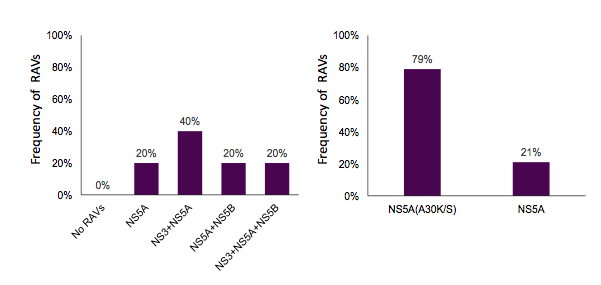
40% of patients failing the 3D regimen showed a combination of NS3 and NS5A RAVs. So far 5 out of 27 patients with G1 and 3D treatment failure have been retreated with a SOF based regimen but up to now no SVR12 data is available. In GT 3 patients failing SOF/RBV 79% showed a NS5A resistance associated strain. 14 of 23 patients with genotype 3 and SOF/RBV treatment failure were retreated with DCV (n=13) or LDV (n=1) + SOF +/- RBV.
Preliminary data shows SVR12 in 7 of 7 retreated patients.
EASL: Retreatment of Patients who failed DAA-Combination Therapies: Real-World Experience from a Large Hepatitis C Resistance Database - (05/27/16)
Overall, it appears that RAVs which can be found after virological failure under DAA combination therapy strongly reflect the selective pressure of the DAAs used for HCV therapy. Retreatment with fully active agents from different drug classes + mostly SOF is associated with very promising HCV cure rates despite extensive RAVs after virological failure under DAA combination therapy. As this approach (retreatment with different drug class) can be done even in the absence of a resistance test results this strategy appears helpful in settings where no access to resistance testing is available. Factors associated with virological failure in this dataset were male sex, cirrhosis and prior treatment failure.
New DAAs under regulatory approval
New data was also presented for the upcoming fix-dose combination of sofosbuvir/velpatasvir (SOF/VEL) which is currently undergoing regulatory approval and is expected to become available in the US shortly. The once-daily fixed-dose combination tablet of SOF/VEL was evaluated for the treatment of genotype 1-6 HCV infection in three phase 3 studies in patients with and without compensated cirrhosis (ASTRAL-1, ASTRAL-2, ASTRAL-3). One of the EASL presentations evaluated the safety of SOF/VEL across these 3 Phase 3 studies (19). Treatment-emergent adverse events (AEs) and laboratory abnormalities were assessed in patients randomized to SOF/VEL or placebo for 12weeks in ASTRAL-1, SOF/VEL or SOF + RBV for 12weeks in ASTRAL-2 and SOF/VEL for 12 weeks or SOF + RBV for 24 weeks in
ASTRAL-3. Data was pooled by treatment regimen. Figure 23 summarizes the adverse events which were observed in more than 10% of patients as well as all grade 3/4 chemistry abnormalities.
Figure 23: Adverse events and laboratory abnormalities within the ASTRAL 1-3 studies.
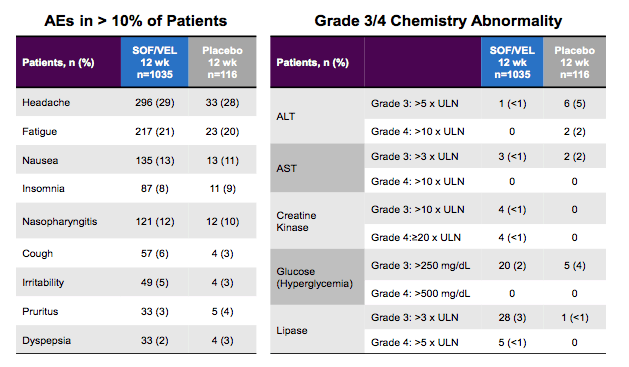
Of note as the ASTRAL studies included a placebo arm the possibility exists to directly compare SOF/VEL with placebo with regard to safety profile. No significant difference was observed between the DAA fix-dose combination and the placebo arm underlining that adverse events also occur at a certain rate in placebo treated patients. The same was also true for lab abnormalities. If at all there were more grade 3 liver enzyme elevations in the placebo group which makes sense as in the placebo arm no active HCV treatment was given. These data clearly underline the very favorable safety profile of this new regimen.
EASL; The Tolerability of Sofosbuvir/Velpatasvir for 12 Weeks in >1000 Patients Treated in the ASTRAL-1, ASTRAL-2, and ASTRAL-3 Studies: An Integrated Safety Analysis - (04/22/16)
A further analysis from the phase III ASTRAL 1-3 studies aimed to explore whether early viral response was predictive of eventual treatment success. This was a retrospective analysis of data from 1,035 patients with genotype 1-6 HCV infection treated with SOF/VEL for 12 weeks in the Phase 3 ASTRAL 1, -2 and -3 studies (20). Patient plasma samples were analyzed using the COBAS Ampliprep/ COBAS Taqman v2.0 with LLOQ = 15 IU/mL. SVR12 rates for subjects with HCV RNA ≥ LLOQ, HCV RNA < LLOQ, and HCV RNA < LLOQ not detected at treatment week 1, 2 and 4 were calculated. Subject who did not achieve SVR12 due to non-virologic reasons (lost to follow-up, death, withdrew consent etc.) were excluded from the analyses. Across all HCV genotypes, there were rapid and sustained declines in HCV RNA during treatment with SOF/VEL. HCV RNA < LLOQ at week 1, 2 and 4 were observed in 18%, 58% and 91% of patients, respectively. 22% and 66% had HCV RNA < LLOQ TND at Weeks 2 and 4, respectively. No difference in SVR rate according to HCV RNA level at week 4 was observed (see figure 24). Also 100% concordance between SVR12 and SVR24 was noted.
Figure 24: SOF/VEL for 12 weeks: On-treatment HCV RNA at treatment week 4 is not predictive of outcomes
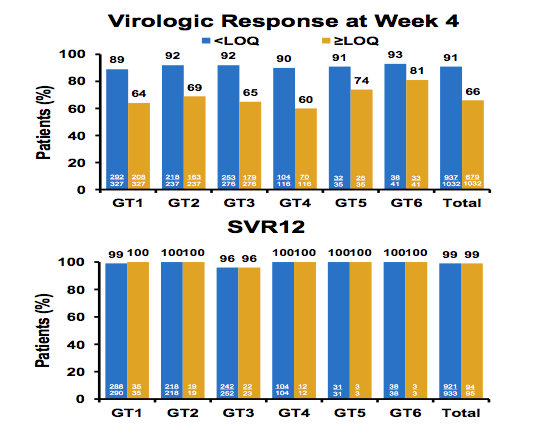
EASL: On-Treatment HCV RNA as a Predictor of SVR12 in Patients With Genotype 1-6 HCV Infection Treated With Sofosbuvir/Velpatasvir for 12 Weeks: An Analysis of the ASTRAL-1, ASTRAL-2, and ASTRAL-3 Studies - (04/26/16)
A further study reported findings from the Phase 1 program which evaluated drug-drug interactions between SOF/VEL or its components and HIV antiretrovirals (ARVs), oral contraceptives (OCs), acid-reducing agents, immunosuppressants, opiates, and drug transporter and CYP probes (21). The effect of concomitant medications used in Phase 2/3 studies on SOF/VEL PK was also assessed by population PK analyses. No clinically relevant changes in atazanavir (ATV)/ritonavir (r), cobicistat, darunavir/r, dolutegravir, efavirenz (EFV), elvitegravir, emtricitabine, lopinavir/r, raltegravir, rilpivirine, or tenofovir alafenamide (TAF) PK were seen when coadministered with SOF/VEL. SOF/VEL increased tenofovir (TFV) exposure (∼40 to 81%) when administered as TDF, but not as TAF. Interestingly, this is sort of in the range of the ledipasvir and tenofovir drug-drug interaction. It is reassuring to see that with the availability of the tenofovir prodrug formulation (TAF) this interaction can be overcome. No clinically relevant changes in SOF or GS-331007 (primary circulating metabolite) PK were observed with ARVs. Efavirenz decreased VEL AUC 53% and ATV/r increased VEL AUC 142%. A small increase in ethinyl estradiol (EE) Cmax (39%) and decrease in Ctau (17%) with no change in AUC occurred when representative OC EE/norgestimate was administered with VEL; norgestrel AUC and Ctau increased (19% and 23%) when was administered with SOF. Another interesting class of drugs studied were H2 receptor blockers and PPIs as the interaction between PPIs and ledipasvir has raised some concern (see above). The ph-based interactions with acid lowering drugs are summarized in table 13.
Table 13: pH-based Interactions: Acid-reducing agents
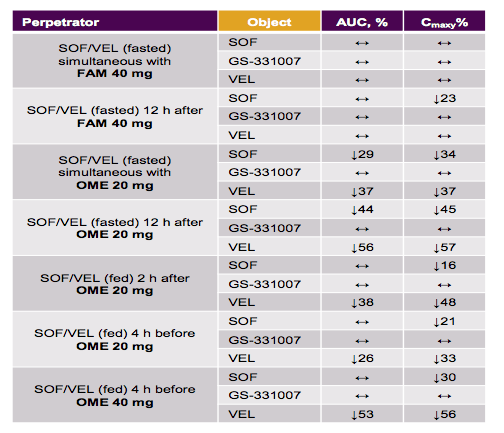
Histamine-2 receptor antagonists (famotidine 40 mg BID) did not impact SOF/VEL exposure. Omeprazole (OME) 20 mg resulted in a decrease in VEL AUC (26-38%) and no impact on SOF or GS-331007 AUC when given with food. Larger decreases in VEL AUC were observed with OME 40 mg (56%) or when administered under fasting conditions (36-57%). Therefore, it appears the safest to follow the same rules as with LED/SOF when there is a need for concomitant PPi therapy (see above; at best 2mg dose together with HCV drugs). Sofosbuvir and VEL AUC decreased 72% and 82%, respectively, when coadministered with rifampin, a potent P-gp and CYP inducer. Pravastatin and rosuvastatin AUC increased 35% and 169%, respectively, when coadministered with VEL. A small increase in digoxin AUC (34%) was observed with VEL. No clinically relevant interactions were observed upon administration of VEL with CsA or SOF with CsA, TAC, or methadone. Anticoagulants, SSRIs, calcium channel blockers, diuretics, or rifaximin did not affect SOF/VEL PK in Phase 2/3 studies. In summary, SOF/VEL exhibits a favorable DDI profile allowing use with various drugs commonly used by HCV-infected patients. The same recommendations for PPIs should be used for concomitant therapy with SOF/VEL as with LED/SOF.
EASL: Drug-Drug Interaction Profile of Sofosbuvir/Velpatasvir Fixed-Dose Combination - (04/21/16)
The next wave of pangenotypic DAA therapies
As the availability of broad pangenotypic DAAs still remains scarce and in particular GT3 treatment especially in treatment experienced cirrhotics remains challenging there is still a search for more potent broad pangenotypic combinations. Obviously, this is of particular importance for the resource limited setting as the availability of a simple broad genotypic HCV therapy would minimize the need for complex baseline virological tests and allow easier access to therapies which could be delivered in simple clinic settings. One of the exciting new combinations with various trial results being presented is the new 2nd generation HCV protease inhibitor ABT-493 (NS3/4A protease inhibitor identified by AbbVie and Enanta) and the NS5A inhibitor ABT-530. Both compounds have demonstrated potent pangenotypic antiviral activity in vitro, with a high barrier to resistance and maintenance of activity against common variants. In Part 1 of the SURVEYOR-I and SURVEYOR-II studies, ABT-493 + ABT-530 for 12 weeks was well tolerated and achieved sustained virologic response (SVR) rates between 97-100% in non-cirrhotic patients with HCV genotype (GT) 1 or 2 infection (22,23). In Part 2 of these studies, ABT-493 + ABT-530 was co-administered for a shorter duration of 8 weeks. The results of this trial were reported here at EASL (24). The study design for the SURVEYOR part 2 studies is shown below in figure 25.
Figure 25: Study design SURVEYOR 1 and 2
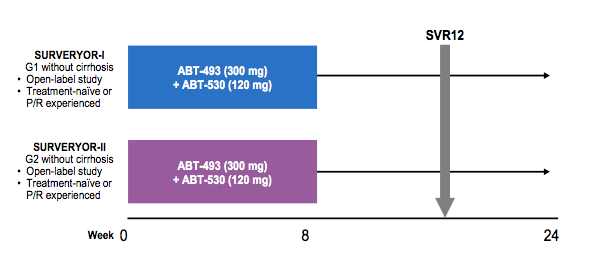
Non-cirrhotic treatment-naïve patients or pegylated interferon/ribavirin treatment-experienced non-responders received once-daily ABT-493 300 mg + ABT-530 120 mg for 8 weeks. In Part 2 of SURVEYOR-I and -II, 34 patients with GT1 infection (71% GT1a; 68% non-CC IL28B genotype; 15% treatment-experienced) and 54 patients with GT2 infection (70% GT2b; 59% non-CC IL28B genotype; 13% treatment-experienced) were enrolled, respectively. Mean baseline HCV RNA log10 IU/mL ± standard deviation was 6.3 ± 1.1 for GT1-infected patients and 6.6 ± 0.8 for GT2-infected patients, with 38% and 57% of patients who had baseline levels ≥6 M IU/mL, respectively. The SVR results are shown below in figure 26.
Figure 26: SVR results of SURVEYOR I and II by HCV genotype (ITT and mITT analyses).
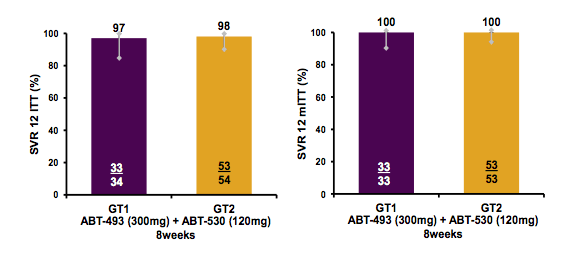
Overall SVR rates were very impressive with no virological failure documented. One GT1-infected patient discontinued study prematurely at week 4 (with undetectable HCV RNA) due to a non-DAA related serious adverse event (AE) of abdominal cancer of unknown origin and subsequently died prior to reaching the SVR12 time point. The GT2-infected patient without SVR4 was lost to follow up after week 6, when HCV RNA was not detected. There were no other discontinuations due to AEs. Across both studies, AEs were mostly mild (Grade 1), with the most common AEs being fatigue and headache. In summary, the combination of ABT-493 and ABT-530 administered for 8 weeks in non-cirrhotic patients with HCV GT1 or GT2 infection was well tolerated and achieved SVR rates of 97-98%, with no virologic failures, regardless of baseline viral load or prior treatment history. Despite the small numbers this is quite impressive as baseline VL does impact SVR with the shorter treatment duration of 8 weeks in ledipasvir/sofosbuvir treated patients. Also this combination promises cure in all GT2 patients without ribavirin and at shorter treatment durations than with velpatasvir/sofosbuvir.
EASL: High SVR Rates With the Combination of ABT-493 + ABT-530 for 8 Weeks in Non-Cirrhotic Patients With HCV Genotype 1 or 2 Infection - (04/18/16)
The next interesting study with the ABT-493 and ABT-530 combination was a pilot) trial in G1 patients with cirrhosis (26). Obviously, cirrhotic patients can be more difficult to cure so how did this new combination perform here. Treatment-naïve or pegylated interferon/ribavirin treatment-experienced patients with GT1 infection and cirrhosis received ABT-493 200 mg + ABT-530 120 mg once daily for 12 weeks. Cirrhosis was determined by either liver biopsy (Metavir F4), Fibroscan (liver stiffness >14.6 KPa) or serum markers (Fibrotest score ≥0.75 and an APRI > 2). Overall, 27 patients were enrolled, 74% were male, 89% white, 74% had a G1a and 85% had a non-CC IL28B genotype. 26% were HCV treatment-experienced all had F4 fibrosis. The SVR rates obtained are shown below in figure 27.
Figure 27: SVR rate in GT1 cirrhotics receiving 12 weeks of ABT-493 and ABT-530
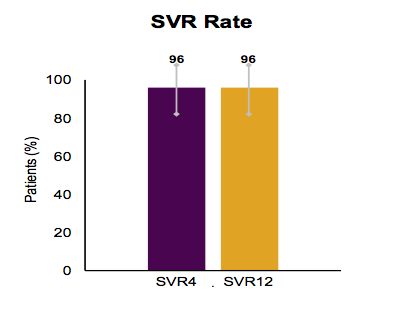
Only, one patient experienced relapse at post-treatment week 4. All adverse events (AEs) were deemed mild or moderate in severity, with no patients reporting severe or serious AEs considered related to study drugs. No patients discontinued treatment prematurely due to AEs and the most frequent AEs reported in >10% of patients were fatigue (11%) and headache (11%). No clinically meaningful abnormal liver function or other laboratory results were observed. In conclusion, very encouraging efficacy and safety data in this small number of patients was reported. Clearly, larger trials are needed to confirm these very promising initial results.
EASL: High Efficacy and Favourable Safety of ABT-493 + ABT-530 Co-Administration for 12 Weeks in HCV Genotype 1-Infected Patients With Cirrhosis (SURVEYOR-I) - (04/18/16)
The next interesting patient population to be studied was a non-cirrhotic genotype 3 patient group (11). In Part 1 of the Phase 2 SURVEYOR-II study, once daily ABT-493 300 mg + ABT-530 120 mg for 12weeks was well tolerated and achieved a sustained virologic response (SVR) in 96% (26/27) of treatment-naïve, non-cirrhotic GT3-infected patients, without virologic failures (26). Part 2 of this study was now presented at EASL which evaluated shorter treatment duration of 8 weeks (11). 29 treatment-naïve HCV GT3-infected patients without cirrhosis received once daily ABT-493 300 mg + ABT-530 120 mg. 52% were male, 90% white, 86% had GT3a, and 62% non-CC IL28B. The median (range) HCV RNA log10 IU/mL was 6.3 (5.0-7.5) and 24% of patients had HCV RNA ≥6M IU/mL at baseline. The SVR12 rates are shown below in figure 28.
Figure 28: SVR 12 rates in GT3 non-cirrhotics by ITT and mITT analysis
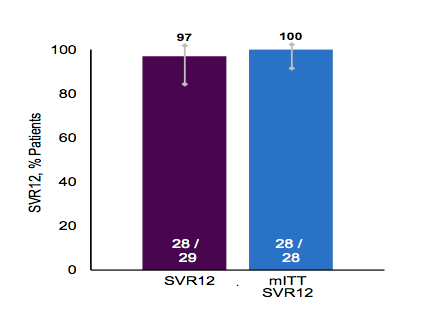
No patients experienced virologic failure. One patient discontinued the study after treatment week 6 (HCV RNA undetectable at this visit) due to intolerance of blood draws. No patients discontinued due to adverse events (AEs) or experienced serious AEs. The majority of AEs were mild in severity, with the most common AEs (>10% of patients) reported for patients being headache and fatigue. In summary, in this limited number of patients almost the best efficacy data until today has been generated in very short treatment duration of 8 weeks again strongly encouraging larger trials with this compound in the GT 3 population.
EASL: HIGH SVR RATES WITH ABT-493 + ABT-530 CO-ADMINISTERED FOR 8 WEEKS IN NON-CIRRHOTIC PATIENTS WITH HCV GENOTYPE 3 INFECTION - (04/18/16)
The next obvious question is how this new combination performs in cirrhotic, treatment experienced GT3 patients as this remains one of the more challenging to treat patient populations to date. Efficacy and safety of ABT-493 + ABT-530 with or without ribavirin (RBV) in GT3-infected patients with cirrhosis were studied accordingly (27). Treatment-naïve GT3-infected patients with compensated cirrhosis were randomized to receive once-daily 300 mg ABT-493 + 120 mg ABT-530, either with or without once daily 800 mg RBV for 12 weeks. Baseline samples were sequenced to assess the presence of resistance-associated variants (RAVs) in NS3 or NS5A. Table 14 summarizes the main results of the HCV sequencing at baseline. Overall, forty-eight patients with cirrhosis were enrolled (65% non-CC IL28B genotype, 65% male, mean age 55, mean viral load 6.3 log10 IU/mL).
Table 14: Baseline Variants in NS3 and NS5A
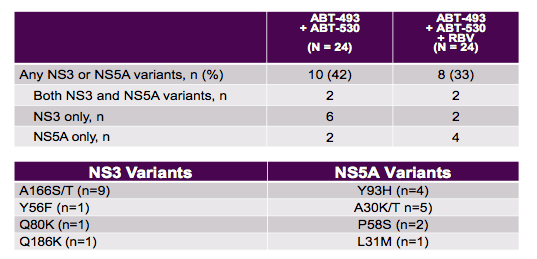
Baseline sequencing results were available for samples from 46 patients: 7 (15%) had variants in NS3 only, 7 (15%) had variants in NS5A only, and 3 (7%) had variants in both targets. The final SVR12 rates are shown in figure 29.
Figure 29: SVR rates by ITT analysis
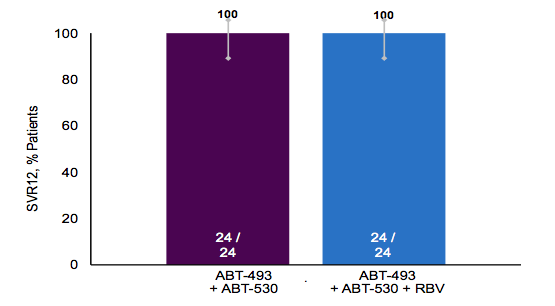
Most impressively, all patients obtained SVR12. Ribavirin had no additional effect on SVR rate. Also presence of RAVs seemed to have no impact on treatment outcome after 12 weeks of combination therapy. The most common adverse events (AEs) were more frequently observed in the RBV-containing arm than the RBV-free arm (headache 33 vs 13%, fatigue 25 vs 8%, and nausea 21% vs 8%, respectively). Three patients experienced serious AEs, of which 1 event (delusional disorder) was considered related to DAAs and RBV (in a patient with admitted amphetamine and alcohol use) and 1 event (anaemia) was considered related to RBV. There were no discontinuations due to AEs and no patients experienced aminotransferase elevations above baseline. In summary, ABT-493 + ABT-530 with or without RBV for 12 weeks was well tolerated in treatment-naïve GT3-infected patients with cirrhosis, achieving SVR4 rates of 100% regardless of the presence of baseline NS3 and/or NS5A variants. Clearly, this combination holds a lot or promise in treatment of GT3 patients. Nevertheless data in treatment experienced cirrhotics is missing which in the end are the most difficult to treat patient group.
EASL: 100% SVR12 WITH ABT-493 AND ABT-530 WITH OR WITHOUT RIBAVIRIN IN TREATMENT-NAÏVE HCV GENOTYPE 3-INFECTED PATIENTS WITH CIRRHOSIS - (04/19/16)
More data on the efficacy and safety of ABT-493 + ABT-530 was presented from non-cirrhotic genotype 4,5 and 6 patients (28). Treatment-naïve or pegylated interferon/ribavirin treatment-experienced patients received once-daily ABT-493 300 mg + ABT-530 120 mg for 12 weeks. A total of 34 patients with genotype 4 (n = 22; 65%), 5 (n = 1; 3%), or 6 (n = 11; 32%) infection were enrolled: 53% male, 59% white, 62% had non-CC IL28B, and 15% were treatment-experienced. The median (range) HCV RNA log10 IU/mL was 6.4 (4.6-7.4) at baseline, and 35% of patients had HCV RNA ≥6,000,000 IU/mL. All patients achieved SVR12 (see figure 30) underlining the great efficacy of this DAA combination. Clearly more GT 5 and 6 patients need to be studied.
Figure 30: SVR12 rates for GT4,5 and 6 patients receiving ABT-493 and ABT-530
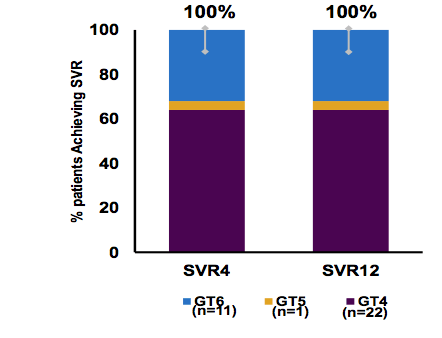
EASL: 100% SVR4 and Favorable Safety of ABT-493 + ABT-530 Administered for 12 Weeks in Non-Cirrhotic Patients With Genotypes 4, 5, or 6 Infection (SURVEYOR-I) - (04/18/16)
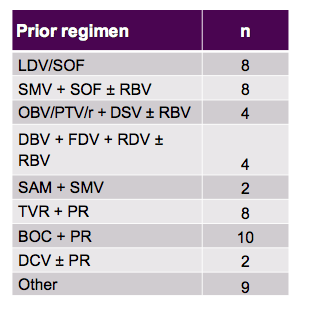
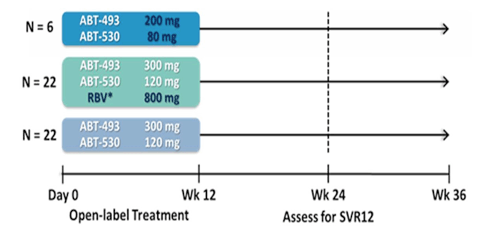
The last group of patients to be studied were patients with GT1 infection who already failed a previous DAA-based HCV therapy (10). Obviously, especially if this was a failure under DAA combination therapy this is a very difficult to treat patient population with a high probability of being enriched with resistance associated variants at baseline which may impact over all SVR chances. The MAGELLAN-I study evaluated the efficacy and safety of the combination regimen of the NS3/4A protease inhibitor ABT-493 plus the NS5A inhibitor ABT-530 with or without ribavirin (RBV) in HCV genotype 1 (GT1)-infected patients without cirrhosis who had failed DAA-containing regimens that included a protease inhibitor and/or NS5A inhibitor, with or without an NS5B polymerase inhibitor. Patients were randomized to receive once-daily ABT-493 and ABT-530 at doses of 200 + 80 mg(Arm A), 300 + 120 mg + 800 mg RBV (Arm B), or 300 + 120 mg (Arm C), respectively, for 12 weeks. Patients who failed previous treatment for reasons other than breakthrough or relapse were excluded. Deep sequencing (Illumina MiSeq) was performed on HCV NS3 and NS5A genes from samples collected from all patients at baseline, and at the time of virologic failure. The previous DAA regimens are shown in table 15.
Table 15: Prior DAA-based regimens
Fifty patients were randomized, of whom 42 (84%) had GT1a infection and 33 (66%) had treatment experience with regimens that contained 2 or 3 DAAs. Deep sequencing revealed baseline resistance associated variants (RAVs) in 41 (82%) patients, 15 in NS3, 10 in NS5A, and 16 with RAVs in both targets. Figure 31 summarizes the study design.
Figure 30: Study design of the MEGALLAN-I study.
Arm A enrolment was stopped early to investigate higher doses of study drugs in other arms. In ITT analysis SVR12 was achieved in 6/6 (100%) arm A patients, 20/22 (91%) arm B patients and 19/22 (86%) arm C patients. Two virologic failures were observed; 1 relapse in an Arm B patient with baseline NS5A RAVs, and 1 breakthrough at treatment week 8 in Arm C in a patient with Crohn's disease on immune suppressant therapy, and with baseline NS3 and NS5A RAVs. Three patients were lost to follow-up (LTFU) (1 LTFU after week 6 with HCV RNA undetectable and 2 patients LTFU after completing treatment
(1 death); both achieved SVR8). The most common adverse events (AEs) were headache (28%), fatigue (26%), and nausea (20%). Two patients experienced serious AEs assessed as unrelated to study drug or RBV (breast cancer and femoral fracture). Overall, very impressive SVR rates in DAA failures with a high percentage of RAVs at baseline. The SVR12 findings for all 3 arms are shown in figure 31.
Figure 32: SVR12 rates (ITT and mITT) in the MEGALLAN-1 study (Arm1-A,B and C).
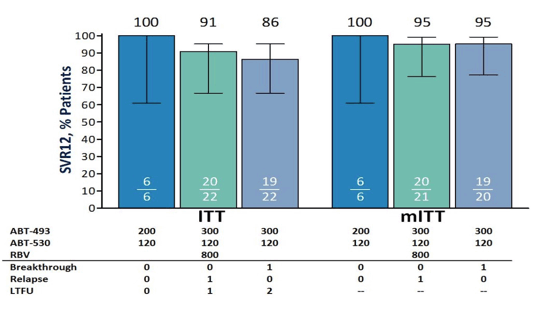
EASL: HIGH EFFICACY OF ABT-493 AND ABT-530 IN HCV GENOTYPE 1-INFECTED PATIENTS WHO HAVE FAILED DIRECT-ACTING ANTIVIRAL-CONTAINING REGIMENS: THE MAGELLAN-I STUDY - (04/15/16)
EASL: ABT-493 AND ABT-530 COMBINATION DEMONSTRATED MINIMAL POTENTIAL FOR CYP-MEDIATED DRUG-DRUG INTERACTIONS - (05/10/16)
In conclusion, the regimen of ABT-493 + ABT-530 with or without RBV for 12 weeks was well tolerated and achieved high SVR12 rates in non-cirrhotic HCV GT1-infected DAA-experienced patients, most of whom had baseline NS3 and/or NS5A RAVs. The addition of RBV did not appear to impact SVR. This study is very important as it offers new treatment strategies for those few patients who are currently failing on all oral DAA combination therapy.
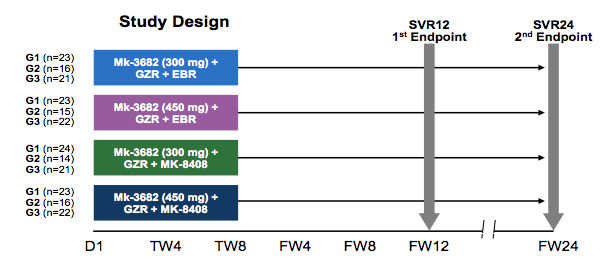
Other interesting new DAA combination studied was the new Merck regimen which consists of grazoprevir (the already licensed HCV PI) together with MK-8408 (an NS5A inhibitor) and MK-3682 an NS5B polymerase inhibitor. Other study arms were treated with grazoprevir/elbasvir and the new NS5B inhibitor MK-3682. These were randomized, open-label, dose-ranging Phase 2 studies in treatment-naïve, non-cirrhotic patients with HCV genotype (GT)1, 2 (C-CREST-1) or 3 (C-CREST-2) infection. Patients received EBR 50 mg/MK-3682 300 mg or 450 mg/GZR 100 mg or GZR 100 mg/MK-8408 60 mg/MK-3682 300 mg or 450 mg. All patients were treated for 8 weeks and followed for 24 weeks after completion of therapy. The respective study design is depicted below in figure 33.
Figure 33: Study design of the C-CREST 1 and 2 studies
240 patients were enrolled (GT1a, n = 46; GT1b, n = 47; GT2, n = 61; GT3, n = 86). The figure 34 summarizes the main SVR24 outcome data for the different genotypes, respectively.
Figure 34: SVR24 rates for the CREST studies
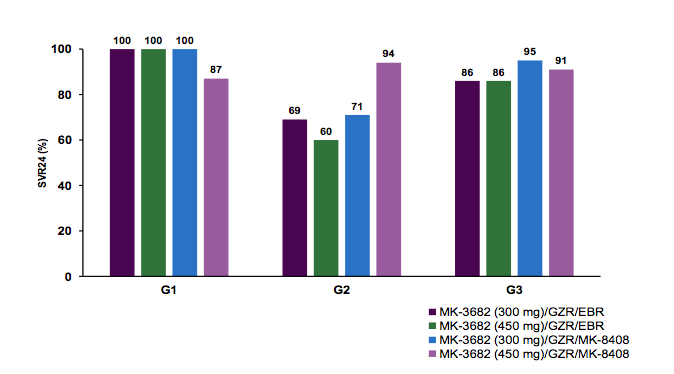
Across treatment arms, 98% (45/46) of GT1a and 98% (46/47) GT1b patients achieved SVR12. The GZR/MK-8408/MK-3682 (450 mg) regimen achieved SVR12 in 94% (15/16) of GT2 patients and 91% (20/22) of GT3 patients. In summary, whereas efficacy was great throughout all combinations and doses for GT1 patients improved SVR rates were observed for GT2 and 3 with the GZR/MK-8408/MK-3682 combination using the higher dose for MK-3682. Of note this was also achieved again with shorter treatment duration of 8 weeks. Current HCV DAA development clearly aims at shortening treatment duration at least for non-cirrhotics. Another important finding was that baseline RAVs in particular NS5A RAVs had no impact on SVR rates which is obviously different than what has been observed for grazoprevir/elbasvir alone in treatment of GT1a patients particularly in cirrhotics (see figure 35).
Figure 35: C-CREST 1 and 2: Grazoprevir/MK-8408/MK-3682: No Impact of Baseline RAVs on SVR.
EASL: High Efficacy of an 8-Week, 3-Drug Regimen of MK-3682/Grazoprevir/MK-8408 in HCV Genotype 1, 2, or 3-Infected Patients: SVR24 Data from the Phase 2 C-CREST 1 and 2 Studies - (04/19/16)
More data on 3-drug combinations was presented from Gilead. The newly developed fix-dose combination of sofosbuvir/velpatasvir was studied in combination with GS-9857 (new pangenotypic HCV protease inhibitor) in various studies. Among them were two Phase 2 studies, GS-US-367-1168 and GS-US-367-1169 which evaluated whether short duration SOF/VEL (400 mg/100 mg) + GS-9857 (100 mg) can effectively treat genotype (GT) 1-6 HCV-infected, treatment-naïve patients with or without cirrhosis (30). Patients were assigned SOF/VEL + GS-9857 administered orally once daily for 6 or 8weeks based on the absence or presence of cirrhosis, respectively. The primary endpoint was sustained virologic response 12 weeks after treatment (SVR12) as assessed by the CAP/CTM HCV 2.0 assay (LLOQ = 15 IU/mL). NS5B, NS5A, and NS3 regions were amplified and deep sequenced (<1% cutoff) at baseline and at the time of virologic failure. The study design is shown below in figure 36.
Figure 36: Study design
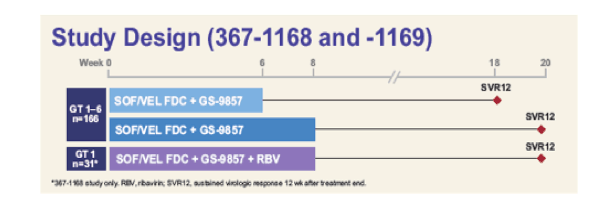
A total of 130 patients were studied (52% GT1, 9% GT2, 30% GT3, 8% GT4, and 1% GT6) : 58% male, 82% white, 68% with non-CC IL28B allele(s), and 48% had documented cirrhosis. The SVR12 rates for the different genotypes are shown below in figure 37 and 38.
Figure 37: SVR12 rates for GT1 patients depending on cirrhosis status
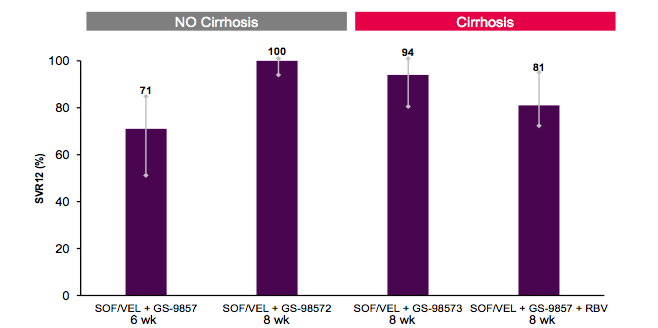
Of note, 6 weeks of 3 DAA combination therapy was associated with a lower SVR12 rate than 8 weeks suggesting that a minimum treatment duration indeed may be needed in HCV therapy despite using very potent drugs. This is very interesting in the context of the acute HCV studies where a 2 drug regimen of ledipasvir/sofosbuvir was sufficient in curing all HCV monoinfected patients with acute HCV (see above) which implies that innate immunity during acute HCV may play a role in achieving SVR in this unique phase of HCV infection. Once established, shorter treatment durations appear less promising. 8 weeks however in non-cirrhotic GT1 patients cured all thereby introducing a further new pangenotypic DAA combination which will cure all in shorter treatment duration regardless of baseline viral load or RAVs. In the cirrhotic patients addition of RBV did not seem to add anything to chance of obtaining SVR.
Figure 38: SVR12 rates for GT2-6 patients depending on cirrhosis status
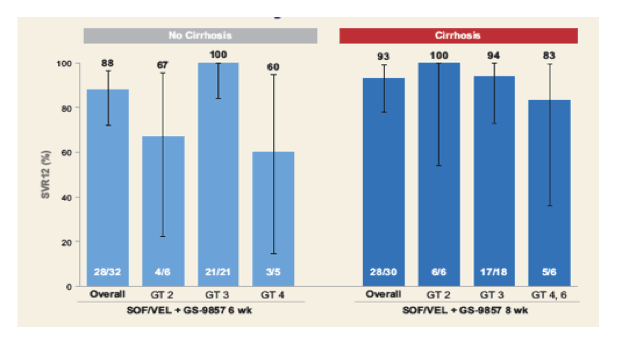
Obviously, data has to be interpreted cautiously as some genotypes are only included with very low numbers. Nevertheless, it appears that 8 weeks lead to higher SVR rates than 6 weeks for GT 2 and 4 whereas 6 weeks were sufficient to reach HCV cure in all GT3 patients. This is very exciting and promises yet another DAA combination with improved GT3 activity. Larger trials will be needed to confirm these findings.
Frequent adverse events (AE, >10%) were headache, fatigue, diarrhea, and nausea; most were mild or moderate in severity. Two (2%) patients had treatment-emergent SAEs of atrial flutter (n = 1) and vertigo (n = 1); and both were considered not related to study drug by the investigator. Two (2%) patients discontinued therapy due to AE(s) of asthenia, diarrhea, vomiting, and dehydration (n = 1) at Week 7; and fatigue (n = 1) at Week 5; both achieved SVR12. No clinically significant laboratory abnormalities were observed.
EASL: Short-Duration Treatment With Sofosbuvir/Velpatasvir Plus GS-9857 in Treatment-Naïve Genotype 1-6 HCV-Infected Patients With or Without Cirrhosis - (04/22/16)
Within the two Phase 2 studies, GS-US-367-1168 and GS-US-367-1169, was also a study arm which studied sofosbuvir/velpatasvir (SOF/VEL, 400 mg/100 mg) + GS-9857 (100 mg) administered once daily for 12 weeks in treatment-experienced genotype (GT) 1-6 infected patients with or without cirrhosis, including those who were previously treated with DAAs (31). Obviously, this is an even more challenging to treat patient group due to the prior treatment failure. Patients with virologic failure to prior treatment with pegylated-interferon (Peg-IFN) plus RBV (GT 2-6) or any DAA (GT 2-6), NS5A-inhibitor or multiple classes of DAAs (GT1) ± Peg-IFN ± RBV regimens received open-label SOF/VEL + GS-9857 for 12 weeks. NS5A, NS3, and NS5B regions were amplified and deep sequenced (<1% cutoff ) at baseline and at the time of virologic failure. Overall, a total of 128 patients (49% GT1, 16% GT2, 27% GT3, 5% GT4, and 2% GT6) were included: 75% male, 82% white, 73% with non CC IL28B alleles and 48% of patients had cirrhosis. Overall, 27% were NS5A-experienced, 52% were non-NS5A, DAA-experienced, and 21% had no prior DAA-experience. The study design is shown below in figure 39.
Figure 39: Study design
Baseline RAVs were detected in 60% of patients (20% NS5A; 15% NS3, 2% NS5B (no S282 T), and 23% had resistance to multiple classes). All patients except one achieved SVR12. One patient relapsed at post-treatment week 8. It was a 58-year-old white female with G3a infection, HCV RNA 7.0 log10 IU/mL, and cirrhosis. She also had a baseline NS5A RAV (Y93H) which may have contributed to development of virological failure. The SVR12 rates are shown below in figure 40.
Figure 40: Overall SVR12 rates and by genotype
Frequently reported adverse events (AE, >10%) were headache, fatigue, diarrhea, and nausea; most were mild or moderate in severity. One patient (<1%) experienced a serious AE of gastroenteritis (<1%); and 1 patient discontinued treatment early due to an AE of gastritis at Week 9 and achieved SVR12; both were considered not related to the study drugs by the investigator. No clinically significant laboratory abnormalities were observed. In summary the combination of sofosbuvir/velpatasvir with the new HCV PI GS-9857 was highly efficacious and safe in a rather challenging patient population with previous treatment failure, genotypes 1-6 and 48% cirrhosis.
EASL: High Efficacy of Sofosbuvir/Velpatasvir Plus GS-9857 for 12 Weeks in Treatment-Experienced Genotype 1-6 HCV-Infected Patients, Including Those Previously Treated With Direct-Acting Antivirals - (04/15/16)
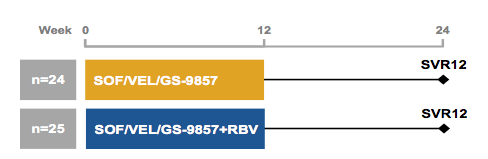
At EASL a further study was presented which evaluated the safety and efficacy of 12 weeks of SOF/VEL/GS-9857 ± ribavirin (RBV) in genotype 1 DAA-experience HCV-infected patients, including those with cirrhosis (32). Patients with prior DAA treatment experience were randomized, stratified by prior NS5A inhibitor experience and the presence of compensated cirrhosis, to receive SOF/VEL/GS-9857 or SOF/VEL/GS-9857 + RBV for 12 weeks. The study design is shown below in figure 41.
Figure 41: Study design
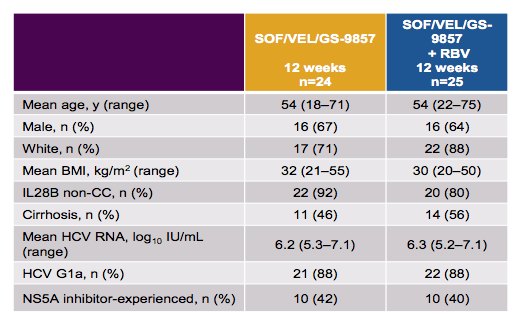
Forty-nine patients were randomized and treated. The majority was male (65%), Caucasian (80%), and had IL28B non-CC alleles (86%) and HCV genotype 1a (88%). Cirrhosis was present in 49% of patients. The median HCV RNA was 6.4 log10 IU/mL. Overall, 41% had previously received an NS5A inhibitor, 53% of patients had prior exposure to one DAA class, 27% had previously received DAAs from 2 classes, and 20% had previously received ≥3 classes of DAA. The baseline characteristics are summarized below in table 16.
Table 16: Baseline characteristics
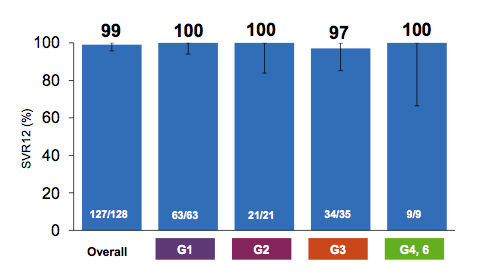
The SVR12 rates overall and for the two study arms are shown below in figure 42.
Figure 42: SVR results
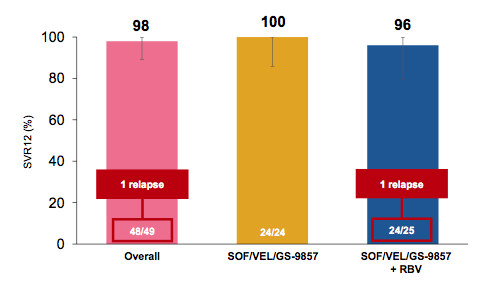
Overall HCV cure rates were excellent with 98% in this rather challenging to treat patient population. Only one relapse was noted in the SOF/VEL/GS-9857 ± RBV arm. Obviously addition of RBV did not help to prevent the relapse. The treatment was safe and well tolerated. There was one SAE (pneumonia). Two patients discontinued RBV due to adverse events (fatigue and rash). Adverse events were generally mild, and Grade 3/4 laboratory abnormalities were infrequent. The most frequent adverse events were fatigue (18%) and anemia (8%) which was only observed in patients that had received RBV. In conclusion, treatment with SOF/VEL/GS-9857 ± RBV for 12 weeks resulted in high rates of SVR4 in genotype 1 HCV-infected DAA experienced patients. The addition of RBV did not result in higher rates of SVR.
EASL: High Efficacy of Sofosbuvir/Velpatasvir/GS-9857 With or Without Ribavirin for 12 Weeks in Direct-Acting Antiviral-Experienced Patients With Genotype 1 HCV Infection - (04/15/16)
Treatment with generic DAAs
One of the most discussed presentations in the hepatitis arena at the EASL conference was without doubt the Australian report back from first treatment experience with DAA generics (33). In Australia the possibility exists to import medication from other countries if not available in the own country for personal use. As price negotiations were ongoing and months passed patients demanded treatment from their doctors as the news of HCV cure under DAA therapy spread. In this first presentation at EASL the efficacy and safety of generic DAA treatment was reported back for the first time (33). Sofosbuvir, ledipasvir, daclatasvir and ribavirin were imported and evaluated using High Precision Liquid Chromatograhy (HPLC) and Nuclear Magnetic Resonance (NMR). Rapid Virological Response (RVR) testing was conducted to confirm clinical efficacy. Patients were assessed pre-treatment, at weeks 4, 8, 12/End Of Treatment (EOT) and then for SVR 4 and SVR 12. Adverse events were recorded. This analysis includes results from 419 patients monitored in two clinics. The SVR rates under generic therapy are shown in figure 43.
Figure 43: Redemption-1 HCV RNA < LLOQ at EOT and SVR4
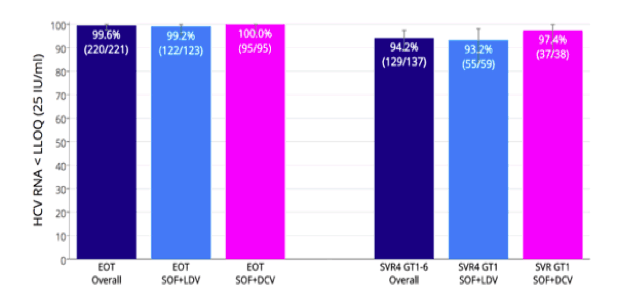
Of the 419 patients treated, 3 received SOF/RBV, 187 SOF/LDV, 20 SOF/LDV/RBV, 183 SOF/DCV and 26 SOF/DCV/RBV. Overall the patients were 56% male and 89% Caucasian with a mean age of 54 years; 30% had cirrhosis; 64% were Genotype 1, 5% Genotype 2, 28%
Genotype 3, and 3% Genotype 4, 5 or 6. The median baseline HCV RNA was 6.06 log10 IU/mL (1,160,000 IU/mL). Using currently available data, overall percentage HCV RNA undetectable was 99.6 at EOT, and 94.2% undetectable at SVR4. Clearly, these results are superimposable to what has been achieved in clinical trials. Obviously, the biggest concern with generics is that patients receive fake generics. By ensuring activity of the regimen through HLPC screening this risk was minimized. The overall SVR rates are impressive and clearly outline that in the setting of financial constraints generics will be the future of DAA based HCV therapy allowing much more patients to benefit from this beneficial treatment.
EASL: High sustained virological response rates using generic direct antiviral treatment for Hepatitis C REDEMPTION-1 - (04/27/16)
Summary
• 6 weeks of Ledipasvir/Sofosbuvir for treatment of acute HCV moninfection was very successful with all patients achieving SVR12. As all patients were symptomatic but treated immediately after diagnosis it can however, be speculated that a considerable proportion of patients would have cleared the infection spontaneously.
• A substantial HCV reinfection risk exists among HIV/HCV coinfected MSM - 7.6/100 person years with 25% reinfected at 3 years. These findings clearly underscore the failure of current prevention interventions and stress the need for developing better prevention strategies.
• Real-life Portuguese data demonstrates that hepatitis C treatment with universal access to ledipasvir/sofosbuvir and sofosbuvir is associated with very high SVR, >95% irrespective of HCV genotype involved. The most difficult patients to treat are those with G3 and cirrhosis.
• Surprisingly high SVR 12 rates were obtained in non-cirrhotic HCV GT3 after receiving 12 weeks of ledipasvir/sofosbuvir and ribavirin, but clearly if negative host factors are added such as cirrhosis, HCV cure rates dramatically drop off underling the somewhat low GT3 activity of this regimen.
• Overall, PPI use was not predictive of SVR in a large real-life cohort. Twice daily PPI however, had a reduced effect with 91.7% SVR in univariate analysis but not multivariate analysis. Caution is advised when using twice daily PPIs.
• The new all oral fix-dose combination of sofosbuvir/velpatasvir for treatment of GT1-6 infection in patients with HIV/HCV coinfection was very successful and well tolerated with an overall SVR12 rate of 95% which was not impacted by presence of cirrhosis or prior treatment failure.
• A 12-week regimen of EBR/GZR was highly efficacious among patients with inherited blood disorders and HCV GT1/4 infection. High efficacy was maintained across many important patient subgroups, including those with cirrhosis and HIV co-infection, and across all IBLDs. A lower response was seen among patients with GT1a infection with baseline NS5A RAVs.
• LDV/SOF for 12 weeks resulted in 100% SVR12 in HCV-infected kidney transplant recipients with or without cirrhosis and/or history of prior treatment failure. LDV/SOF was generally safe and well tolerated in kidney transplant recipients with HCV infection and no clinically meaningful reductions in renal function were observed.
• Within the TARGET cohort study no impact of baseline NS5A or NS3 RAVs was detected on SVR outcome.
• Overall, it appears that RAVs which can be found after virological failure under DAA combination therapy strongly reflect the selective pressure of the DAAs used for HCV therapy. Retreatment with fully active agents from different drug classes + mostly SOF is associated with very promising HCV cure rates despite extensive RAVs after virological failure under DAA combination therapy.
• SOF/VEL was associated with a very favourable safety profile in the phase III ASTRAL program with no difference in adverse events or laboratory abnormalities as seen in the placebo group.
• On-treatment HCV RNA at treatment week 4 is not predictive of outcomes after 12 weeks of SOF/VEL therapy.
• SOF/VEL exhibits a favourable DDI profile allowing use with various drugs commonly used by HCV-infected patients. The same recommendations for PPIs should be used for concomitant therapy with SOF/VEL as with LED/SOF.
• The combination of ABT-493 and ABT-530 administered for 8 weeks in non-cirrhotic patients with HCV GT1 or GT2 infection was well tolerated and achieved SVR rates of 97-98%, with no virologic failures, regardless of baseline viral load or prior treatment history.
• The combination of potent next generation antivirals, ABT-493 and ABT-530, co-administered for 8 weeks in treatment naïve, non-cirrhotic patients with HCV GT3 infection was well tolerated and achieved a high SVR rate of 97%. These encouraging data warrant confirmation in a larger cohort of GT3-infected patients.
• ABT-493 + ABT-530 with or without RBV for 12 weeks was well tolerated in treatment-naïve GT3-infected patients with cirrhosis, achieving SVR4 rates of 100% regardless of the presence of baseline NS3 and/or NS5A variants. Clearly, this combination holds a lot or promise in treatment of GT3 patients. Nevertheless data in treatment experienced cirrhotics is missing which in the end are the most difficult to treat patient group.
• The regimen of ABT-493 + ABT-530 with or without RBV for 12 weeks was well tolerated and achieved high SVR12 rates in non-cirrhotic HCV GT1-infected DAA-experienced patients, most of whom had baseline NS3 and/or NS5A RAVs. The addition of RBV did not appear to impact SVR.
• An 8-week regimen of GZR/MK-8408/MK-3682 (450 mg) was highly effective, with SVR12 >90% in treatment-naïve, non-cirrhotic patients with HCV GT1, 2 or 3 infection, including those with NS5A RAVs at baseline.
• Treatment with SOF/VEL + GS-9857 administered once daily for 8 weeks is safe, well tolerated, and highly effective in treatment-naïve, genotype 1-6 HCV-infected patients with cirrhosis. The 6 week treatment duration was associated with a higher relapse rate.
• Treatment with SOF/VEL + GS-9857 for 12 weeks in treatment-experienced GT1-6 HCV-infected patients resulted in a high SVR12 rate of 99%, including those who failed prior DAA(s)-containing regimens. SOF/VEL + GS-9857 was safe and well tolerated.
• Treatment with SOF/VEL/GS-9857 ± RBV for 12 weeks resulted in high rates of SVR4 in genotype 1 HCV-infected DAA experienced patients. The addition of RBV did not result in higher rates of SVR.
• Treatment with legally imported generic DAAs was demonstrated to lead to SVR4 rates of 93% on SOF/LDV and 97% on SOF/DCV. These SVR rates are similar to those seen in Phase 3 trials of the branded treatments. Mass treatment with generic DAAs is a feasible alternative where high prices prevent access to branded treatment.
References
1. Hinrichsen H, et al. Real-world safety and effectiveness of ombitasvir/paritaprevir/r with dasabuvir and/or ribavirin in the German Hepatitis C Registry. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract GS07.
2. Kattakuzhy S, et al. A novel task shifting model to expand the HCV care continuum: The ascend investigations. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LBP524.
3. Welzel T, et al. Effectiveness and safety of DAA combination therapies for treatment of HCV in elderly patients (>70 yrs): Results from the German Hepatitis C-Registry. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-274.
4. Afdhal N, et al. No effect of proton pump inhibitor (PPI) use on SVR with ledipasvir/sofosbuvir (LDV/SOF): Real world data from 2034 genotype 1 patients in the TRIO networks. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LBP519.
5. Deterding K, et al. Six weeks of sofosbuvir/ledipasvir (SOF/LDV) are sufficient to treat acute hepatitis C virus genotype 1 monoinfection: The HEPNET acute HCV IV study. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LB08.
6. Hezode C, et al. C-EDGE IBLD: Efficacy and safety of elbasvir/grazoprevir in patients with chronic hepatitis C virus infection and inherited blood disorders. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-128
7. Colombo M, et al. Ledipasvir/Sofosbuvir for 12 or 24 weeks is safe and effective in kidney transplant recipients with chronic genotype 1 or 4 HCV infection. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract GS13.
8. Wang G, et al. Prevalence and impact of baseline resistance-associated variants on ledipasvir/sofosbuvir or simeprevir/sofosbuvir against GT1 HCV infection: HCVTARGET interim analysis. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract PS102.
9. Vermehren J, et al. Retreatment of patients who failed DAA-combination therapies: Real-world experience from a large hepatitis C resistance database. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract PS103.
10. Poordad F, et al. High efficacy of ABT-493 and ABT-530 in HCV genotype 1 infected patients who have failed direct-acting antiviral-containing regimens: The MAGELLAN-I study. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract GS11.
11. Muir AJ, et al. High SVR rates with ABT-493 + ABT-530 co-administered for 8 weeks in non-cirrhotic patients with HCV genotype 3 infection. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract PS098.
12. Rockstroh JK et al.: Ledipasvir/Sofosbuvir for 6 weeks in HIV-infected patients with acute HCV infection. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract LB154
13. Martin T, et al. Hepatitis C virus reinfection incidence and outcomes among HIV-positive MSM in Western Europe. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abst. PS098.
14. Rodriguez J et al. Evidence of impressive real world SVR from the Portuguese ledipasvir/sofosbuvir and sofosbuvir universal coverage program to eradicate (eliminate) hepatitis C. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LBP523
15. Terrault N et al.: Treatment outcomes with 8, 12 and 24 week regimens of Ledipasvir/Sofosbuvir for the treatment of hepatitis C Infection: Analysis of a multicenter prospective, observational Study. 66th AASLD; San Francisco, CA; November 13-17, 2015; Abstract 94.
16. Neukam K et al.: HIV coinfection impairs the response to DAA-based HCV therapy. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LBP513
17. Wyles D, et al.: Sofosbuvir/Velpatasvir for 12 weeks in patients coinfected with HCV and HIV-1: The ASTRAL-5 Study. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract
18. Sarrazin C, et al.: Effect of baseline resistance-associated variants on SVR with the 3D regimen with and without RBV in GT1a and GT1b-infected patients. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LBP503
19. Jacobson I et al.: The tolerability of SOF/VEL for 12 weeks in >1,000 patients treated in the ASTRAL-1, ASTRAL-2, and ASTRAL-3 studies: An integrated safety analysis. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-168
20. Alqatani S et al.: On treatment HCV RNA as a predictor of SVR12 in patients with genotype 1-6 HCV infection treated with sofosbuvir/velpatasvir fixed dose combination for 12 weeks: An analysis of the ASTRAL-1, ASTRAL-2, and ASTRAL-3 studies. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-257
21. Mogalian E et al.: Drug-drug interaction profile of sofosbuvir/velpatasvir fixed dose combination. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract FRI-168
22. Poordad F, et al.: 98%-100% SVR4 in HCV Genotype 1 Non-Cirrhotic Treatment-Naïve or Pegylated Interferon/Ribavirin Null Responders With the Combination of the Next Generation NS3/4A Protease Inhibitor ABT-493 and NS5A Inhibitor ABT-530 (SURVEYOR-1). 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 41
23. Wyles DL, et al.: High SVR4 Rates Achieved With the Next Generation NS3/4A Protease Inhibitor ABT-493 and NS5A Inhibitor ABT-530 in Non-Cirrhotic Treatment-Naïve and Treatment-Experienced Patients With HCV Genotype 2 Infection (SURVEYOR-2). 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 250
24. Poordad F et al.: High SVR rates with the combination of ABT-493 + ABT-530 for 8 weeks in non-cirrhotic patients with HCV genotype 1 or 2 infection. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-157
25. Gane E et al.: High efficacy and favorable safety of ABT-493 and ABT-530 co-administration for 12 weeks in HCV genotype 1-infected patients with cirrhosis (SURVEYOR-I). 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-135
26. Kwo P, et al.: High SVR4 Rates Achieved With the Next Generation NS3/4A Protease Inhibitor ABT-493 and NS5A Inhibitor ABT-530 in Non-Cirrhotic Treatment-Naïve and Treatment-Experienced Patients With HCV Genotype 3 Infection (SURVEYOR-2). 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 248
27. Kwo P et al.: 100% SVR4 with ABT-493 and ABT-530 with OR without ribavirin in treatment-naïve HCV genotype 3-infected patients with cirrhosis. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LB01
28. Gane E et al.: 100% SVR4 and favourable safety of ABT-493 + ABT-530 administered for 12 weeks in non-cirrhotic patients with genotypes 4,5 or 6 infection (SURVEYOR-I). 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-137
29. Gane E et al.: High efficacy of an 8-week drug regimen of grazoprevir/MK-8408/MK-3682 in HCV genotype 1,2 and 3-infected patients: SVR24 data from the phase 2 C-CREST 1 and 2 studies. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-139
30. Gane E et al.: Short duration treatment with sofosbuvir/velpatasvir plus GS-9857 in treatment-naïve genotype 1-6 HCv infected patients with or without cirrhosis. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract SAT-138
31. Lawitz E, et al. High efficacy of sofosbuvir/velpatasvir plus GS-9857 for 12 weeks in treatment-experienced genotype 1-6 HCV infected patients including those previously treated with direct-acting antivirals. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract PS008
32. Lawitz E, et al.: High efficacy of sofosbuvir/velpatasvir/GS-9857 with or without ribavirin for 12 weeks in direct acting antiviral-experienced patients with genotype 1 HCV infection. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract PS021
33. Freeman J et al.: High sustained virological response rates using generic direct antiviral treatment for hepatitis C imported into Australia. 51st EASL; Barcelona, Spain; April 13-17, 2016. Abstract LB03
|
| |
|
 |
 |
|
|