 |
 |
 |
| |
High Efficacy of an 8-Week, 3-Drug Regimen of MK-3682/Grazoprevir/MK-8408 in HCV Genotype 1, 2, or 3-Infected Patients: SVR24 Data from
the Phase 2 C-CREST 1 and 2 Studies
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Gane EJ1; Pianko S2; Roberts SK3; Thompson AJ4; Zeuzem S5; Zuckerman E6; Ben-Ari Z7; Foster GR8; Agarwal K9; Laursen AL10; Gerstoft J11; Gao W12; Huang H-C12; Fitzgerald B12; Fernsler D12; Li JJ12; Wan S12; Zeng Z12; Chen H-L12; Dutko F12; Nguyen B-Y12; Wahl J12; Barr E12; Butterton JR12; Esteban R13
1Auckland Clinical Studies, Auckland, New Zealand; 2Monash Health and Monash University, Melbourne, VIC, Australia; 3Alfred Hospital, Melbourne, VIC, Australia; 4St. Vincent's Hospital Melbourne, Melbourne, VIC, Australia; 5JW Goethe University Hospital, Frankfurt, Germany; 6Carmel Medical Center, Haifa, Israel; 7Sheba Medical Center, Ramat Gan, Israel; 8Queen Mary University of London, London, United Kingdom; 9King's College Hospital, London, United Kingdom; 10Aarhus University Hospital, Aarhus, Denmark; 11University of Copenhagen, Copenhagen, Denmark; 12Merck & Co., Inc., Kenilworth, NJ, USA; 13Hospital Universitario Val d'Hebron, Barcelona, Spain
EASL: Merck HCV Nuk MK3682 7-Day Monotherapy - (05/05/15)
AASLD/2014: MK-8408, a Potent and Selective NS5A Inhibitor With a High Genetic Barrier to Resistance and Activity Against HCV Genotypes 1-6 - (11/12/14)




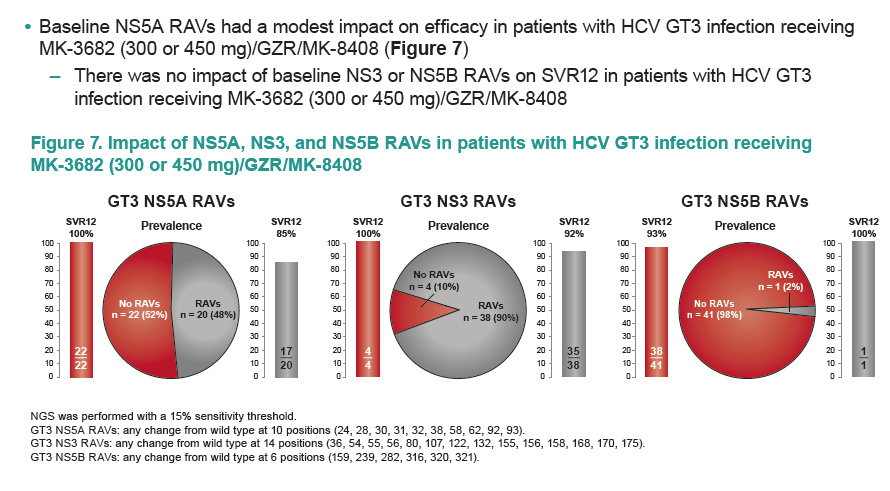
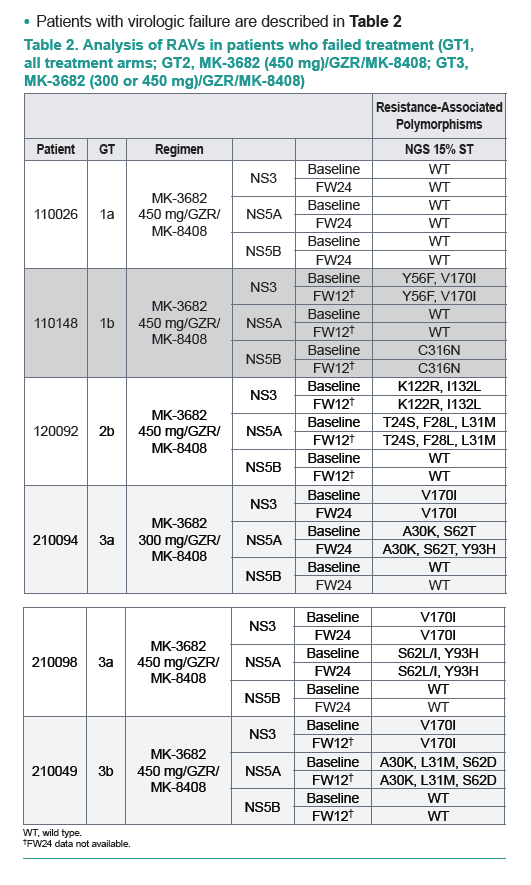
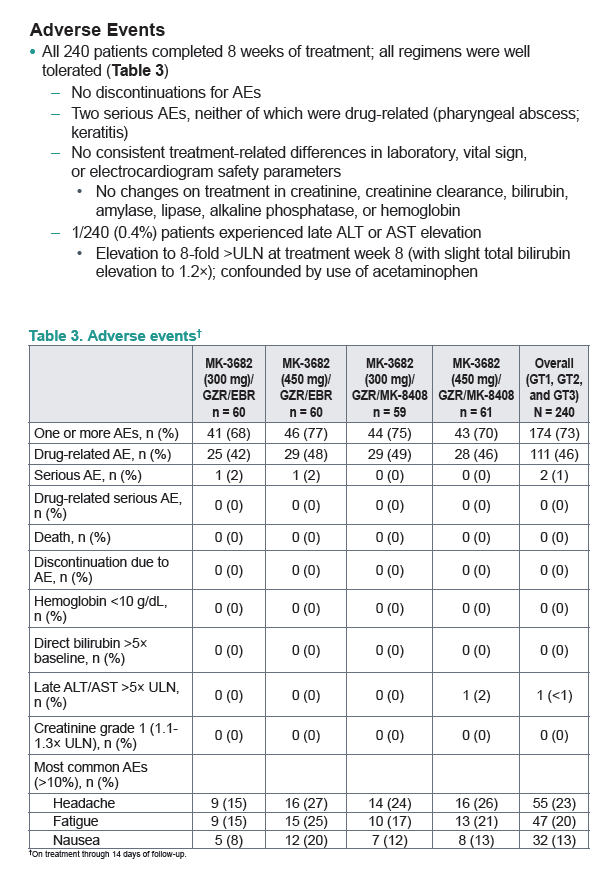

|
| |
|
 |
 |
|
|