 |
 |
 |
| |
TREATMENT OF HEPATITIS C VIRUS IN PATIENTS WITH ADVANCED CIRRHOSIS:
ALWAYS JUSTIFIED? ANALYSIS OF THE HEPA-C REGISTRY
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Carlos Fernandez-Carrillo* 1, Sabela Lens2, Elba Llop1, Juan Manuel Pascasio3, Inmaculada Fernandez4, Carme Baliellas5, Javier Crespo6, María Buti7, Lluís Castells7, Manuel Romero-Gomez8, Clara Pons9, Jose María Moreno10, Agustín Albillos11, Conrado Fernandez-Rodríguez12, Martín Prieto13, Miguel Fernandez-Bermejo14, Javier García-Samaniego15, Jose Antonio Carrion16, Manuel de la Mata17, Ester Badia18, Javier Salmeron19, Jose Ignacio Herreros20, Magdalena Salcedo21, J. Javier Moreno22, Juan Turnes23, Rafael Granados24, Michel Ble25, Jose Luis Calleja1 and The Hepa-C Registry
1Digestive, Liver, Hospital Universitario Puerta de Hierro-Majadahonda, IDIPHIM, Majadahonda, Madrid, 2Liver Unit, Hospital Clínic, IDIBAPS, CIBERehd, Barcelona, 3Digestive diseases, Hospital Universitario Virgen del Rocío, IBIS, CIBERehd, Sevilla, 4Digestive, Hospital Universitario 12 de Octubre, Madrid, 5Digestive, Hospital Universitari de Bellvitge, L'Hospitalet de Llobregat, Barcelona, 6Digestive, Hospital Universitario Marques de Valdecilla, Santander, 7Internal Medicine-Hepatology, Hospital Universitario Vall d'Hebron, Barcelona, 8Digestive diseases, Hospital Universitario Virgen de Valme, CIBERehd, Sevilla, 9Digestive, Hospital General Universitario de Castellon, Castellon de la Plana, 10Digestive, C.H.U de Albacete, Albacete, 11Digestive, Hospital Universitario Ramon y Cajal, IRYCIS, CIBERehd, Madrid, 12Digestive, Hospital Universitario Fundacion Alcorcon, Alcorcon, Madrid, 13Digestive Medicine, Hospital Universitario de La Fe, CIBERehd, Valencia, 14Digestive, Hospital San Pedro de Alcantara, Caceres, 15Infectious Diseases, Hospital Carlos III, CIBERehd, Madrid, 16Digestology, Hospital del Mar, IMIM, Barcelona, 17Digestive, Hospital Universitario Reina Sofía, IMIBIC, CIBERehd, Cordoba, 18Digestive, Hepatology, Hospital Universitario de Burgos, Burgos, 19Digestive, Hospital Universitario San Cecilio, CIBERehd, Granada, 20Liver Unit, Clínica Universitaria de Navarra, IdiSNA, CIBERehd, Pamplona, 21Liver Unit, Hospital General Universitario Gregorio Maranon. IiSGM, Madrid, 22Internal Medicine, Hospital General de Segovia, Segovia, 23Digestive, Complejo Hospitalario de Pontevedra, Pontevedra, 24Internal Medicine, H. U. de Gran Canaria Dr. Negrín, Las Palmas de Gran Canaria, 25Digestive, Servidigest Centre, Barcelona, Spain

Program Abstract
Background and Aims: Direct-acting antivirals (DAA) have recently revolutionized HCV treatment, which is being extended to special populations. The past year has seen considerable debate on the risk/benefit of treating patients with advanced or decompensated liver disease. We aimed at finding factors to identify the candidates most likely to experience an improved natural course of their chronic liver disease without compromising safety.
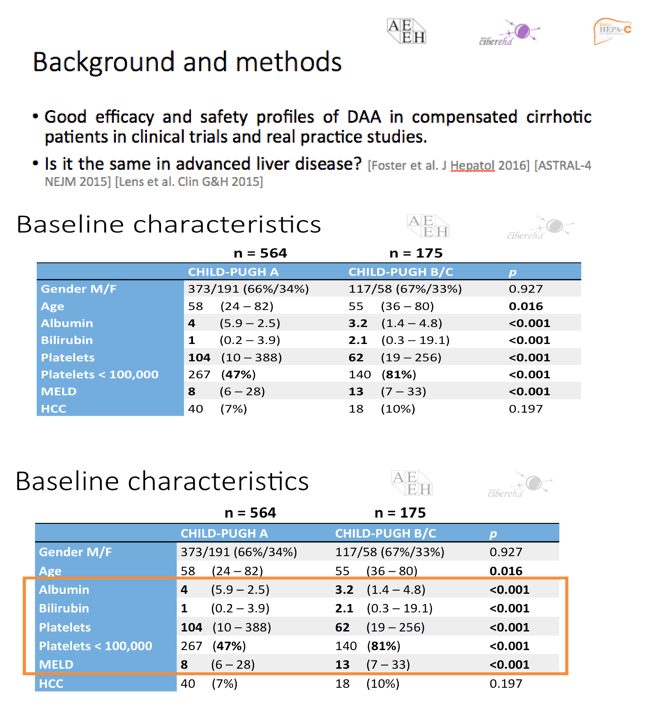
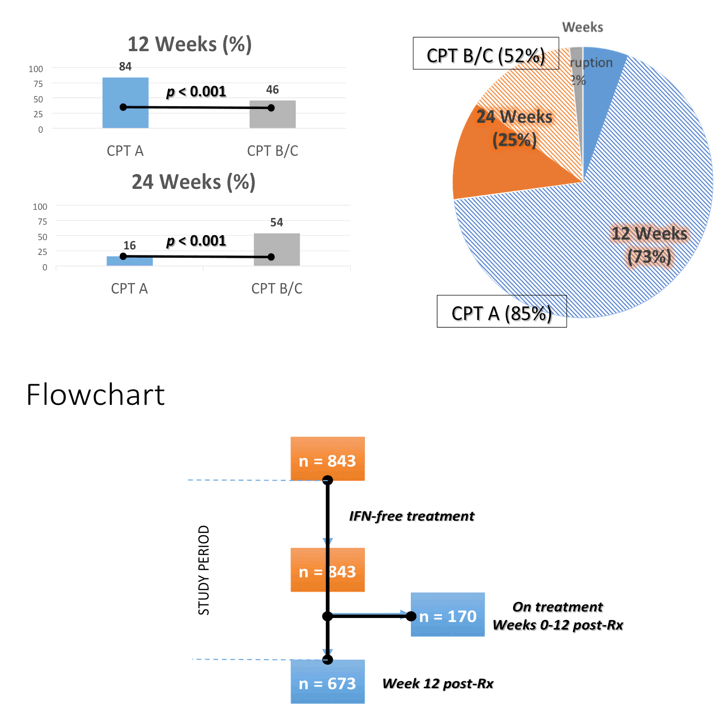
Methods: Hepa-C is a collaborative, monitored national registry of HCV patients directed by the Spanish Association for the Study of the Liver. A total of 564 patients, many of them included in the SOF compassionate use program, with cirrhosis on biopsy, FibroScan, and/or clinical symptoms, not transplanted during or within 12 weeks after treatment (study period), were registered.
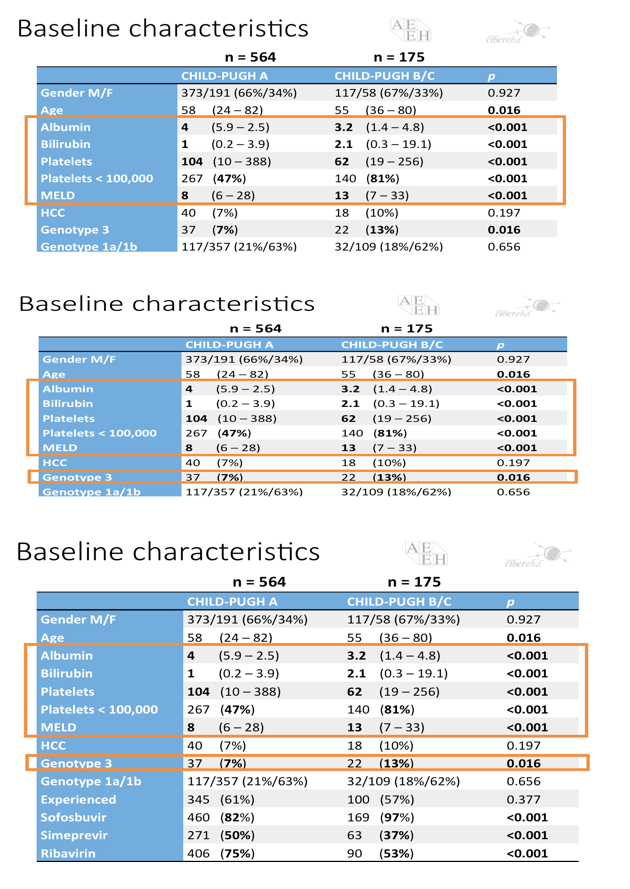
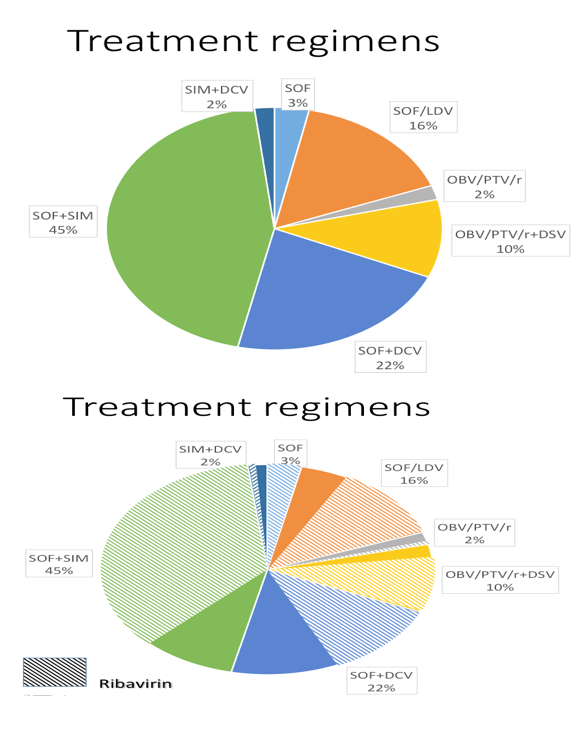
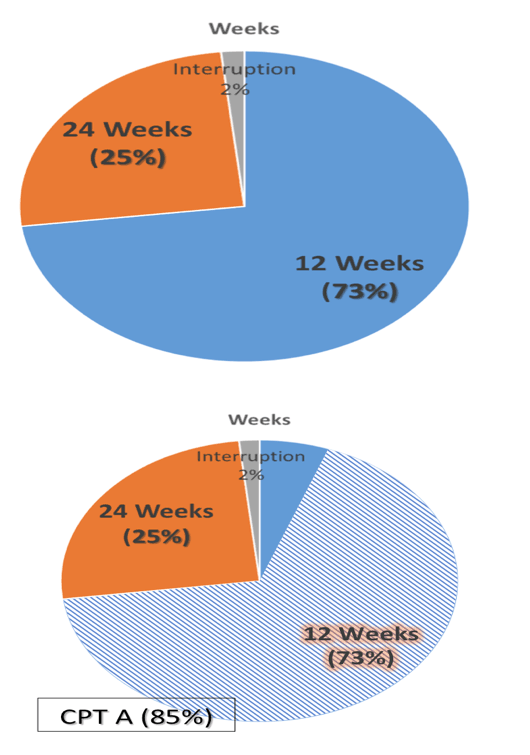
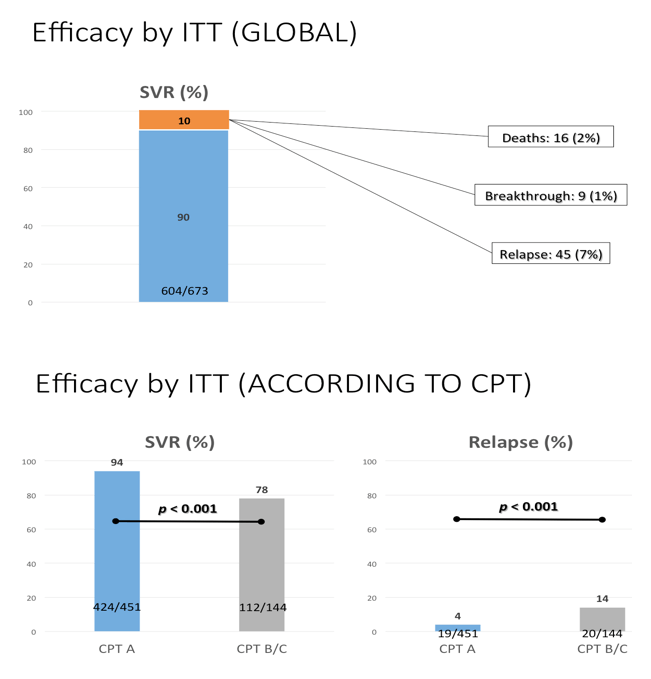
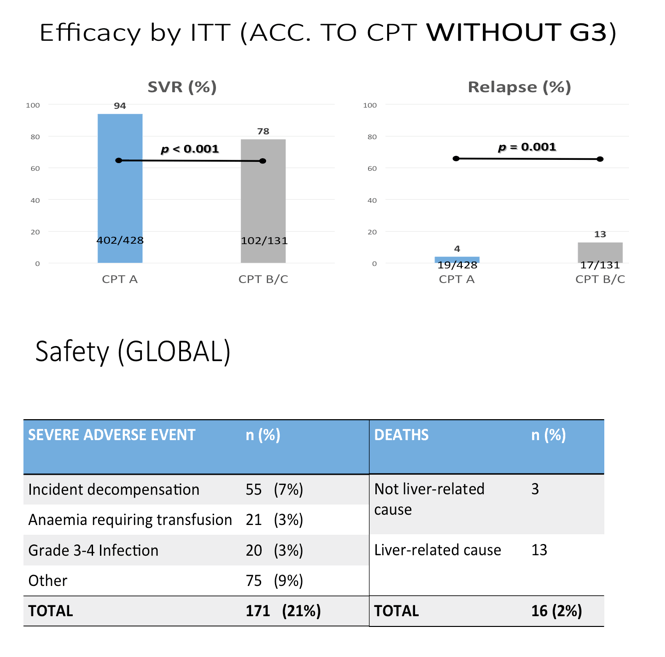
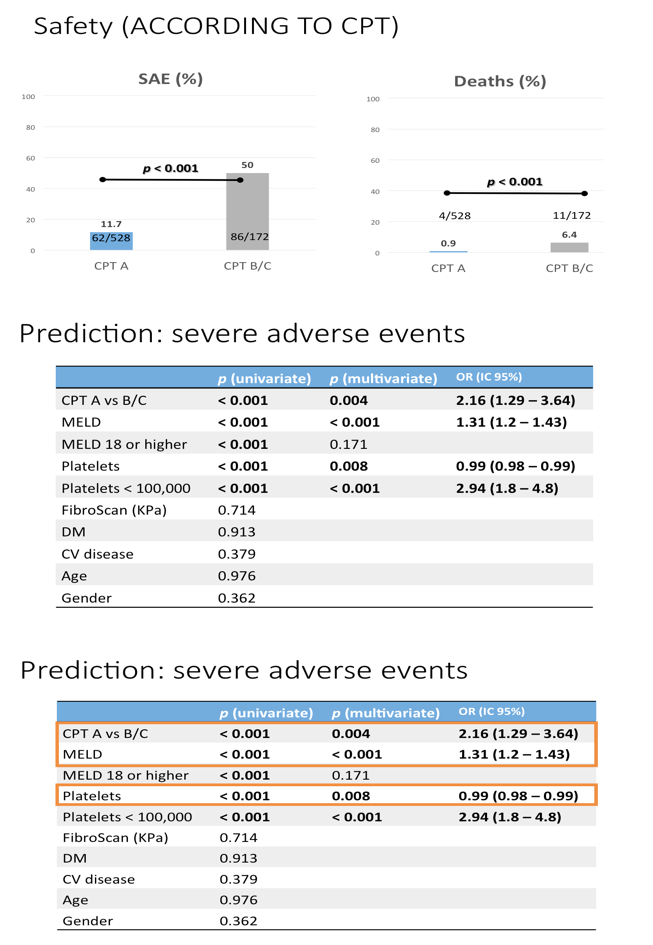
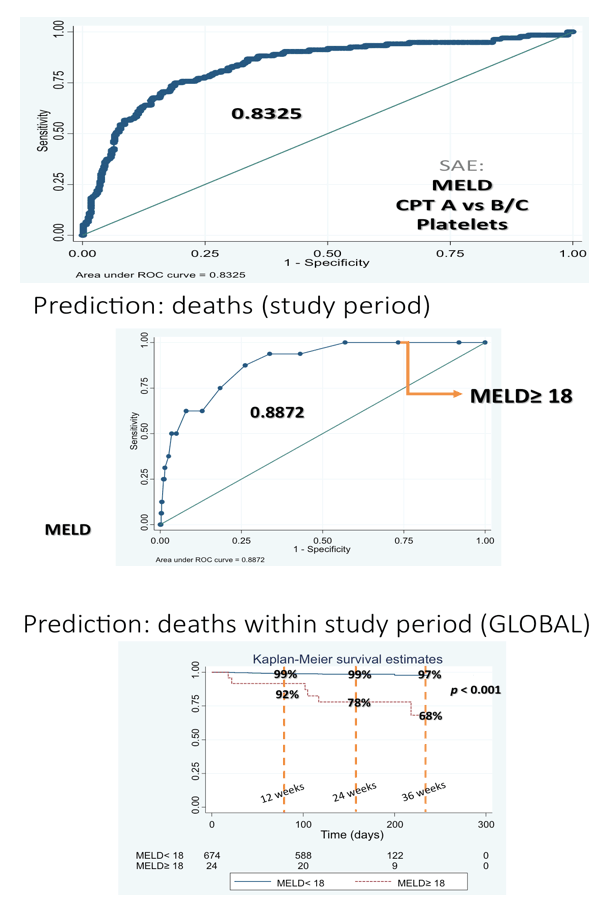
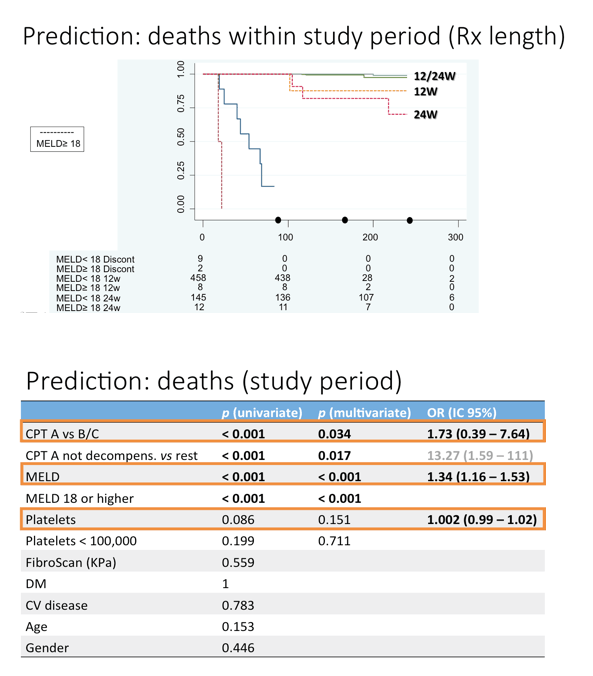
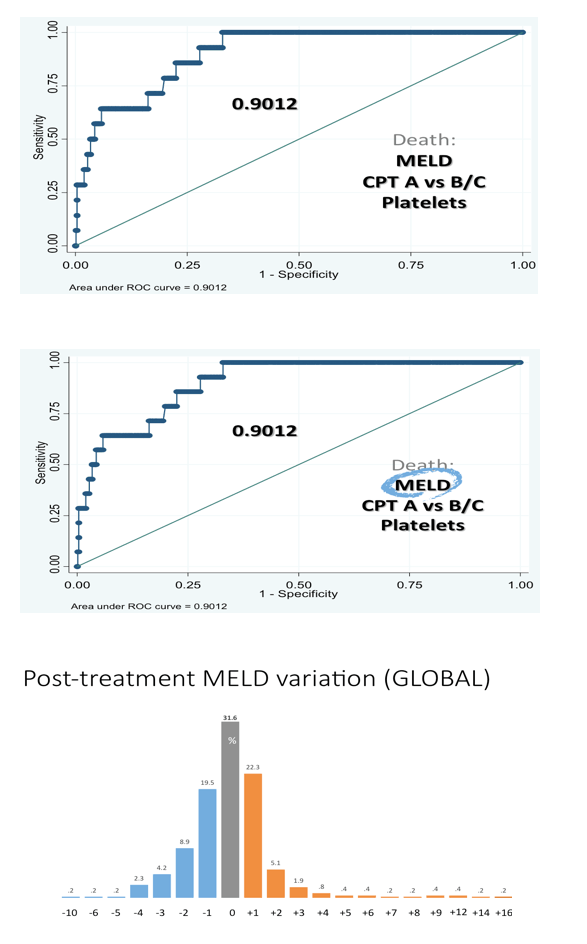
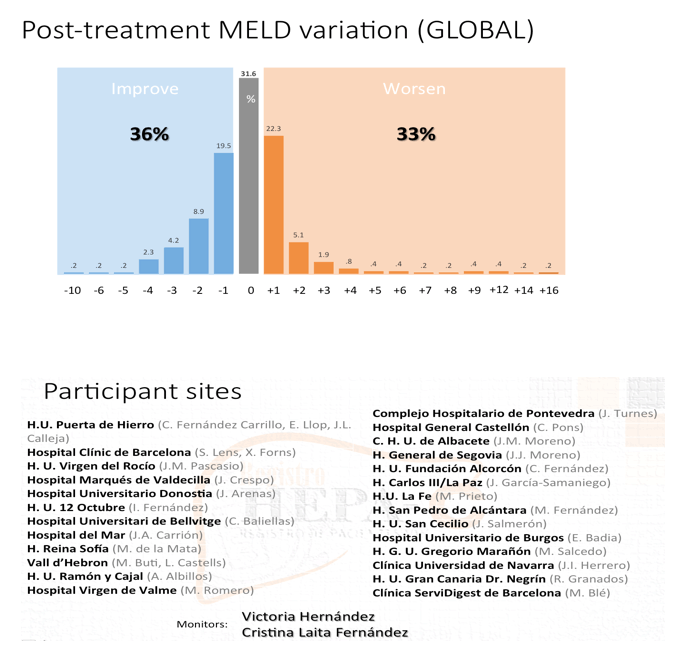
Results: The study included 393/564 (70%) patients with compensated cirrhosis (Child-Pugh A) and 171/564 (30%) patients with Child-Pugh (CPT) B/C. The treatment regimens were: SMV + SOF: 292/564 (52%), SOF + DCV: 133/564 (24%), SOF/LDV: 70/564 (12%), OBV/PTV/r+/-DSV: 28/564 (5%), SMV + DCV: 12/564 (2%), all with or without ribavirin (R), and SOF+ R: 28/564 (5%). Overall percentages of SVR, relapse and grade 3-4 adverse events (AE) were 88% by intention to treat (ITT), 10%, and 27%, respectively. Patients with baseline CPT B/C had lower SVRs, and more relapses and grade 3-4 AE than CPT A (81% vs. 95% ITT, p <0.001; 21% vs. 5%, p <0.001; 61% vs. 12%, p <0.001). More deaths occurred during the study period in patients with advanced baseline CPT (B/C vs. A: 12 vs. 6, p = 0.003). Baseline MELD >17 independently identified a group of patients with 39% deaths vs. 1.5% (p <0.001) in this period. However, patients with CPT B/C patients had greater post-treatment improvement in liver function (MELD) compared with CPT A ones, even after excluding regimens with SMV from the analysis. Baseline FibroScan and MELD values were independently associated with this improvement, yet their influence was very small (OR:1.05, CI:1.01-1.1, p = 0.01; OR:0.72, CI:0.56-0.92, p <0.01).
Conclusions: Compared with patients with less severe disease, patients with advanced liver disease are more likely to die during or within 12 weeks after HCV treatment and had more AE and worse virologic response. Treating HCV in patients with baseline MELD >17 might imply an ethical issue due to a high risk of death in this period. The sample is being enlarged to confirm these findings.
|
| |
|
 |
 |
|
|