 |
 |
 |
| |
PREDICTING HBSAG CLEARANCE RESPONSES DURING ARC-520 RNA
INTERFERENCE (RNAI) THERAPY BASED ON HBSAG EPITOPE PROFILE ANALYSIS
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-17 Barcelona
Renae Walsh1, Rachel Hammond1, Lilly Yuen1, Joshua Deerain1, Bruce Given2, Thomas Schluep2, Man-Fung Yuen3, Henry Lik-Yuen Chan4, Ching-Lung Lai3, James Hamilton2, Johnson Yn Lau5, Carlo Ferrari6, Robert G Gish7, Stephen A. Locarnini* 1
1Victorian Infectious Diseases Reference Laboratory, Melbourne, Australia, 2Arrowhead Research Corporation, Pasadena, California, United States, 3The University of Hong Kong, 4The Chinese University of Hong Kong, 5Hong Kong Polytechnic University, Hong Kong SAR, China, 6University of Parma, Parma, Italy, 7Hepatitis B Foundation, Doylestown , United States
Curing HBV, New Drugs.....http://www.natap.org/2014/HBV/012215_01.htm
Curing HBV....attacking cccDNA "Suppression of hepatitis B virus DNA accumulation in chronically infected cells using a bacterial CRISPR/Cas RNA-guided DNA endonuclease"......http://www.natap.org/2014/AASLD/AASLD_151.htm............."..... in order to effectively clear an HBV infection and produce full remission, the viral cccDNA intermediates need to be destroyed

Program abstract
Background and Aims: Functional cure in chronic hepatitis B requires HBV DNA negativity, HBsAg loss and anti-HBs seroconversion. ARC-520 RNAi drug therapy targets cccDNA derived mRNA, including the full-length HBsAg transcript. Using a 19plex anti-HBs panel to map HBsAg epitopes, we have developed a predictive algorithm of an HBsAg clearance profile in patients undergoing HBsAg loss during tenofovir therapy, defined as reduced recognition at loop 1 and loop 2 HBsAg "a" determinant epitopes. Complimentary to this, we have developed assays to detect co-existing complexed anti-HBs (with HBsAg), and analysed the ARC-520 cohorts with the aim of evaluating the impact of RNAi therapy on HBsAg loss, the identification of an HBsAg clearance profile and the development of co-existing anti-HBs.
Methods: Analysis of HBsAg clearance profiles and concomitant anti-HBs was performed for 40 ARC-520 study HBeAg-negative (n=32) and HBeAg-positive (n=8) patients (under code: 30 ARC-520; 10 placebo), from pre-treatment to day 85, and then compared to the quantitative HBsAg responses. All were entecavir suppressed prior to (mean 5 years) and during ARC-520 therapy.
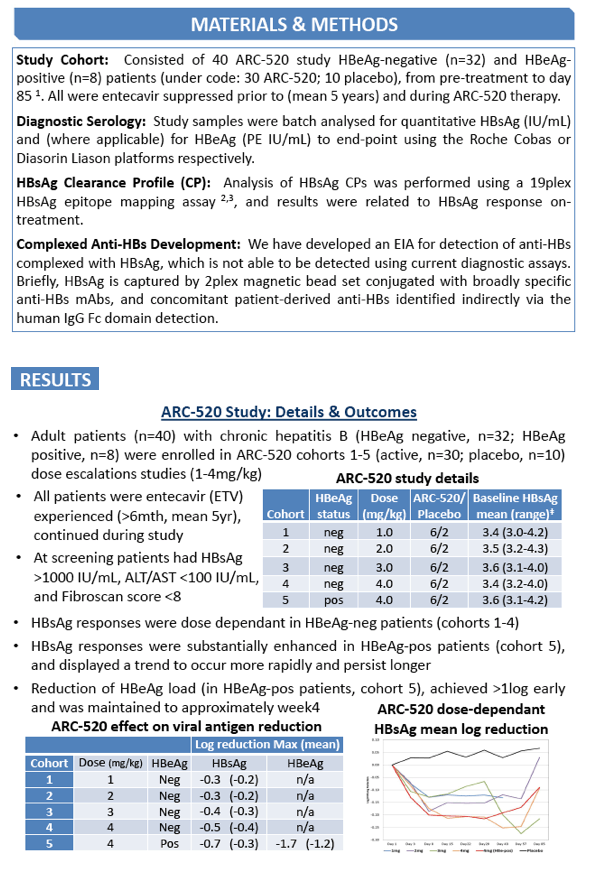
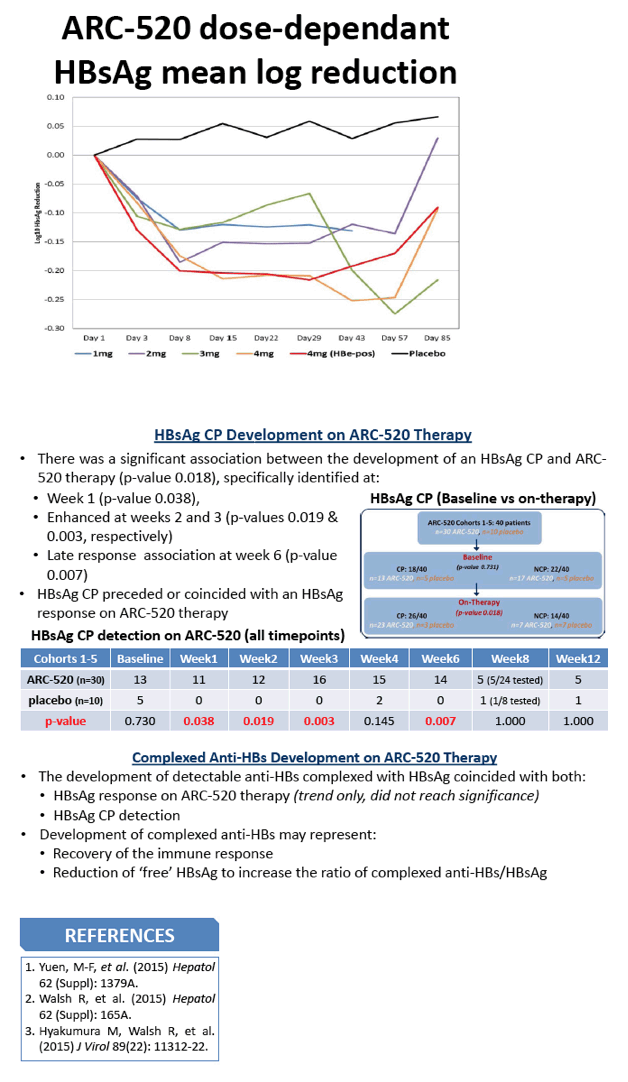
Results: ARC-520 therapy resulted in a dose response maximum decline in HBsAg of 0.3 log IU/ml observed at 1 mg/kg vs 0.5 log at 4 mg/kg in the HBeAg-negative patients (n=24), and 0.7 log at 4mg/kg in HBeAg-positive (n=6) patients. Analysis of the treated group identified that an HBsAg clearance profile preceded and/or coincided with HBsAg decline. A significant association between HBsAg clearance profile development and ARC-520 treatment emerged at week1 (11/30, p=0.038), and strengthened at week2 (12/30, p-value 0.019) and week3 (16/30, p= 0.003). A late HBsAg response at week6 was associated with development of an HBsAg clearance profile (14/30, p= 0.007). Clearance profiles were not observed in the placebo group. Complexed anti-HBs development coincided with HBsAg decline and HBsAg clearance profile detection.
Conclusions: Development of the HBsAg clearance profile was predictive of HBsAg decline due to ARC-520 therapy, with an increasing significant association from week1-3 coinciding with or preceding the HBsAg decline, and the detection of complexed anti-HBs, reflective of recovery of the anti-HBs response. Further longitudinal and multiple dose studies will assess the magnitude and persistence of HBsAg loss on ARC-520 therapy, in the context of the predictive potential of HBsAg clearance profile and anti-HBs response.
|
| |
|
 |
 |
|
|