 |
 |
 |
| |
PRECLINICAL CHARACTERIZATION OF POTENT CORE PROTEIN ASSEMBLY
MODULATORS FOR THE TREATMENT OF CHRONIC HEPATITIS B
|
| |
| |
Reported by Jules Levin
EASL 2016 April 14-27 Barcelona
Qi Huang1, Alexandre Mercier* 1, Yi Zhou1, Kirk Henne1, G. Renuka Kumar1, Shawn Sun1, Lida Guo1, Jean Liu1, Yuhua Zong1, Tian Sun1, Katherine Nabel1, Pao-Chen Li1, Cathal Mahon1, Geoffrey Chen1, Mark Bures2, Lichun Li2, Earl May2, Emily Connelly1, Jason Deer2, Sarah Katen2, Samson Francis2, William W. Turner2, Adam Zlotnick2, 3, Lee D. Arnold2, Uri Lopatin1
1Assembly Biosciences, Inc., San Francisco, 2Assembly Biosciences, Inc., 3Indiana University, Bloomington, United States

Program abstract
Background and Aims: There is currently no curative therapy for chronic HBV (CHB). Core Protein Allosteric Modulators (CpAMs) are a novel class of direct acting antivirals (DAA) for the treatment of CHB. Herein we characterize a series of potent, pan-genotypic CpAMs that can be used alone or in combination with current standard of care therapies to achieve higher rates of cure.
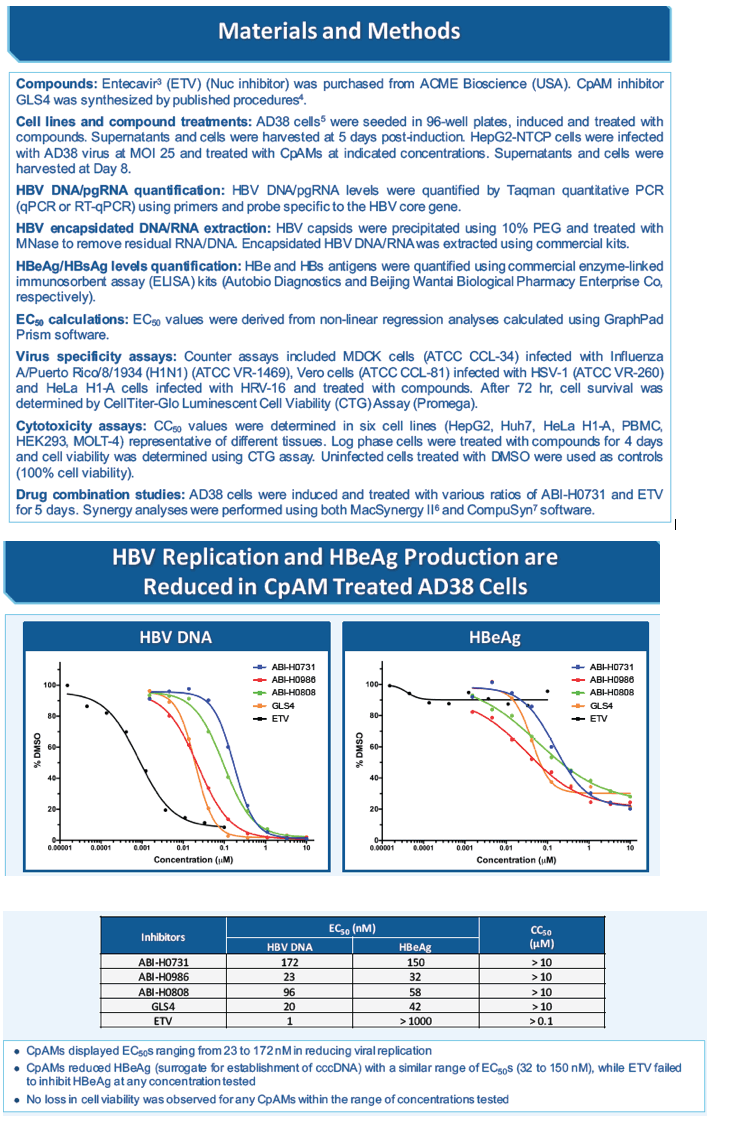
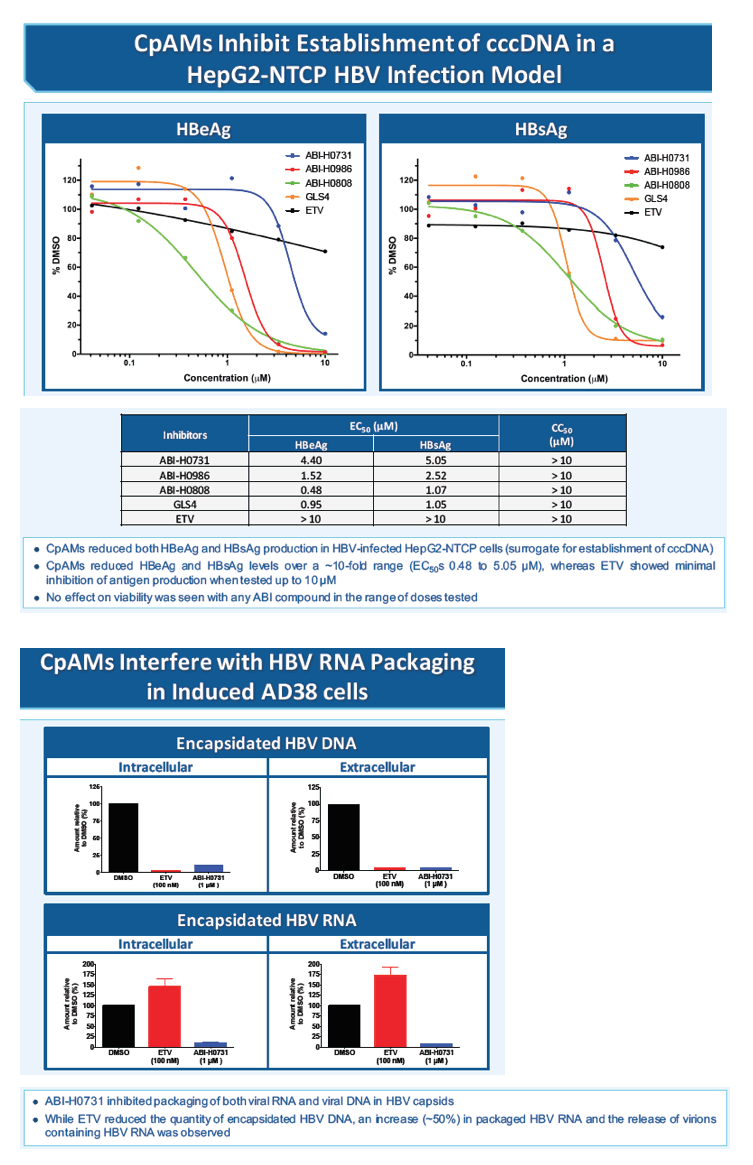
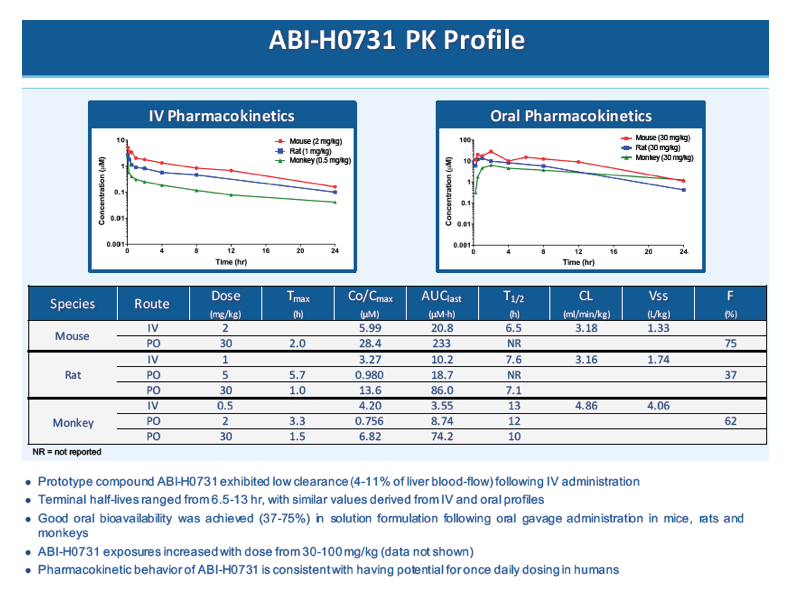
Methods: Antiviral activities of representative CpAMs were determined by measuring viral DNA replication and HBeAg production in the HepAD38 cell line (Gt D) and HepG2 cells transiently transfected with terminally redundant (1.05x), replication competent HBV clinical genomes (Gt A,B,C,D). Secreted (HepAD38) and intracellular (transiently transfected HepG2 cells) HBV viral DNA was measured by qPCR. Secreted HBeAg levels were determined with commercial HBeAg ELISA kits. Pharmacokinetic parameters were determined by non-compartmental analysis of plasma concentration-time data following intravenous and oral administration to mice, rats, dogs or monkeys.
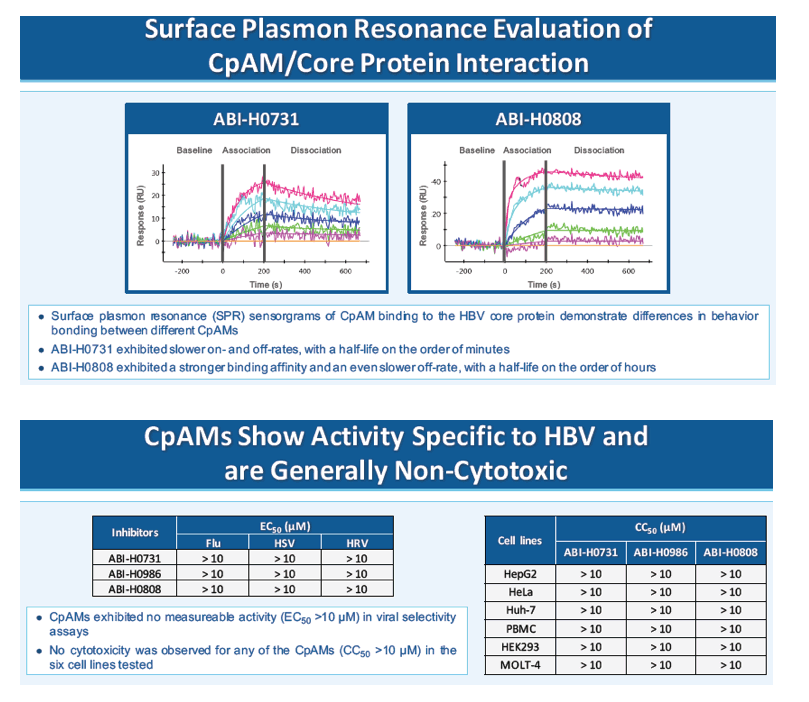
Results: Several representative CpAMs from a new series of inhibitors exhibited potent inhibition of both viral DNA replication and HBeAg production in HepAD38 cells (EC50 ranging from 20 to 200 nM). They also showed antiviral activities against other major HBV genotypes in transiently transfected HepG2 cells and demonstrated no activity against other viruses tested or cytotoxicity in a panel of cell lines, indicating that the compounds are selective inhibitors of HBV. Ongoing safety profiling of these molecules has not identified any significant safety liabilities. Several CpAMs from this new series exhibited limited or no inhibition of major CYP isoforms, suggesting low potential for drug-drug interactions with other co-administered agents. Pharmacokinetic analyses of candidate compounds showed oral bioavailability ranging from 20-75% in all species tested. Overall, pharmacokinetic properties are suitable for QD administration in clinical studies and robust exposures can be achieved following oral administration.
Conclusions: Representative molecules from a novel series of pan-genotypic CpAMs demonstrated potent HBV antiviral activity, good in vitro safety, and promising pharmacokinetic properties. The potency and favorable preclinical activity and safety profiles support selection of a CpAM from this series to advance into Phase 1 clinical studies.
|
| |
|
 |
 |
|
|