| |
DAAs /Restrictions-Denials - New Article:
Cost Pushback Limiting Hepatitis C Drug Utilization
|
| |
| |
from Jules: I have been saying for a long time as the author here says that if this were cancer or heart disease or diabetes, all mainstream diseases, and there were a cure there is no way The White House & Congress would allow any restrictions, they would make sure everyone got the drugs, the public would not let them do otherwise. YES its stigma because of who HCV affects, not proper citizens, drug users!, not the normal regular citizenry.
By Joseph Haas
https://www.pharmamedtechbi.com/publications/the-pink-sheet/78/25/cost-pushback-limiting-hepatitis-c-drug-utilization
June 20 2016
Executive Summary
Study finds 22% of patients prescribed drug therapy for HCV were not started on care or delayed 150-plus days beyond the writing of the prescription. Coverage decisions don't seem to be linked clearly to disease severity, but denials were higher among Medicaid patients and among certain private-sector plans.
With the pushback against pricing of the hepatitis C drugs that have entered the US market in the past few years, disease severity often has been cited as a reason why some HCV patients are denied coverage, but data analysis by Trio Health indicates that controlling costs remains a primary factor in determining which patients are treated.
Hepatitis C: Access To Care Methodology
A disease management consulting firm headquartered in Boulder, Colo., Trio Health performed an assessment of access to HCV care by reviewing the cases of 2,111 patients for whom an initial prescription for a direct-acting antiviral (DAA) regimen was written between October 2014 and March 2015.
The data were obtained from 197 physicians and eight specialty pharmas in 25 states. Patients in the sample were prescribed a 12-week regimen of either: Gilead Sciences Inc.'s Harvoni (sofosbuvir/ledipasvir) with or without ribavirin; Johnson & Johnson's Olysio(simeprevir) with sofosbuvir plus or minus ribavirin; or AbbVie Inc.'s Viekira Pak (ombitasvir/paritaprevir/ritonavir; dasabuvir) plus or minus ribavirin. Nine academic researchers compiled the data; the study was funded by Gilead.
New HCV drugs typically have posted substantial sales almost immediately after reaching market, leading to jockeying between companies over pricing and formulary position in the US private-sector health care market. The consistent market leader has been Gilead Sciences Inc., thanks to regimens featuring Sovaldi (sofosbuvir), but competitors have had their successes and lately eaten into Gilead's market share a bit ("Sales Down, Discounts Up: Hepatitis C Market Feeling Zepatier Launch" - "The Pink Sheet," May 9, 2016).
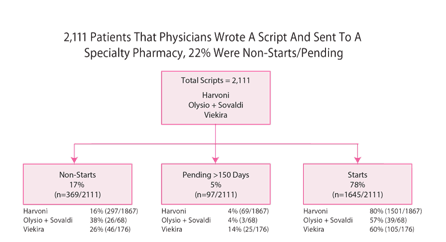
Trio Health, a disease management consulting firm that works with academic medical centers, community physicians and specialty pharmacies, found that of the 2,111 cases it reviewed, 22% of patients either did not start therapy or the decision was still pending more than 150 days after the initial prescription was written. There were 369 non-starts of therapy (17% of the overall sample) and 97 pending cases (5%). Most of the non-starts were due to denials of coverage by payers/insurers, comprising both private- and public-sector care, Trio Health reported.
By regimen, 80% (1,501/1,867) of patients prescribed Gilead Sciences
Inc.'s Harvoni started treatment, compared to 60% of those prescribed AbbVie Inc.'s Viekira Pak and 57% of those who were to take Johnson & Johnson's Olysio in tandem with sofosbuvir. The non-start rates for the three therapeutic regimens were 38% for Olysio/sofosbuvir, 26% for Viekira and 16% for Harvoni.
Rate Of Non-Starts Across HCV Regimens
Source: Trio Health
Non-starts/pending decisions occurred more frequently in the community health care setting, 26% (272/1,035), than in the academic health center setting, 18% (194/1,076).
Stigmatization Of "High-Risk" Behavior Likely A Factor
In an interview, Trio Health CEO Brent Clough suggested that denials of coverage occurred at least in part because some view HCV as "a shameful disease" and argued that similar improvements in therapy for cancer would not have resulted in denials of coverage to the same extent. Since the first DAA drugs for HCV reached market in 2011 (protease inhibitors Incivek (telaprevir) from Vertex Pharmaceuticals Inc. and Victrelis (boceprevir) from Merck & Co. Inc.), cure rates have risen and safety and tolerability have improved, while the duration of therapy has decreased significantly.
Prior to the launch of Incivek and Victrelis, the standard of care was a 48-week course of oral ribavirin daily and injectable pegylated interferon weekly, with cure rates in the 55% range. During the DAA era, duration of treatment decreased to 24 weeks, then 12 and now eight in some cases, with cure rates rising above 90% and fewer severe side effects, such as the flu-like symptoms that often are seen with interferon therapy.
"Think about if Gilead, Merck and AbbVie came up with a cure for cancer, and they said, 'if you take one pill a day with virtually no side effects … for eight weeks or 12 weeks, you have a 95% chance that you'll be cured of your cancer,'" Clough said. "There's not one payer and no one in our country that I can think of that would say, "Sorry, we're not going to make that drug available to you.'"
The 22% denial or delayed start of care that Trio Health found indicates that HCV patients are stigmatized for presumably having engaged in "high-risk behavior and so forth," the exec added.
"What you saw in a lot of the press last year was that the pricing of these drugs was 'breaking the bank,'" Clough continued. And although the perception has been that patients with less-severe disease were not being treated, the Trio analysis suggests non-treatment extended beyond such patients.
"When you look at our data, and especially as we look specifically at Medicaid, we saw that Tennessee had almost a 96% denial rate. And, specifically, 32% of the Medicaid patients with F4 [fibrosis scores] were non-starter or pending. And we saw the same thing with F3 patients."
"I would argue that discriminating against any patient is a problem, but we're talking about a cirrhotic patient who potentially could be hospitalized, need a liver transplant and end up costing the system half-a-million dollars," Clough noted.
Source: Trio Health
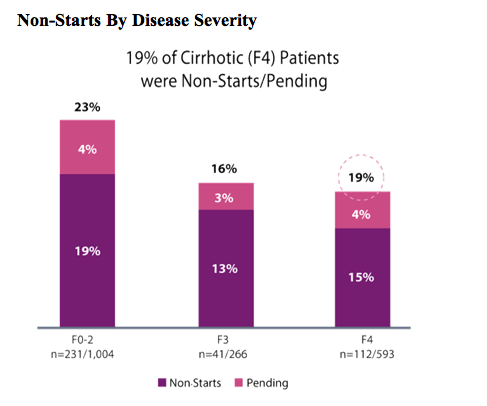
Overall, Trio Health found that 23% (231/1,004) of HCV patients with Metavir fibrosis scores ranging from F0 to F2 (non-cirrhotic, ranging from no fibrosis to fibrosis just beginning to spread) were not treated, with 19% being non-starts and 4% being decision-pending. Of F3 patients (those with fibrosis that is spreading within the liver), 16% (41/266) went untreated, with 13% being non-starts and 3% pending. Even among patients with scores of F4 (cirrhosis or advanced fibrosis) on the Metavir liver biopsy scale, 19% (112/593) were not approved for therapy, with 15% denied treatment and 4% pending.
While disease severity didn't seem to greatly affect treatment decisions, baseline viral load also was not shown to be a determining factor, the data indicated. For patients with lower baseline viral loads 22% (357/1,642) did not start therapy, while for higher baseline loads, 20% (79/378) went untreated. In both cases, 17% were denied treatment, while 5% with lower baseline viral loads and 3% with higher levels were pending decisions.
Medicaid Patients Denied Treatment More Frequently
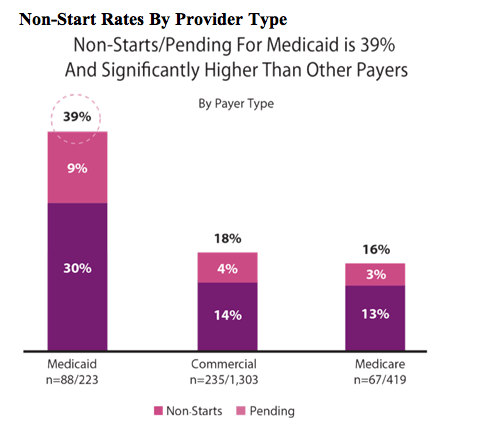
What did tend to be a significant gating factor to therapy was whether or not the patient's health care coverage was through Medicaid. Thirty-nine percent (88/223) of Medicaid patients went uncovered, with 30% turned down for treatment and 9% left pending. Meanwhile, for those with private, commercial insurance, 18% (235/1,303) went untreated (14% non-start; 4% pending), while Medicare patients had a 16% (67/419) non-treated rate (13% non-start; 3% pending).
Source: Trio Health
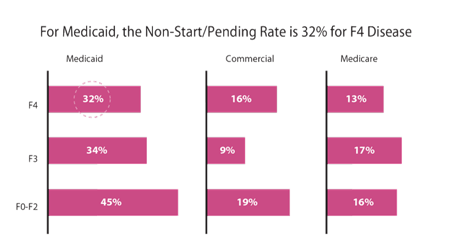
Of patients with F4 Metavir scores, 32% were non-start/pending in Medicaid, compared to 16% of the commercial insurance sample and 13% of Medicare recipients. For F3 patients, 34% in Medicaid, 17% in Medicare and 9% under commercial plans did not start therapy. Among those with scores in the F0-F2 range, the non-start/pending rates were 45% for Medicaid, 19% commercial and 16% Medicare.
Non-Start Rates For F4 Fibrosis Score Patients
Source: Trio Health
Within Medicaid, geography proved to be a decisive factor in whether an HCV patient got treated, as Trio Health's data showed 96% (24/25) in Tennessee's program did not get treated. Similarly, 73% (11/15) of Medicaid patients in Arkansas, 55% (6/11) in Oklahoma and 52% (23/44) in Illinois were non-starts or awaiting a decision.
Specific state situations could be related to burning through funding or setting too low of a budget, Clough acknowledged. "With respect to TennCare, for a period of time they denied all patients since they had not budgeted for the cost for hep C patients. Thereafter, they severely restricted access by changing the reimbursement to [specialty pharmacy providers] that would have cost each SPP over $1,500 per dispense. [There are] lots of games that can be played to prevent access to care."
HCV patients covered by the Department of Veterans Affairs also ran into coverage issues, as the department spent $370m on DAA therapies in 2014 but ran out of funding, bringing treatment to a standstill temporarily. Going forward, VA planned to spend about $1.3bn on HCV therapy during fiscal years 2015-2016 ("VA Spent $370 Million On Sovaldi And Olysio In Fiscal 2014" - "The Pink Sheet" DAILY, Dec. 3, 2014).
When Trio Health shared the data with the Centers for Medicare and Medicaid Services, "the federal government came out issuing awarning letter to all the states, telling them what they were doing was illegal," he said ("CMS Wants To Facilitate Value-Based Discounts For HCV Drugs In Medicaid" - "The Pink Sheet" DAILY, Nov. 6, 2015). "CMS said that denying care to hep C patients is illegal."
"There was kind of nowhere [for payers] to hide," Clough added.
Trio Health believes that policy announcement began "opening the floodgates" for treatment across the range of fibrosis scores, he added. The firm will conduct a follow-on study this year to see if patient access has improved, Clough said.
Among private payers, the highest non-start/pending cases were in Express Scripts plans, followed by CVS Caremark and Blue Cross.
The data analysis also found that, among patients with private-sector insurance, the non-start/pending rate was highest among those within Express Scripts Holding Co. plans at 24% (72/295), followed by 21% for CVS Health Corp./Caremark Rx Inc. plans and 14% (34/251) for Blue Cross Blue Shield plans.
Asked how to assess the varying coverage rates within different commercial plans and also among different states' Medicaid programs, Clough said he couldn't find any rationale other than a desire to hold the line on spending.
"I think that's the reason," he said. "It's the same thing with TennCare or Tennessee Medicaid. We clearly demonstrated that Tennessee has F4 cirrhotic patients who were denied access. You have a highly efficacious, highly safe drug in terms of treatment options. The mode of administration is easy, simple and straightforward. You have patients who clearly have the severity and require treatment that are being denied. So, I'm not sure what else is left other than cost and price."
Trying to determine specifically why coverage was not started or delayed by a pending decision, Trio Health found that financial or insurance reasons were the cause 70% of the time, physician-directed 17% of the time, patient-determined 11% of the time and unspecified in 2% of cases.
Of the 369 non-starts in the sample, 25% (93) were under appeal of an insurance denial, 22% (81) were an insurance denial with reason not specified and 19% (71) were due to patient action or an appeal that was awaiting a physician's documentation. Only 2% that were financial or insurance related were due to the patient not being able to afford treatment (7) or the decision being in pre-authorization (6).
Of the 62 cases (17%) in which start of therapy did not occur due to physician's orders, all were due to patient non-compliance (continuation of drug use) prior to treatment, the study found. Where patient action was the reason for a non-start, there were 25 cases (7%) in which the patient was unreachable and 16 (4%) in which the patient had not complied with the need for lab testing such as drug screening.
The upside of the study was confirming that the drugs remain highly efficacious, with the success rates in the real world at least approaching the high sustained virologic response (SVR) rates seen in clinical studies. Of those who got treated, 97% (896) were cured on Harvoni, 91% (20) on the Olysio/sofosbuvir combination and 85% (36) on Viekira Pak. SVR was not achieved by 2% (20) of the Harvoni patients, with 13 discontinuations (1%). Two patients (9%) did not achieve SVR on the Olysio/sofosbuvir regimen, with no discontinuations, while Viekira saw two treatment-failures (5%) and four discontinuations (10%).
Clough said these rates indicate that the era of "warehousing" of patients for better therapeutic regimens should be all but ended. "I think cure rates are almost a commodity," he asserted. "What's the difference between 93% and 96%? I understand that every basis point counts, but it's really in my mind about access and affordability."
|
|
| |
| |
|
|
|