 |
 |
 |
| |
Novel Integrase Mutation in Macaque Cabotegravir Study--But "Limited" Clinical Impact
|
| |
| |
HIV Research for Prevention (HIVR4P), October 17-19, 2016, Chicago
Mark Mascolini
Subtherapeutic doses of long-acting injected cabotegravir (CAB LA), an integrase inhibitor, led to emergence of a novel mutation in macaques that resulted in high-level resistance [1]. This five amino acid (5AA) duplication at integrase position 232 also made simian immunodeficiency virus (SIV) resistant to currently licensed integrase inhibitors. But researchers from the Aaron Diamond AIDS Research Center who conducted this study believe the 5AA mutation will have "limited" clinical impact.
Formulated as a 200-mg/mL nanosuspension, CAB LA is under study for PrEP [2] and for maintenance therapy with rilpivirine [3]. CAB LA, which may be amenable to dosing every 4 to 8 weeks, protects macaques from intrarectal, intravaginal, and intravenous challenges with SHIV162P3 and SIVmac251 [4,5]. Cabotegravir plasma levels attained in macaques are similar to those in humans [4].
In macaque studies of CAB LA, resistance did not emerge in breakthrough infections. In humans treated with cabotegravir, emergent resistance is associated with the Q148R mutation, which also confers resistance to elvitegravir and raltegravir.
The Aaron Diamond team conducted this study to assess selection of virus resistant to cabotegravir in SIVmac251-infected rhesus macaques. The analysis involved 4 intravenously infected macaques given CAB LA at a subtherapeutic 10-mg/kg dose 13 weeks after infection, followed by 50-mg/kg therapeutic doses at 2- to 4-week intervals. The researchers monitored viral loads and cabotegravir concentrations weekly and searched for mutations in the integrase region of viral RNA.
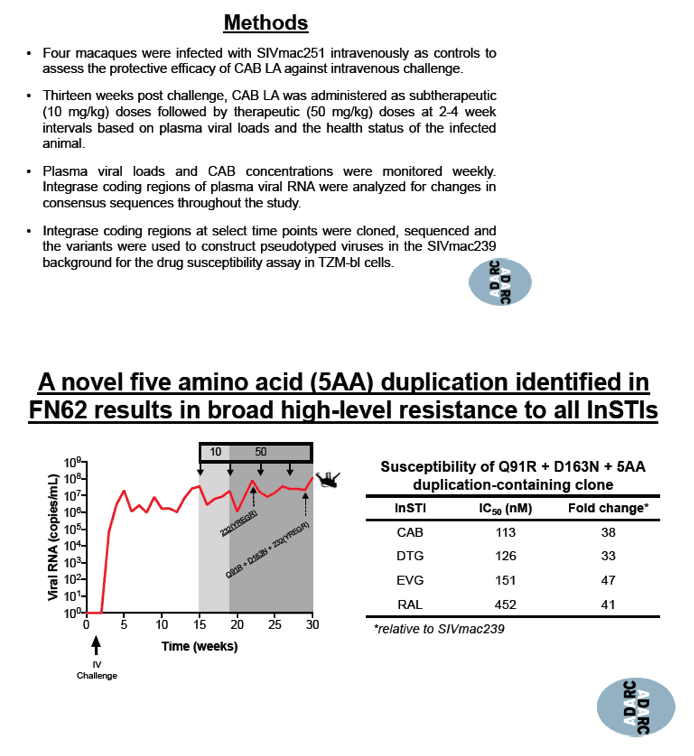
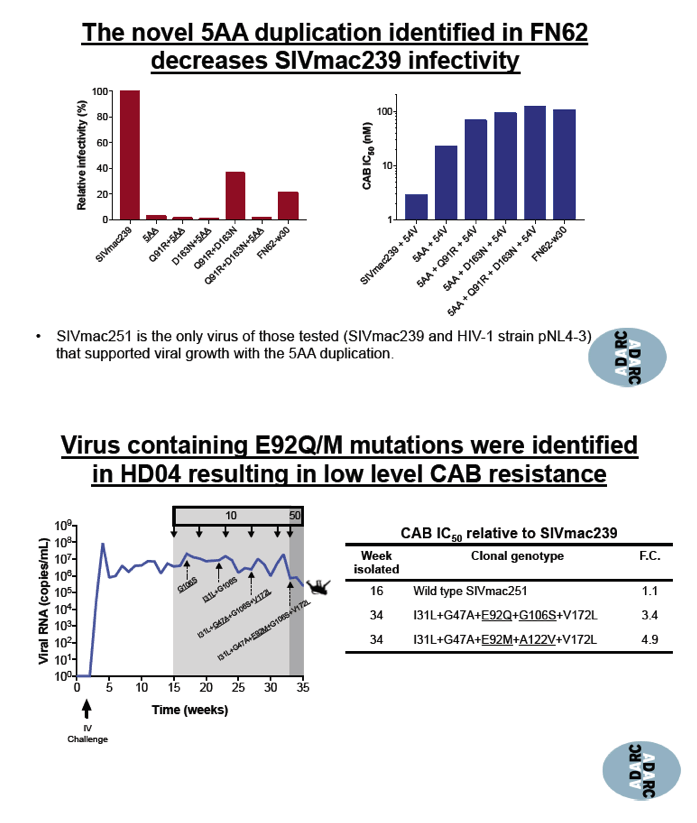
A novel 5 amino acid duplication detected in 1 macaque and designated 5AA resulted in high-level resistance to cabotegravir, dolutegravir, elvitegravir, and raltegravir. Combined with the Q91R and D163N mutations, 5AA yielded a 38-fold increase in 50% inhibitory concentration (IC50) with cabotegravir, a 33-fold increase with dolutegravir, a 47-fold increase with elvitegravir, and a 41-fold increase with raltegravir. Of 3 viruses tested--SIVmac251, SIVmac239, and HIV-1 pNL4-3--SIVmac251 was the only virus that allowed viral growth while harboring the 5AA duplication. This novel duplication decreased infectivity of SIVmac239 and the HIV-1 strain.
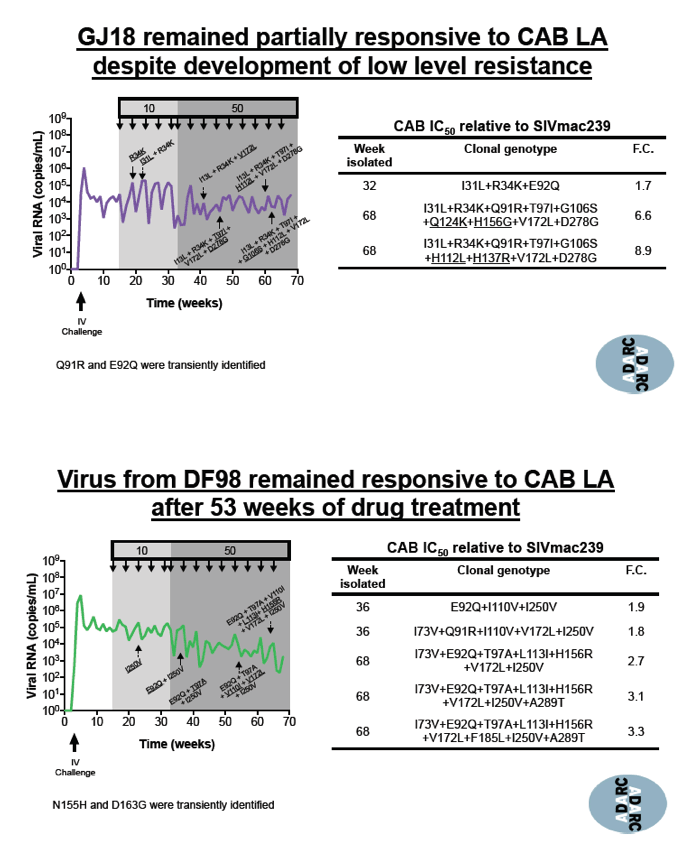
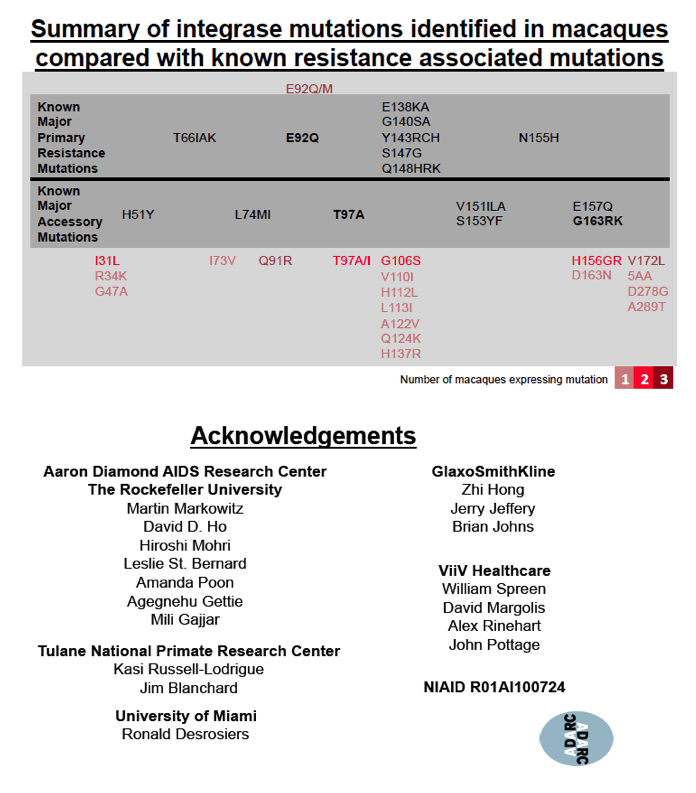
In the other 3 macaques, 10 to 50 mg/kg of CAB LA for up to 53 weeks prompted emergence of 7 other cabotegravir-resistant mutations: I31L, Q91R, E92Q/M, T97A/I, G106S, H156G/R, and V172L. These mutations resulted in low-level resistance with IC50 increases ranging from 3.3-fold to 8.9-fold. The researchers stressed that these mutations "were forcefully induced through conditions that do not mimic clinical practice." No single mutation affected IC50 in these macaques, as Q148R does with HIV-1. Thus the investigators concluded that the clinical relevance of this induced low-level resistance remains unknown.
As for the 5AA duplication, the Aaron Diamond team proposed that "the clinical significance of this mutation is limited."
References
1. Andrews CD, Mohri H, St. Bernard L, et al. Evaluation of resistance to cabotegravir long-acting (CAB LA) in SIVmac251-infected macaques. HIV Research for Prevention (HIVR4P 2016), October 17-19, 2016, Chicago. Abstract OA03.04.
2. Markowitz M, Frank I, Grant R, et al. ECLAIR: Phase 2A safety and PK study of cabotegravir LA in HIV-uninfected men. Conference on Retroviruses and Opportunistic Infections (CROI), February 22-25, 2016, Boston. Abstract 106.
3. ViiV Healthcare. A phase IIb study evaluating a long-acting intramuscular regimen of GSK1265744 plus TMC278 for the maintenance of virologic suppression following an induction of virologic suppression on an oral regimen of GSK1265744 plus abacavir/lamivudine in HIV-1 infected, antiretroviral therapy-naive adult subjects. NLM Identifier NCT02120352. http://www.clinicaltrials.gov/ct2/show/NCT02120352
4. Andrews CD, Spreen WR, Mohri H, et al. Long-acting integrase inhibitor protects macaques from intrarectal simian/human immunodeficiency virus. Science. 2014;343:1151-1154. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4308974/
5. Andrews CD, St. Bernard L, Poon A, et al. Cabotegravir long-acting injection protects macaques against intravenous challenge. Conference on Retroviruses and Opportunistic Infections (CROI), February 22-25, 2016, Boston. Abstract 105. http://www.natap.org/2016/CROI/croi_38.htm


|
| |
|
 |
 |
|
|