 |
 |
 |
| |
Pre-exposure Prophylaxis with EFdA Offers Strong Protection against High Dose Mucosal HIV Challenges - [Long-Acting New NRTI for Treatment & PrEP]
|
| |
| |
Reported by Jules Levin
HIVR4P Oct 18-20 2016 Chicago
"Our data demonstrated that EFdA efficiently prevents both vaginal and oral HIV transmission. Together with EFdA's relatively low toxicity and high potency against drug-resistant HIV strains, these data support further clinical development of EFdA as a potential pre-exposure prophylaxis (PrEP) agent to prevent HIV transmission in women and their infants."
EFdA / MK-8591 - HIV pre-exposure prophylaxis for women and infants prevents vaginal and oral HIV transmission in a preclinical model of HIV infection - (08/05/16)
Parenteral formulations exhibit >180 day extended release after a single injection in rats. Ongoing studies suggest the potential to provide coverage for durations up to a year.....http://natap.org/2016/CROI/croi_14.htm
....virologic activity of MK-8591 in macaques given once weekly oral doses ranging from 1.3 to 18.2 mg/kg, or a once daily oral dose of 0.19 mg/kg. The maximal virologic decline (approximately 1.5 log decrease in viral load) was seen with weekly doses of 3.9 to 18.2 mg/kg once weekly, and was correlated with intracellular-triphosphate concentrations of MK-8591 of ≥ 0.53 pmol/10 million cells.
Significant gender inequalities limit the ability of women to access and exercise HIV prevention options resulting in earlier acquisition of infection, higher transmission rates and increased mother-to-child transmission of HIV. Collectively, our in vivo results demonstrating 4'-ethynyl-2-fluoro-2'-deoxyadenosine's excellent efficacy in preventing both vaginal and oral HIV transmission together with its relatively low toxicity and high potency against drug-resistant HIV strains support 4'-ethynyl-2-fluoro-2'-deoxyadenosine as a potential PrEP agent for use in women and infants, two of the most vulnerable populations at risk for acquiring HIV.
Viral suppression was maintained for at least 7 days after the last dose. This study provides proof of concept for a once weekly oral dosing strategy of MK-8591. In Q&A, the authors stated their plan to develop MK-8591 for prevention and treatment, and acknowledged for the latter they would need other agents to form a complete, once weekly regimen and that they were committed to that goal.

"EFdA Distributes and Penetrates into Mucosal Sites of Vaginal and
Oral HIV Transmission"
Martina Kovarova1, Uma Shanmugasundaram1, Caroline Baker1, Rae Ann Spagnuolo1, Chandrav De1, Christopher Nixon1, Angela Wahl1, J. Victor Garcia1
1University of North Carolina, School of Medicine, United States
Background: EFdA (4'-Ethynyl-2-Fluoro-2'-Deoxyadenosine) is a new NRTI with antiretroviral potency several orders of magnitude higher than any of the current clinically used NRTIs. It has favorable toxicity profiles in vitro and in vivo and effectively inhibits clinically important drug resistant HIVs. Using humanized BLT (bone marrow-liver-thymus) mice as a pre-clinical model to evaluate HIV prevention strategies, we investigated the potential of EFdA to prevent mucosal transmission of HIV at the two primary sites of HIV acquisition in women and infants (vagina and oral mucosa).
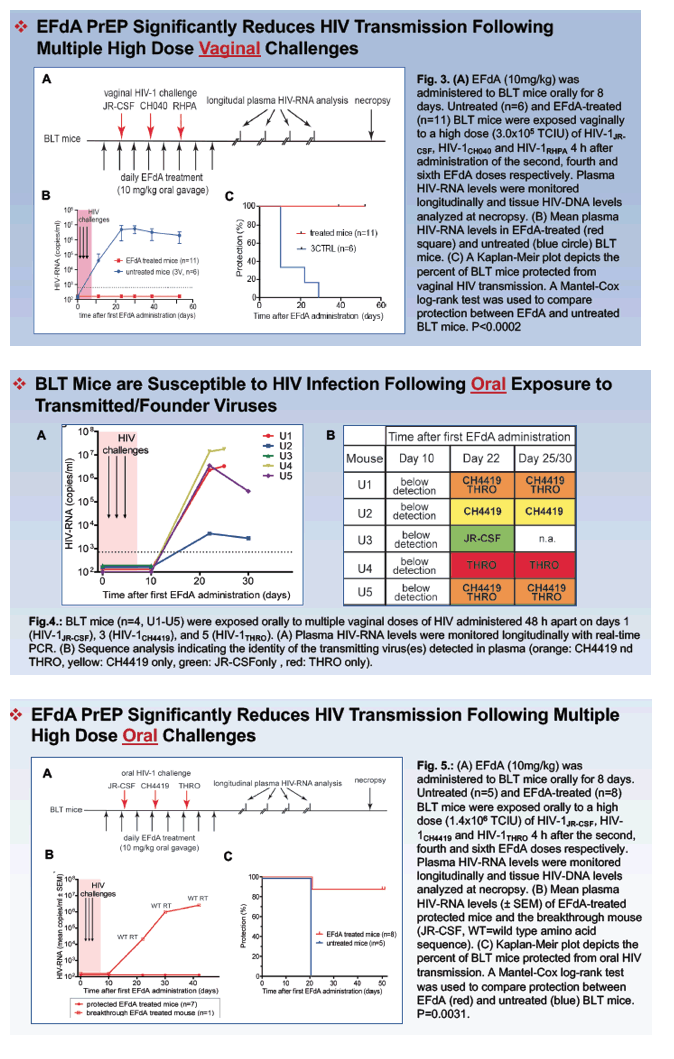
Methods: EFdA was administered orally to NSG mice and to humanized BLT mice. HIV inhibitory activity in serum, cervicovaginal secretions and saliva was evaluated 4 h after administration. EFdA ability to prevent vaginal and oral HIV transmission after multiple high-dose HIV exposures was evaluated using highly relevant transmitted-founder (T/F) viruses in BLT mice.
Results:
After a single oral dose of EFdA we observed strong HIV inhibitory activity in the peripheral blood serum (p=0.0031), cervico-vaginal secretions (p=0.0016), and saliva (p=0.0161) from EFdA-treated mice compared to untreated controls. These data indicate systemic distribution of active EFdA after a single oral administration and efficient penetration into the female reproductive tract and the oral cavity. Consistent with these results, a single daily oral dose of EFdA also resulted in efficient prevention of vaginal (p=0.0002) and oral (p=0.0031) HIV transmission after multiple high-dose exposures to various T/F viruses in BLT humanized mice.
Conclusions: Our data demonstrated that EFdA efficiently prevents both vaginal and oral HIV transmission. Together with EFdA's relatively low toxicity and high potency against drug-resistant HIV strains, these data support further clinical development of EFdA as a potential pre-exposure prophylaxis (PrEP) agent to prevent HIV transmission in women and their infants.
|
| |
|
 |
 |
|
|