 |
 |
 |
| |
Microbiome Composition & Function Drives Would-Healing Impairment in the Female Genital Tract
|
| |
| |
IAC 2016/Durban / Discussed / 4-5 studies /plenary session on these issues:
New Evidence: Why Do Young Women in Africa Have High Rates of HIV Infection? - (08/03/16)
Microbiome Dysfunction/ HIV Acquistion
http://webcasts.hivr4p.org/console/player/33026?mediaType=audio&
Alexander Zevin1, Irene Xie2, Kenzie Birse2, Kelly Arnold3, Laura Romas2, Garrett Westmacott2, Richard Novak4, Lyle McKinnon5, Romel Mackelprang1, Jairam Lingappa1, Douglas A. Lauffenburger6, Nichole Klatt1, Adam D. Burgener2
1University of Washington, United States, 2Public Health Agency of Canada, Canada, 3University of Michigan, United States, 4University of Illinois at Chicago, United States, 5University of Manitoba, Canada, 6Massachusetts Institute of Technology, United States
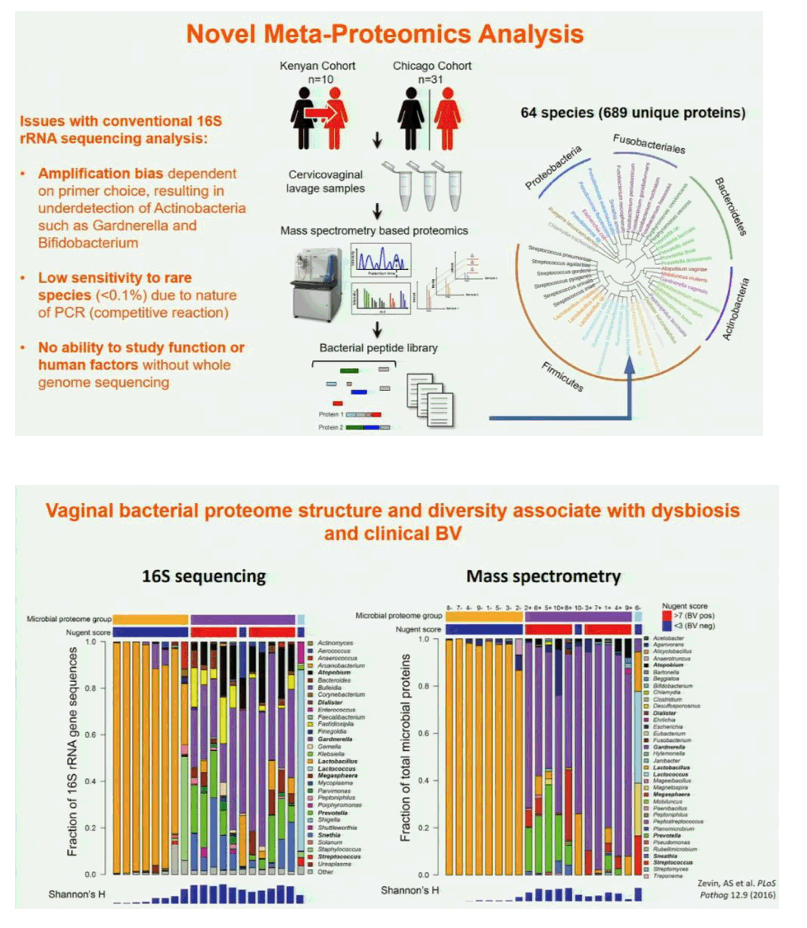
Background: The mechanism by which microbial communities impact disease susceptibility in the female genital tract (FGT), such as HIV, are poorly understood. We utilized a combination of metaproteomic, metagenomic, and in vitro epithelial models to better understand bacterial:host interactions in the FGT, which represents the first study to take this comprehensive approach.
Methods: Both longitudinal (Cohort 1, n=10) and cross-sectional (Cohort 2, n=31) cervicovaginal samples were collected. Nugent/Amsel's criteria was used for diagnosis of bacterial vaginosis. Samples were analyzed using a combination of mass spectrometry, 16s rRNA gene sequencing, and in vitro bacterial:epithelial cell co-culture assays.
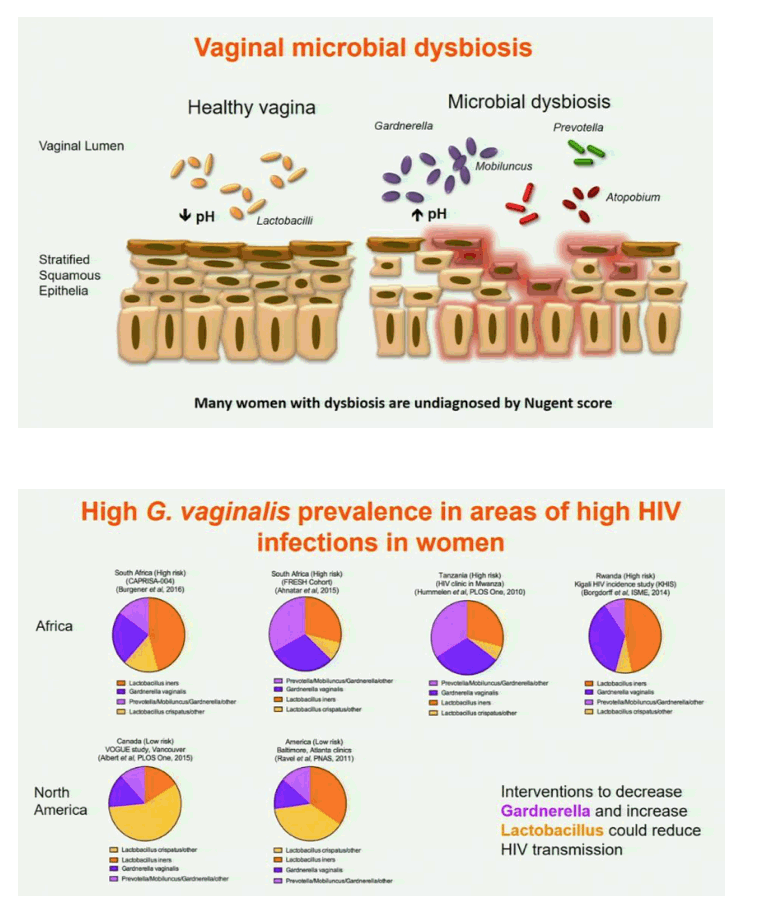
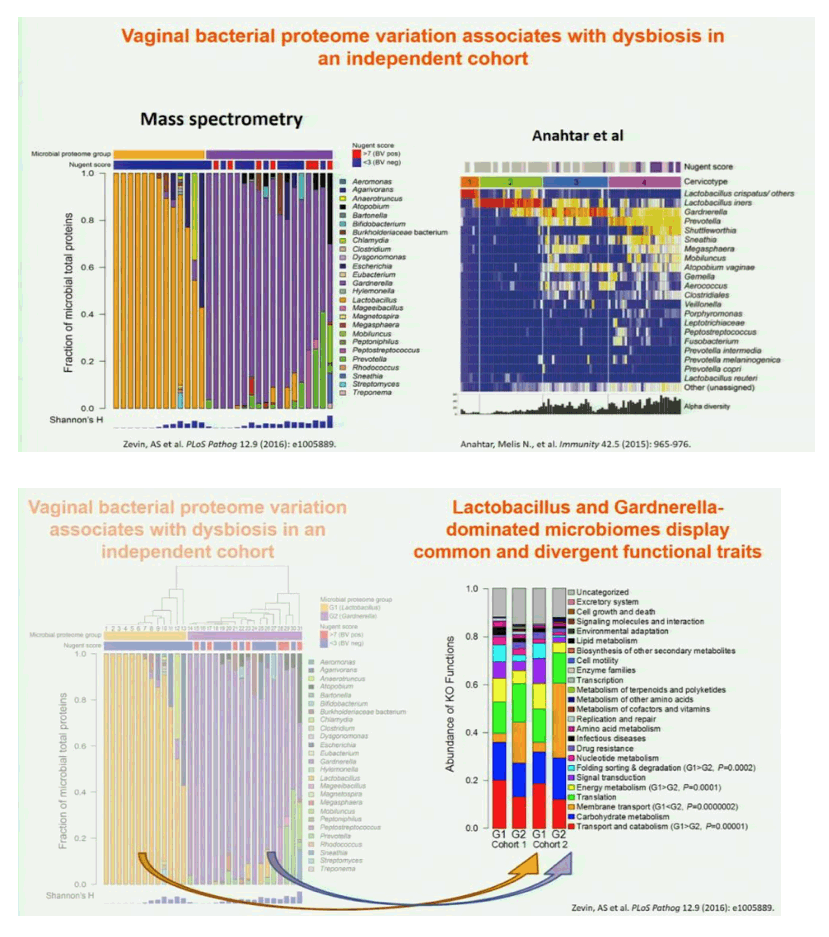
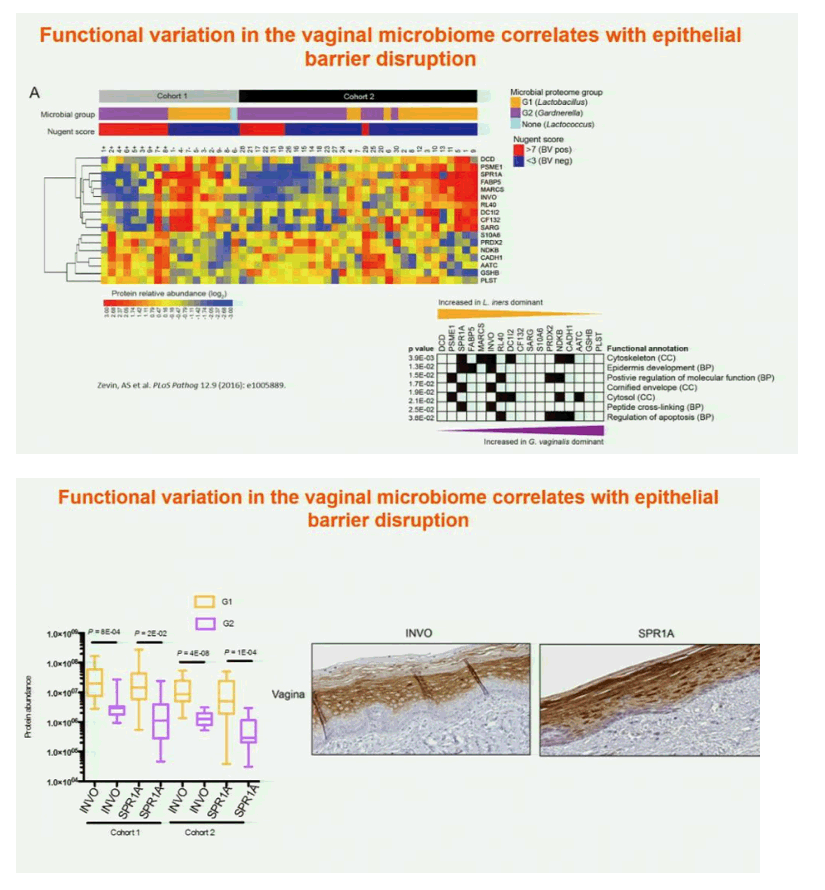
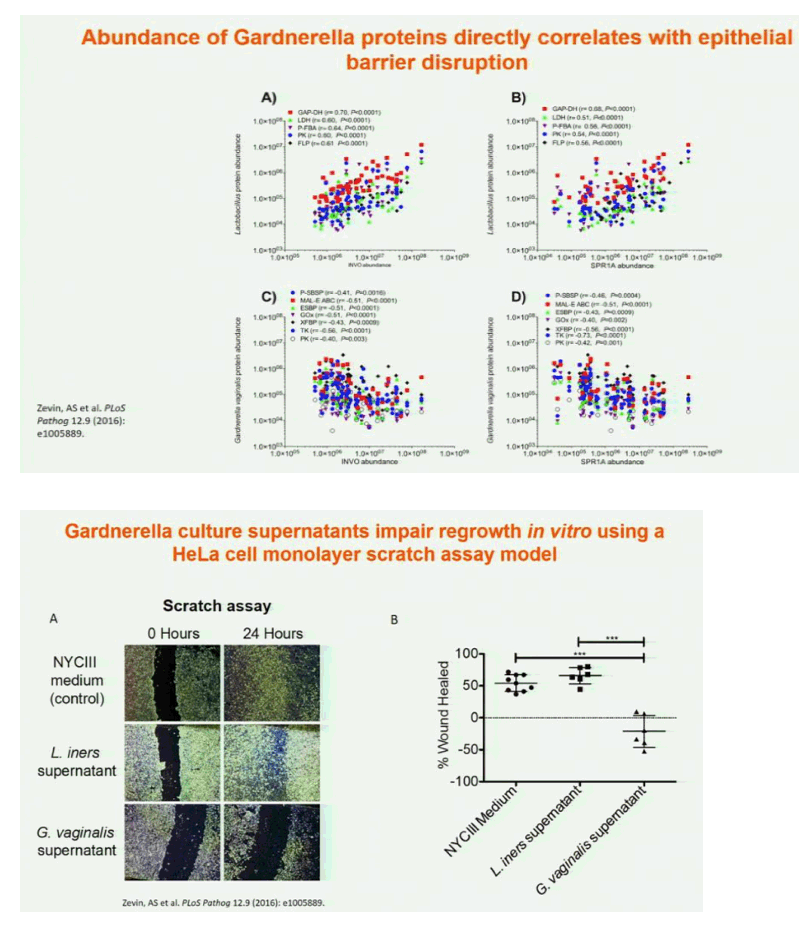
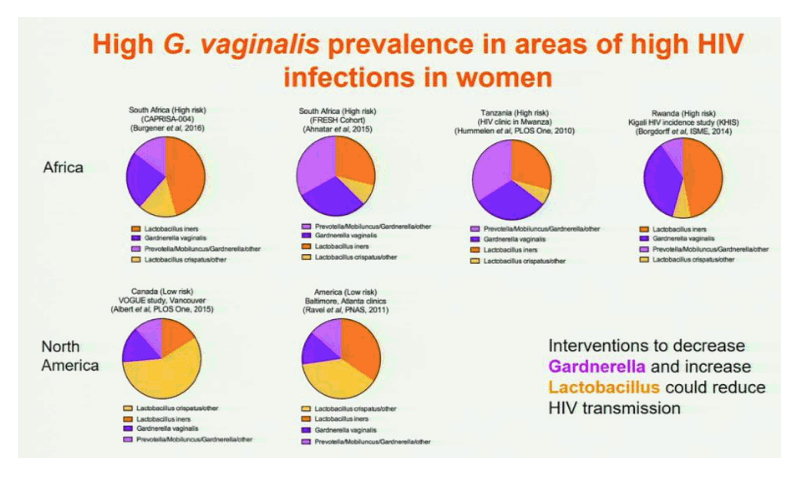
Results: In both cohorts, highly diverse, G. vaginalis dominated FGT microbial communities associated with significant shifts in microbial function, higher Nugent scores and vaginal pH. Host pathways relating to epithelial barrier disruption and inflammation were enhanced in G. vaginalis dominated profiles, associating with extracellular and metabolic proteins belonging to G. vaginalis. Conversely, host proteins important for maintaining epithelial integrity such as envelope cornification, tight-junction formation, and wound-healing, were enhanced in Lactobacillus spp. dominated profiles, associating with glycolysis and homolactic fermentation, lower Nugent scores and pH. Soluble products derived from G. vaginalis culture inhibited wound healing of cervical epithelial cells, while soluble products derived from L. iners culture resulted in effective wound healing.
Conclusions: These data identify the first mechanisms demonstrating diverse, G. vaginalis bacterial communities drive key functional microbial pathways, which decrease epithelial barrier integrity and prevent wound healing in the FGT. Thus, future studies addressing how to combat diminished epithelial integrity, via altering bacterial communities or inhibiting host/microbial pathways identified here, may lead to novel prevention strategies.

|
| |
|
 |
 |
|
|