 |
 |
 |
| |
Raltegravir containing antiretroviral therapy for prevention of mother to child transmission in a high risk population of HIV-infected pregnant women in Buenos Aires, Argentina: maternal and neonatal outcomes....
|
| |
| |
"No vertical transmission was documented to date"
Reported by Jules levin
Durban 2016 July 18-22
Presenter
Diego Martin Cecchini
Authors
D.M. Cecchini1, M. Martinez2, L. Morganti1, C. Rodriguez1
Institutions
1Hospital Cosme Argerich, Infectious Diseases Unit, Buenos Aires, Argentina, 2Hospital Cosme Argerich, Neonatology Unit, Buenos Aires, Argentina

Abstract
Background: Mother-to-child-transmission of HIV-1 in Buenos Aires remained high (4-7%) in recent years: novel strategies for reduction are needed. There is limited information regarding the use of raltegravir (RAL) in subpopulations of HIV-infected pregnant women (HIPW) with high transmission rates (late presenters [LP] and those with detectable antepartum viral load [VL] despite antiretroviral therapy [ART]). We aimed to evaluate: 1) trends in RAL prescription in HIPW in our institution; 2) maternal/neonatal outcomes after RAL exposure.
Methods: Retrospective study in a general hospital in Buenos Aires, Argentina (2009-2015).
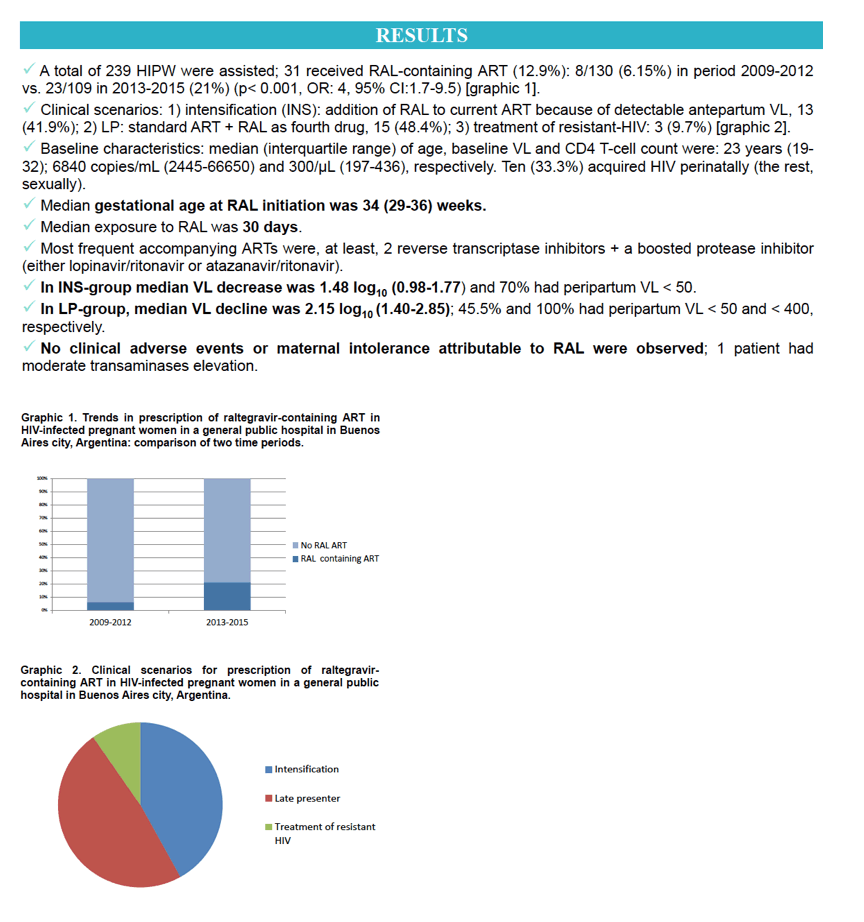
Results: A total of 239 HIPW were assisted; 31 received RAL-containig ART (12.9%): 8/130 (6.15%) in period 2009-2012 vs. 23/109 in 2013-2015 (21%) (p< 0.001 OR: 4, 95%CI:1.7-9.5). Clinical scenarios: 1) intensification (INS): addition of RAL to current ART because of detectable antepartum VL, 13 (41.9%); 2) LP: standard ART + RAL as fourth drug, 15 (48.4%); 3) treatment of resistant-HIV: 3 (9.7%). The median (interquartile range) of age, baseline VL and CD4 T-cell count were: 23 years (19-32); 6840 copies/mL (2445-66650) and 300/μL (197-436), respectively. Ten (33.3%) acquired HIV perinatally (the rest, sexually). Median gestational age at RAL initiation was 34 (29-36) weeks and median exposure was 30 days. In INS-group median VL decrease was 1.48 log10 (0.98-1.77) and 70% had peripartum VL < 50. In LP-group, median VL decline was 2.15 log10 (1.40-2.85); 45.5% and 100% had peripartum VL < 50 and < 400, respectively. No clinical adverse events or maternal intolerance attributable to RAL were observed; 1 had moderate transaminases elevation. One patient had a stillbirth. Elective cesarean section was done in 51.7%; 18.5% of births were preterm. Neonatal prophylaxis: AZT in 57.1% (the rest, combined prophylaxis). Mild elevation of transaminases was observed in 35% of neonates; 14.8% required phototherapy. No vertical transmission was documented to date (all infants had a negative PCR in the first week, 70% at >2 months and 23% had negative 18-month ELISA).
Conclusions: RAL prescription in HIPW increased in the recent years in our institution, mainly in high risk populations. A rapid maternal virological response with RAL containg ART was observed, without major cautionary signals or perinatal transmission.
|
| |
|
 |
 |
|
|