 |
 |
 |
| |
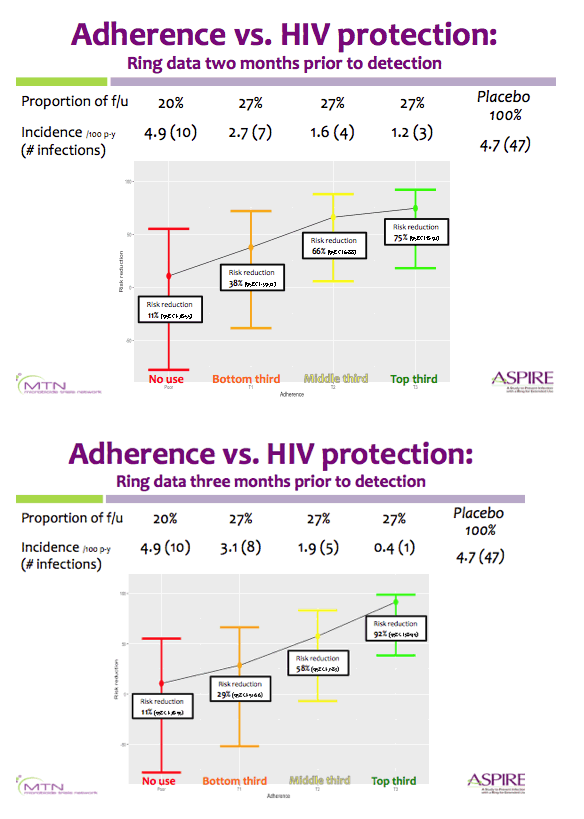
Residual dapivirine ring levels indicate higher adherence to vaginal ring is associated with HIV-1 protection ......."Results suggest ring use is associated with at least 56% and potentially >75% protection when used consistently"
|
| |
| |
Reported by Jules Levin
Durban 2016 July 18-22
E. Brown1,2, T. Palanee-Philips3, M. Marzinke4, C. Hendrix4, C. Dezutti5, L. Soto-Torres6, J. Baeten7
Institutions
1Fred Hutchinson Cancer Research Center, Seattle, United States, 2University of Washington, Seattle, United States, 3University of the Witswaterand, Johannesburg, South Africa, 4Johns Hopkins University, Baltimore, United States, 5University of Pittsburgh, Pittsburgh, United States, 6NIAID, Rockville, United States, 7Univeristy of Washington, Seattle, United States


Abstract
Background: In MTN-020/ASPIRE, a vaginal ring containing dapivirine was found to decrease the risk of HIV-1 acquisition by 27% overall in an intention-to-treat analysis compared to placebo and by 37% in an analysis excluding data from two sites with lower adherence/retention.
In subgroup analyses, no HIV-1 protection was seen in women ≤ age 21, for whom adherence appeared lower. In studies of tenofovir-based prophylaxis, objective markers of adherence have been important in understanding HIV-1 protection when products are used.
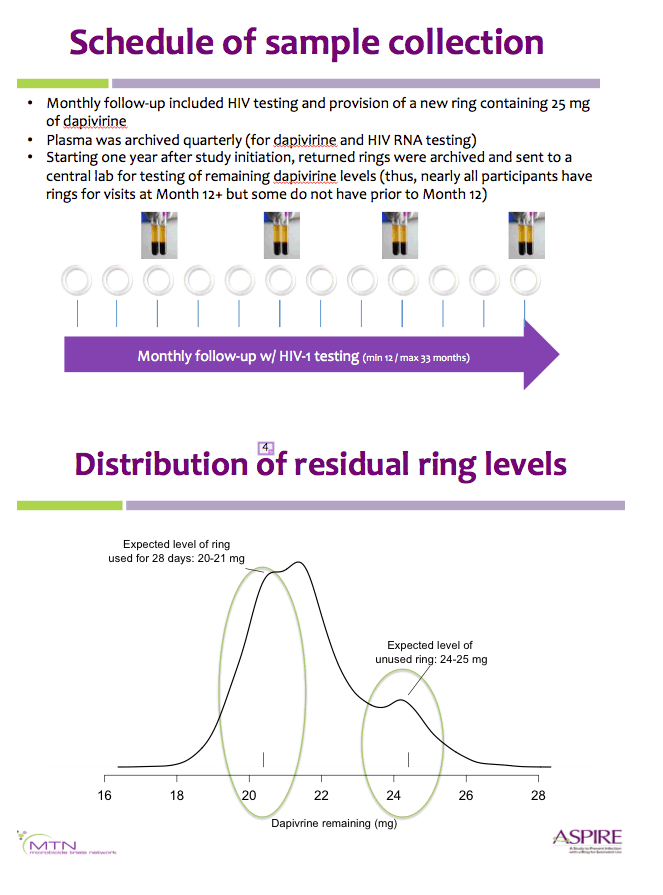
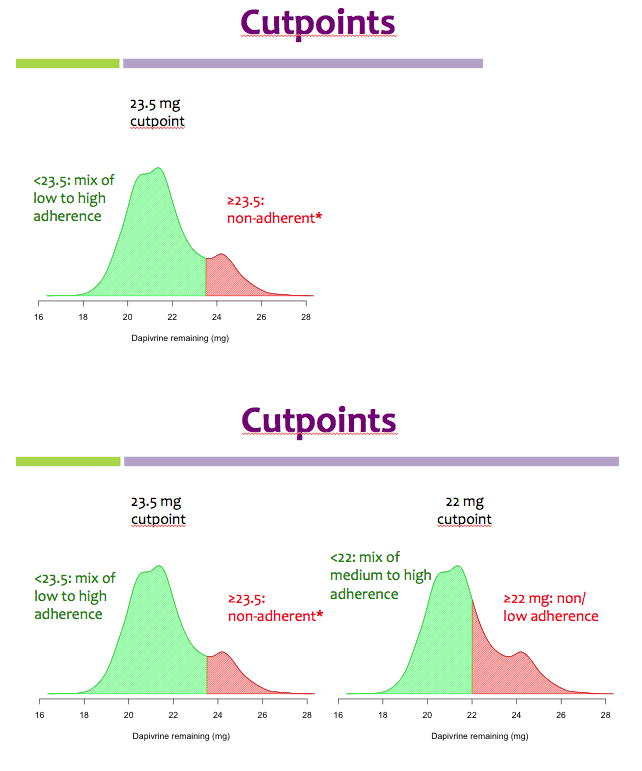
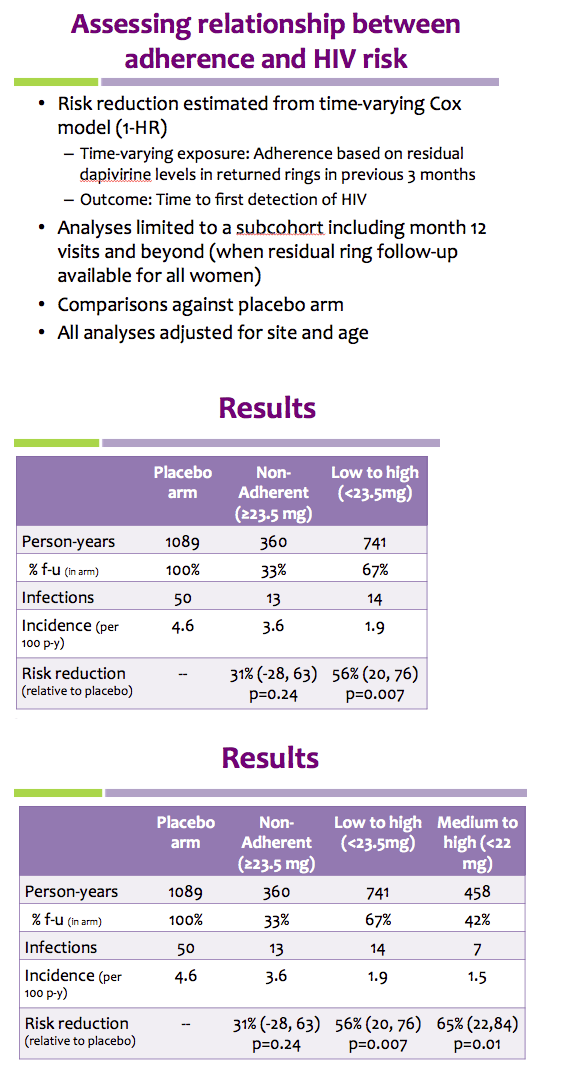
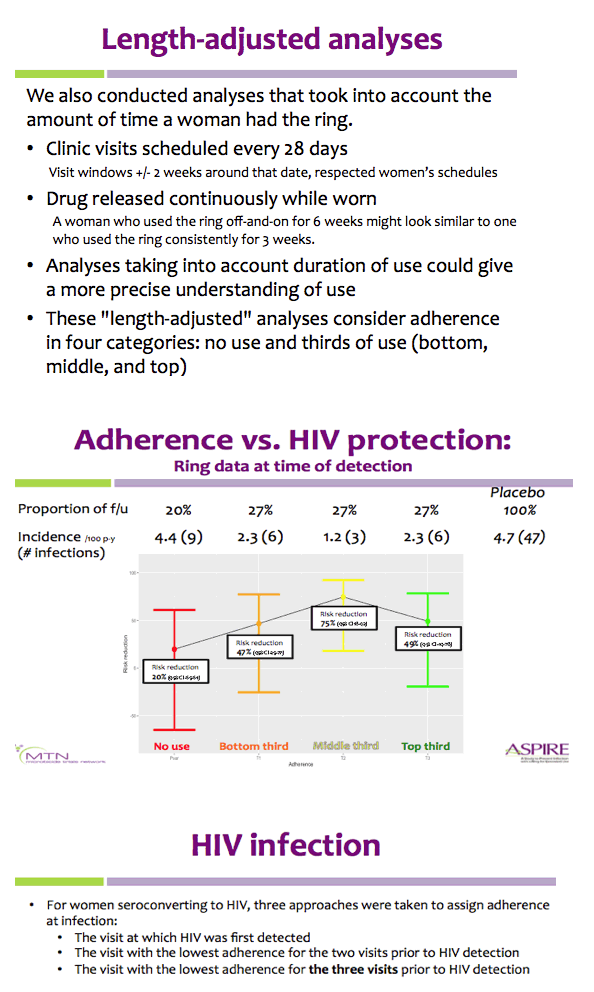
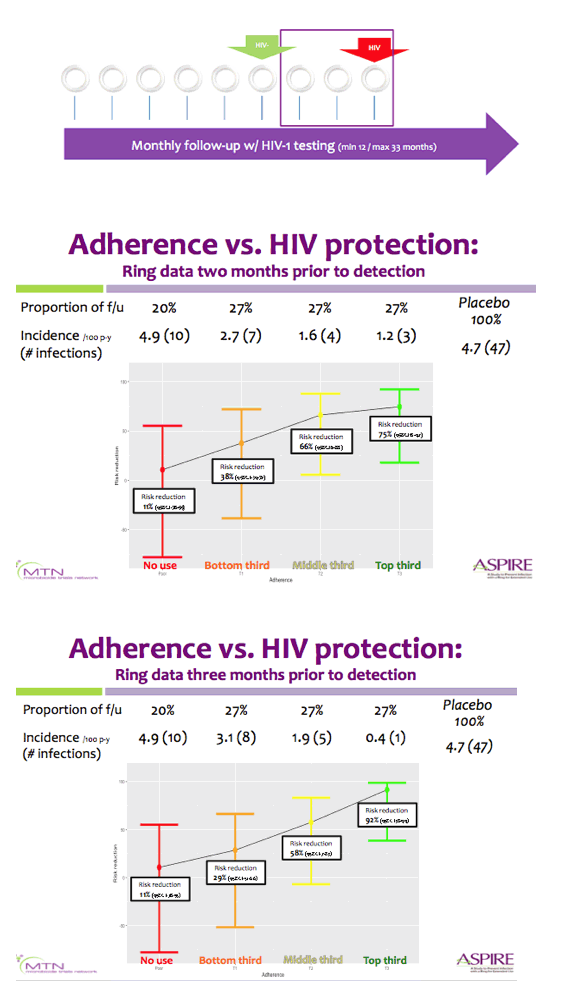
Methods: Rings were manufactured with 25 mg of dapivirine, and phase I studies indicated that ∼4 mg of dapivirine on average are released during four weeks of continuous use; therefore, levels ≤22 mg were defined as having higher adherence for the present analysis. Starting one year into the trial, we tested the residual dapivirine levels (RDL) remaining in returned, used rings in ASPIRE. Visits at which participants did not return the ring, did not have access to the ring due to product hold or refusal or had RDL >22 mg were categorized as less or non-adherent. The association between HIV-1 acquisition and adherence was assessed using time-varying covariate Cox models adjusted for age and study site, including visits occurring at month 12 and beyond.
Results: Of the 2629 women enrolled in ASPIRE, 2359 were included in this analysis. Compared to placebo, higher adherence to the active dapivirine ring (i.e., RDL≤22 mg) was associated with a 65% (95% CI 23-84, p=0.009) reduction in HIV-1 risk.
Results were similar for the full study population and when excluding the two sites with lower adherence/retention (risk reduction 67%, 95% CI 23-86), and point estimates suggested HIV-1 protection for both women >21 years (risk reduction 72%, 95% CI 21-90) and ≤21 years of age (risk reduction 50%, 95% CI -78-86).
Partial/low adherence (i.e., RDL>22 mg) was not significantly associated with HIV-1 protection (relative risk reduction 35%, 95% CI -10-61, p=0.12).
Conclusions: Residual dapivirine levels in returned rings, an objective marker of adherence, indicate that higher adherence to the dapivirine vaginal ring may provide >65% protection from HIV-1 acquisition.


|
| |
|
 |
 |
|
|