 |
 |
 |
| |
Switch to Single-Pill DTG/ABC/3TC Noninferior to Keeping Current ART
|
| |
| |
21st International AIDS Conference (AIDS 2016), July 18-22, 2016, Durban, South Africa
Mark Mascolini
Switching to one-pill once-daily dolutegravir plus abacavir/lamivudine (DTG/ABC/3TC) for 24 weeks proved virologically noninferior to staying with a suppressive regimen in the randomized open-label STRIIVING trial [1]. There were no virologic failures in either study arm through 48 weeks.
In North American DTG/ABC/3TC is licensed as Triumeq--a single-pill once-daily fixed-dose coformulation--for people at least 18 years old. From April to October 2014, STRIIVING enrolled people with a viral load below 50 copies while taking a regimen including two nucleos(t)ides plus a protease inhibitor, a nonnucleoside, or an integrase inhibitor. Researchers randomized them to maintain their current regimen for 24 weeks or switch to DTG/ABC/3TC (the early-switch group). At week 24 everyone who stayed with their initial regimen switched to DTG/ABC/3TC for another 24 weeks (the late-switch group), while the early-switch group kept taking DTG/ABC/3TC for another 24 weeks. The primary endpoint was the proportion of participants with a viral load below 50 copies at week 24 in an FDA snapshot analysis.
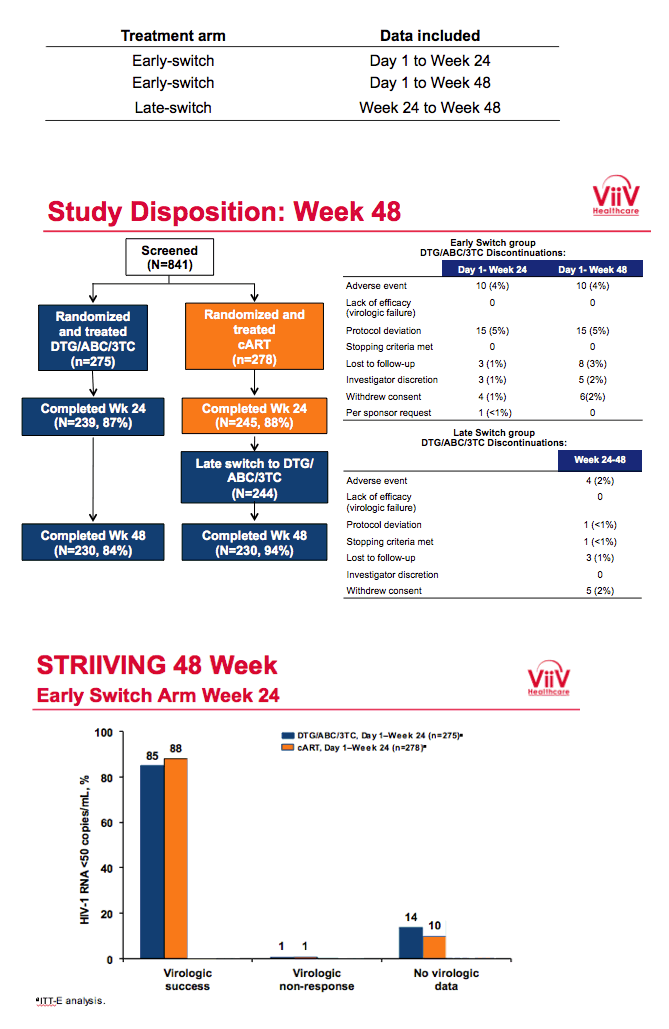
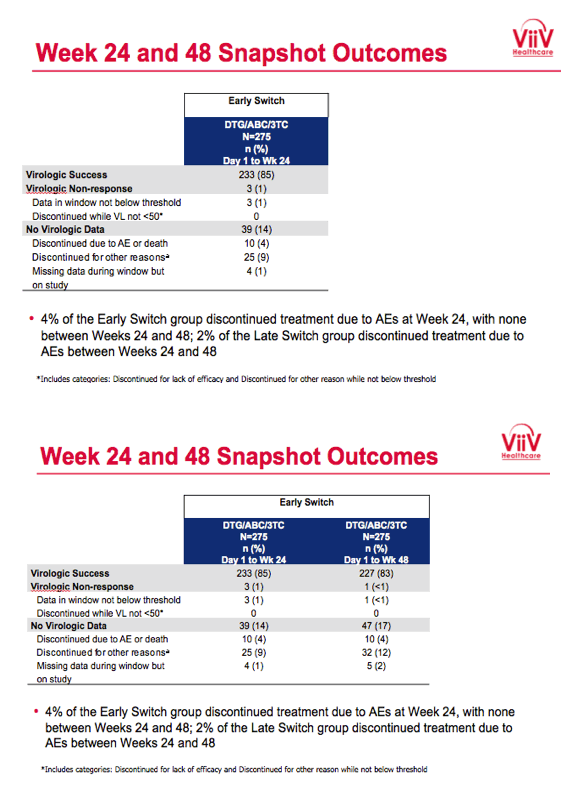
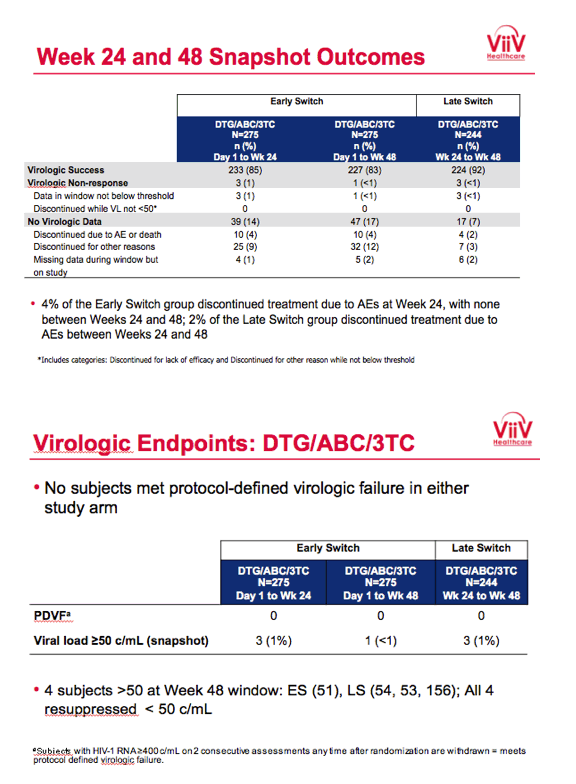
Baseline characteristics did not differ substantially between study arms. Of the 275 people randomized to receive DTG/ABC/3TC immediately, 239 (87%) completed week 24 and 230 (84%) completed week 48. Of the 278 people randomized to maintain their initial regimen, 245 (88%) completed week 24, of whom 244 switched to DTG/ABC/3TC and 230 (83% of 278) completed week 48. No one in either study arm dropped out because of lack of efficacy. Ten participants (4%) quit the early-switch DTG/ABC/3TC group because of an adverse event by week 24, and no more quit from week 24 to week 48 for that reason. Four people (2%) quit the late-switch group between weeks 24 and 48 because of an adverse event.
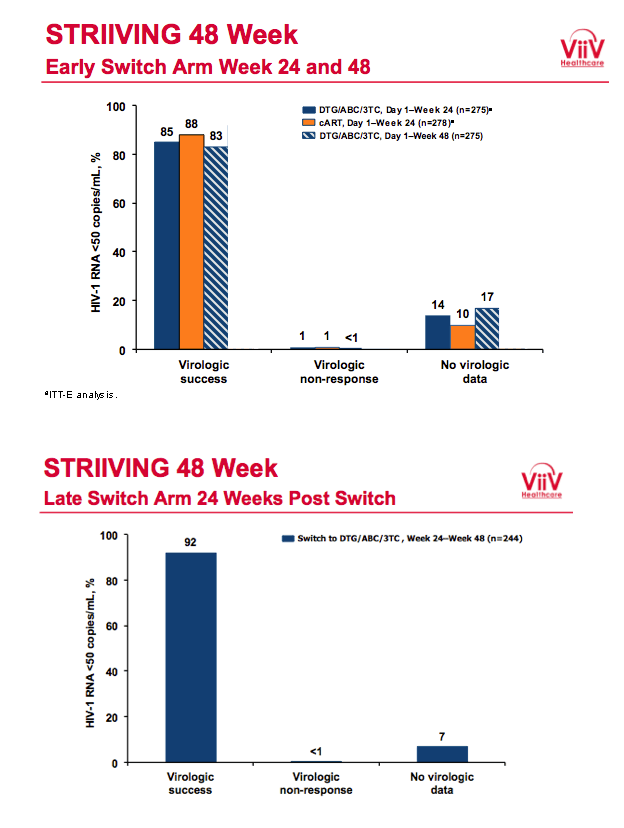
After 24 weeks 85% who switched to DTG/ABC/3TC and 88% who kept their initial regimen had a viral load below 50 copies in the snapshot analysis, a result establishing the noninferiority of switching to DTG/ABC/3TC. Among people taking DTG/ABC/3TC for the entire 48 weeks, 83% had a viral load below 50 copies at week 48. Among people who switched to DTG/ABC/3TC after 24 weeks, 92% had a viral load below 50 copies at week 48. No study participants in either arm had protocol-defined virologic failure, which meant a viral load above 400 copies on two consecutive assessments after randomization.
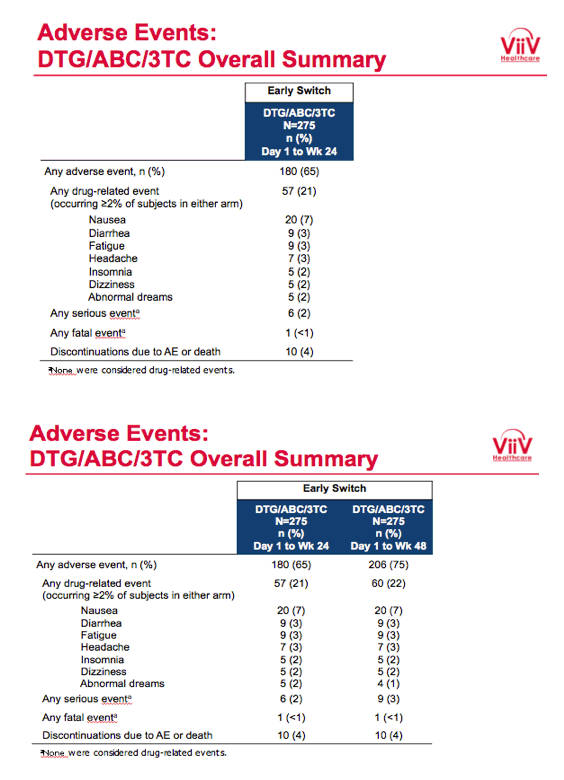
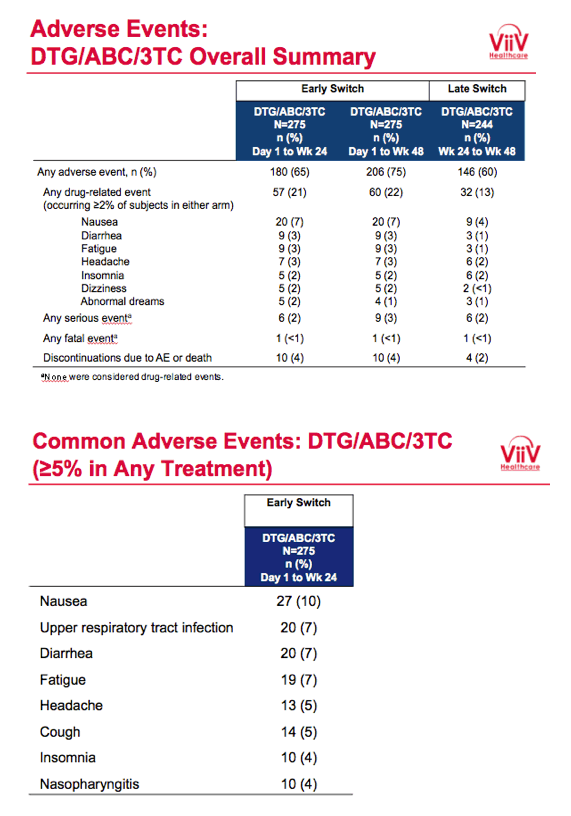
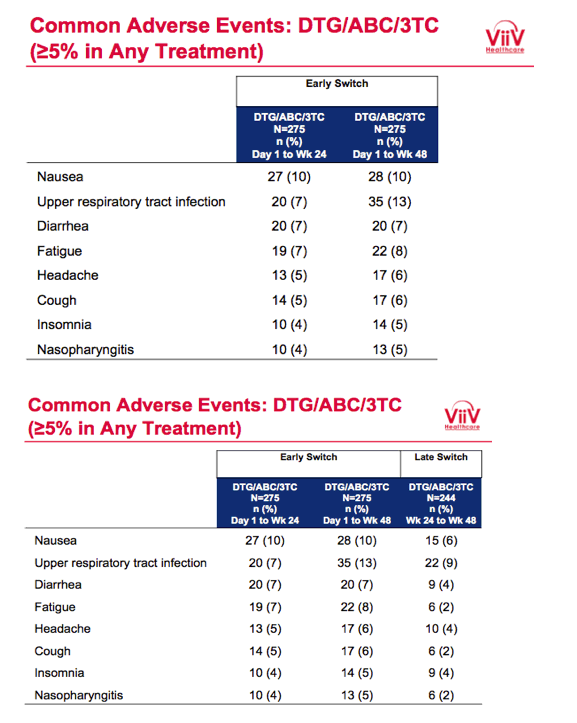
Proportions of participants with a drug-related adverse event in (1) the early-switch group between day 1 and week 24, (2) the early-switch group between day 1 and week 48, and (3) the late-switch group between week 24 and week 48 were 21%, 22%, and 13%. Respective rates of any serious event were 2%, 3%, and 2%, and respective rates of discontinuation due to an adverse event or death were 4%, 4%, and 2%. The most frequent adverse events were nausea, upper respiratory tract infection, diarrhea, and fatigue.
STRIIVING investigators believe these findings support switching from a stable suppressive regimen to once-daily coformulated DTG/ABC/3TC.
Reference
1. Lake JE, Trottier B, Garcia-Diaz J, et al. STRIIVING: switching to abacavir/dolutegravir/lamivudine fixed dose combination (ABC/DTG/3TC FDC) from a PI, INI or NNRTI based regimen maintains HIV suppression at week 48. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract THAB0203.
----------------------
Switching to Dolutegravir/Abacavir/Lamivudine Fixed Dose Combination
(DTG/ABC/3TC FDC) from a PI, INI or NNRTI Based Regimen
Maintains HIV Suppression at 48 Weeks
Reported by Jules Levin
Durban 2016 July 18-22
J Lake,1 B Trottier,2 J Garcia-Diaz,3 H Edelstein,4 P Kumar,5 UF Bredeek,6
M Loutfy,7 C Brennan,8 J Koteff,8 B Wynne,9 J Hopking,10 M Aboud11
1University of California, Los Angeles, CA, USA; 2Clinique Médicale l‘Actuel, Montreal, QC, Canada; 3Ochsner Clinic Foundation, New Orleans, LA, USA; 4Highland Hospital, Alameda Health System, Oakland, CA, USA; 5Georgetown University, Washington, DC, USA; 6Metropolis Medical, San Francisco, CA, USA; 7Maple Leaf Research, Toronto, Ontario, Canada; 8ViiV Healthcare, Research Triangle Park, NC, USA; 9ViiV Healthcare, Collegeville, PA, USA; 10GlaxoSmithKline, London, United Kingdom; 11ViiV Healthcare, London, United Kingdom


|
| |
|
 |
 |
|
|