 |
 |
 |
| |
Enhanced Infection Prophylaxis Cuts Mortality 25% in ART Starters With Low CD4s
|
| |
| |
21st International AIDS Conference (AIDS 2016), July 18-22, 2016, Durban, South Africa
Mark Mascolini
Multidrug infection prophylaxis in Africans starting antiretroviral therapy (ART) with very low CD4 counts reduced mortality about 25% compared with standard prophylaxis in the four-country REALITY trial [1]. The same study found that adding raltegravir to three-drug first-line ART did not cut mortality through 24 or 48 weeks of follow-up [2] (reported separately by NATAP).
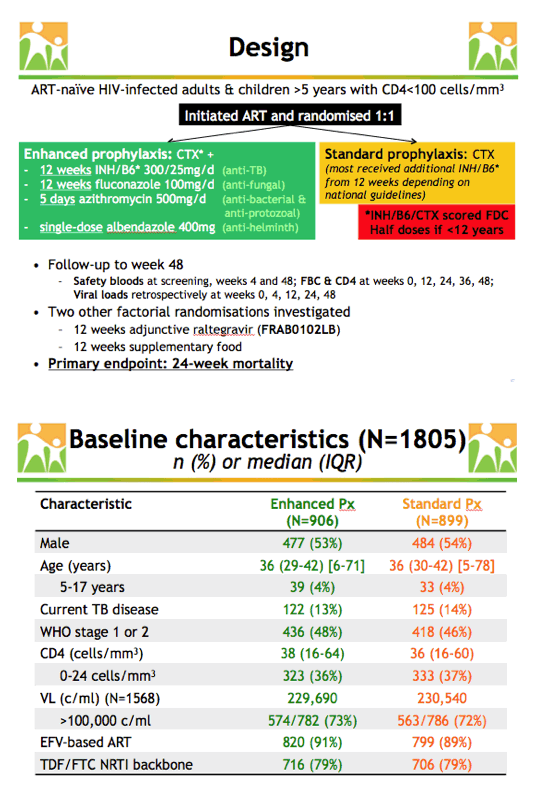
People starting ART with a low CD4 count face a high risk of death in the first 6 months of therapy, often from infections including tuberculosis, other bacterial infections, and fungal and protozoal infections. Guidelines for infection prophylaxis in people with advanced HIV infection vary from country to country in Africa but usually include cotrimoxazole (CTX), isoniazid, and little else. REALITY trial researchers planned this analysis to see whether an "enhanced package" of infection prophylaxis when ART began would lower mortality.
In Kenya, Malawi, Uganda, and Zimbabwe, REALITY enrolled 1733 adults and 72 children, all without antiretroviral experience, and randomized them 1-to-1 to standard prophylaxis (CTX and usually additional isoniazid/B6 depending on national guidelines) or to enhanced prophylaxis (CTX plus 12 weeks of anti-TB isoniazid/B6, 12 weeks of the antifungal fluconazole, 5 days of the antibacterial/antiprotozoal azithromycin, and a single dose of the antihelminth albendazole). Isoniazid, B6, and CTX were formulated into one pill. The primary endpoint was mortality at week 24.
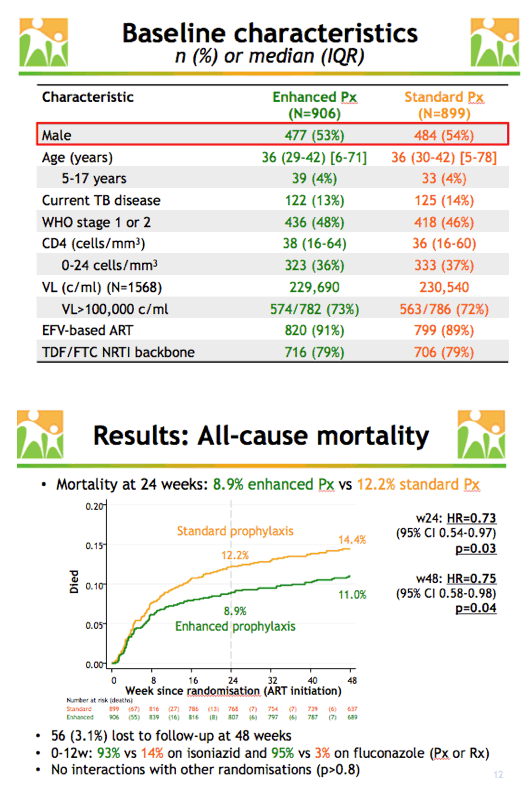
The enhanced and standard prophylaxis arms were similar in proportions of males (53% and 54%), current TB disease (13% and 14%), median pretreatment CD4 count (38 and 36), median viral load (229,690 and 230,540 copies), efavirenz-based ART (91% and 89%), and use of tenofovir/emtricitabine (79% and 79%). Median age was 36 in both the enhanced and standard groups, and 4% in each group were 5 to 17 years old.
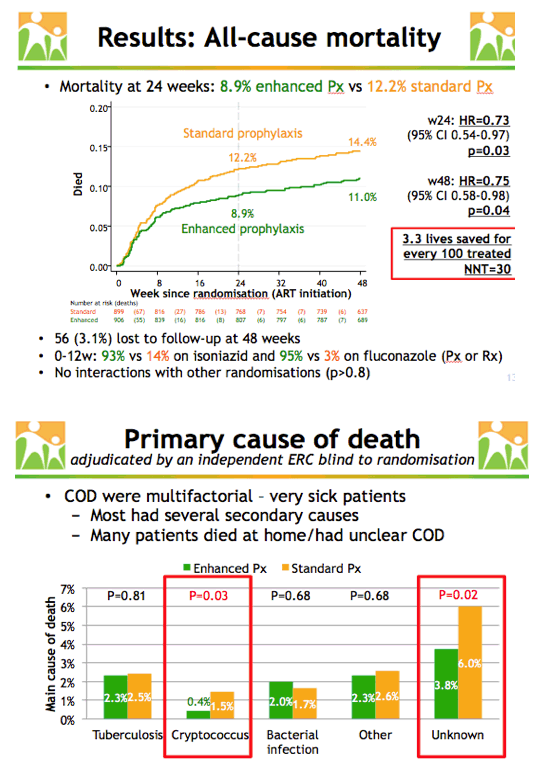
After 24 weeks 8.9% in the enhanced prophylaxis group and 12.2% in the standard group died to yield a hazard ratio (HR) indicating a 27% relative reduction in mortality in the enhanced group (HR 0.73, 95% confidence interval [CI] 0.54 to 0.97, P = 0.03). The absolute reduction in mortality at 24 weeks was 3.3%. After 48 weeks 11.0% in the enhanced arm and 14.4% in the standard arm had died, rates indicating a 25% lower relative death risk with enhanced prophylaxis (HR 0.75, 95% CI 0.58 to 0.98, P = 0.04). The researchers calculated 3.3 lives would be saved for every 100 people who received enhanced prophylaxis.
Enhanced prophylaxis resulted in significantly lower mortality from cryptococcal infection (0.4% versus 1.5%, P = 0.03) and from unknown causes (3.8% versus 6.0%, P = 0.02). Most deaths had several causes, and many people died at home from uncertain causes.
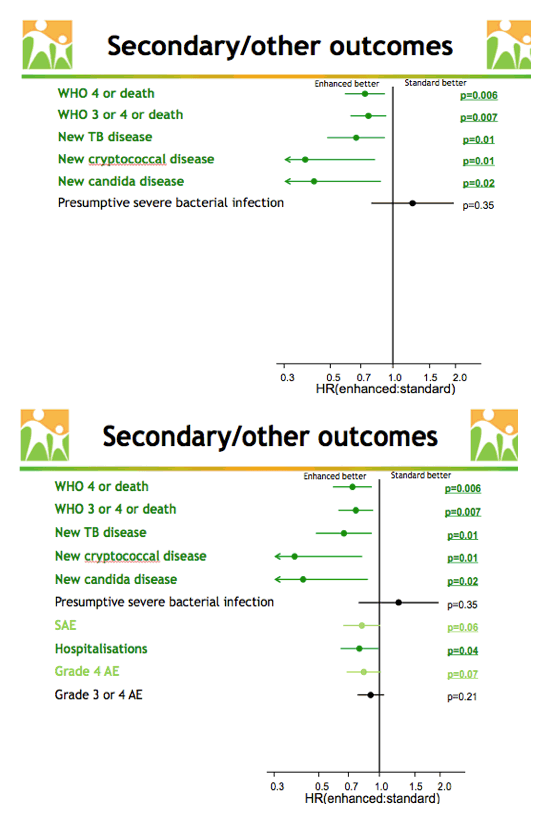
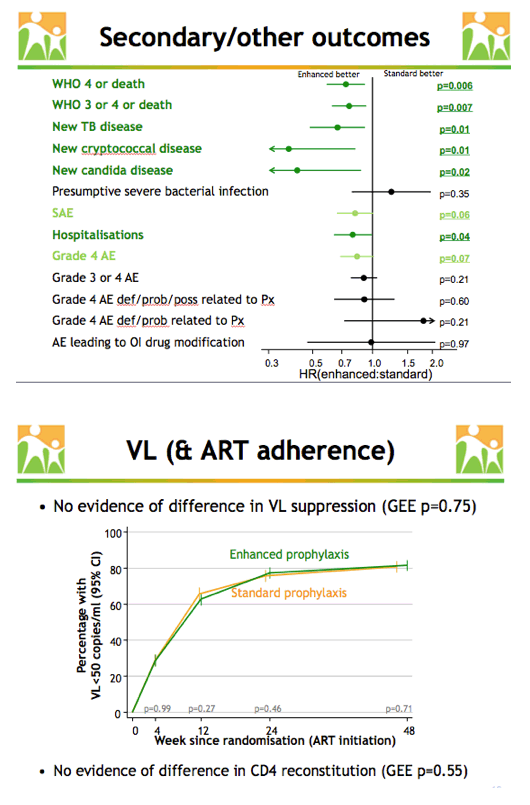
Further analysis determined that enhanced prophylaxis was associated with lower rates of World Health Organization (WHO) stage 4 disease or death (P = 0.006), WHO stage 3 or 4 disease or death (P = 0.007), new TB disease (P = 0.01), new cryptococcal disease (P = 0.01), new candida disease (P = 0.02), and hospital admissions (P = 0.04). Enhanced prophylaxis was marginally linked to fewer serious adverse events (P = 0.06) and fewer grade 4 adverse events (P = 0.07).
Generalized estimating equation analysis indicated that enhanced prophylaxis had no impact on HIV load when compared with standard prophylaxis, and no impact on CD4 gains. Combining drug costs in the four participating countries, the researchers determined that the 12-week enhanced package would cost a little over $30 in Zimbabwe and Malawi, about $10 in Uganda, and about $7 in Kenya.
The REALITY team urged policy makers to "consider adopting and implementing this low-cost broad infection prevention package which would save 3.3 lives for every 100 individuals treated."
References
1. Hakim J, Musiime V, Szubert AJ, et al. Enhanced infection prophylaxis reduces mortality in severely immunosuppressed HIV-infected adults and older children initiating antiretroviral therapy in Kenya, Malawi, Uganda and Zimbabwe: the REALITY trial. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract FRAB0101LB.
2. Kityo C, Siika A, Szubert AJ, et al. 12-week raltegravir-intensified quadruple therapy versus triple first-line ART reduces viral load more rapidly but does not reduce mortality in severely immunosuppressed African HIV-infected adults and older children: the REALITY trial. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract FRAB0102LB.



|
| |
|
 |
 |
|
|