 |
 |
 |
| |
CD8-Cell Exhaustions Predicts Primary HIV Infection Progression -
HIV Causes CD8 Exhaustion-Occurs Early/Predicts HIV Progression
|
| |
| |
[from Jules: suggesting starting ART immediately after infection is the best & might prevent or limit immune exhaustion & depletion; or the earliest start of ART as possible]
21st International AIDS Conference (AIDS 2016), July 18-22, 2016, Durban, South Africa
Mark Mascolini
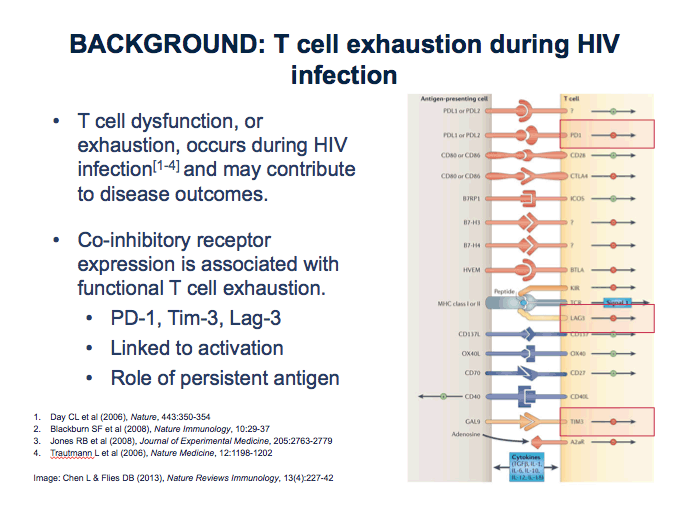
Expression of three T-cell exhaustion markers predicted clinical progression of primary HIV infection in a 138-person analysis [1]. Researchers at the University of Oxford and colleagues found that expression of all three markers on CD8 cells correlated positively with viral load.
Prior research suggested that T-cell dysfunction, or exhaustion, during HIV infection may contribute to clinical outcomes [2-4]. These studies and others tied expression of three markers--PD-1, Tim-3, and Lag-3--to functional T-cell exhaustion, to the size of the HIV reservoir [5], and to HIV rebound after antiretroviral therapy (ART) stops. The Oxford team conducted this study to determine whether T-cell exhaustion during early HIV infection affects disease outcomes.
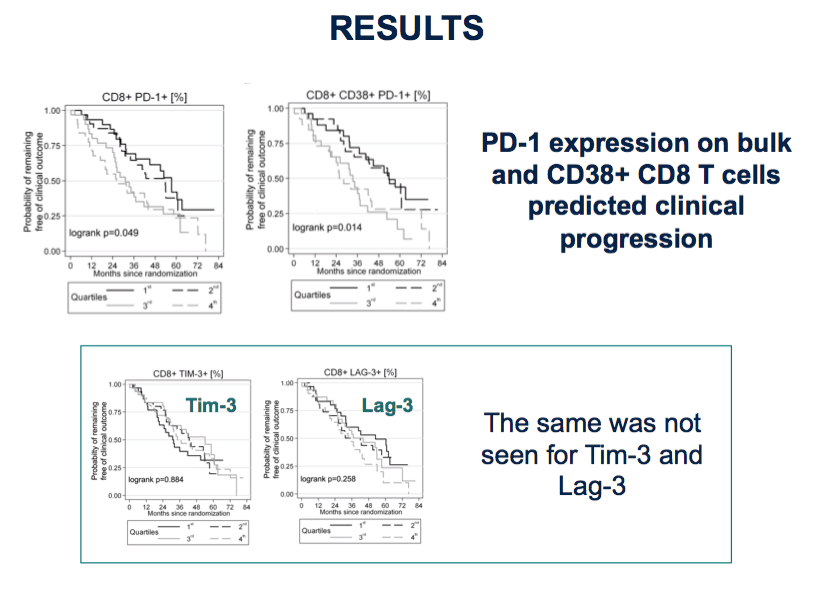
The analysis began with measurement of PD-1, Lag-3, and Tim-3 expression on CD8 cells in 122 UK participants in the SPARTAC study, a randomized trial of short-course ART in primary HIV infection [6]. For the exhaustion analysis, researchers defined progression as a CD4 count below 350 or starting long-term ART. PD-1 expression on CD38 CD8 cells did predict progression, but expression of Tim-3 or Lag-3 did not.
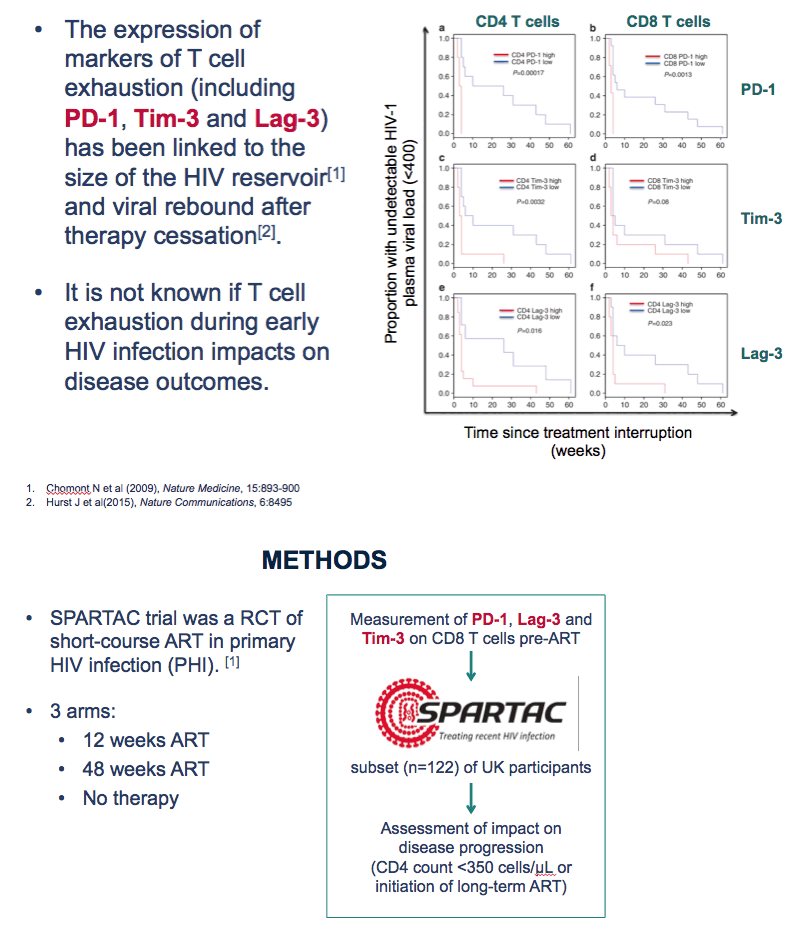
The impact of PD-1 on progression proved more evident in SPARTAC participants sampled within 12 weeks of HIV seroconversion than in those sampled later. Expression of all three markers on bulk CD8 cells and CD38 CD8 cells correlated positively with viral load. For example, for PD-1 on bulk CD8s rho = 0.41 (P < 0.001), and for PD-1 on CD38 CD8s rho = 0.50 (P < 0.001).
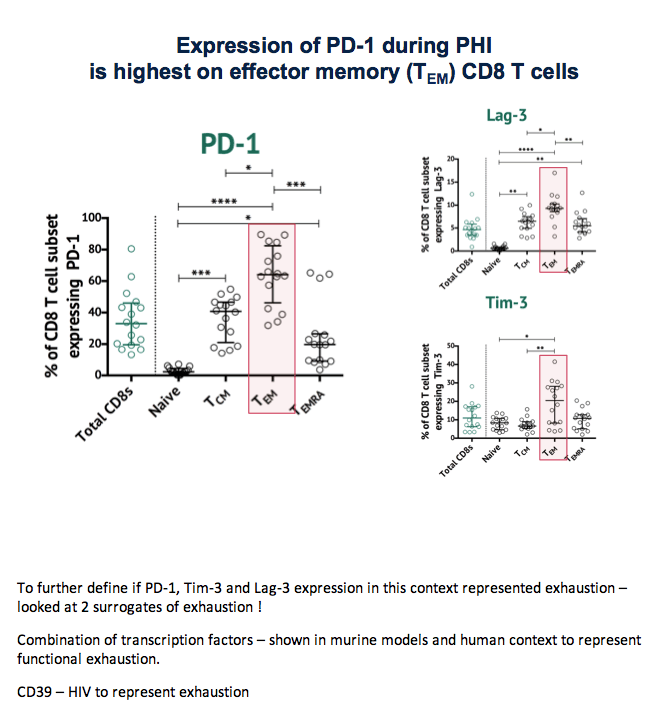
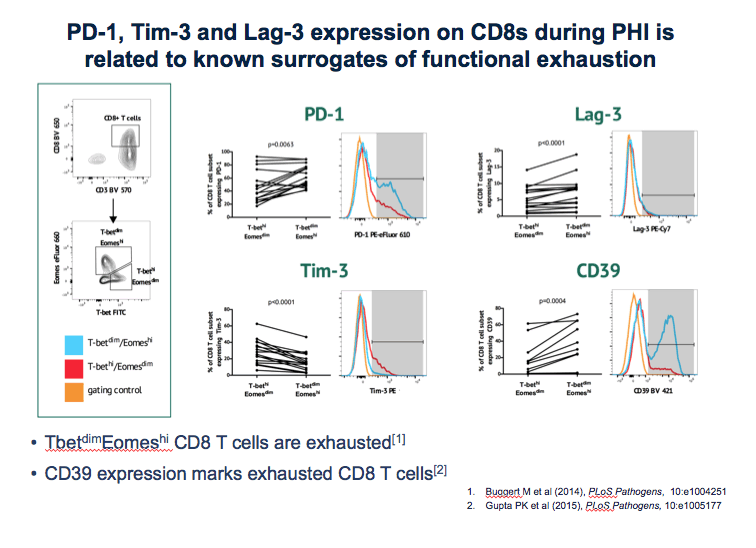
The second part of the CD8-cell exhaustion analysis involved 16 participants in HEATHER, an observational cohort of people who started ART during primary infection. Expression of PD-1, Lag-3, and Tim-3 during primary HIV infection proved significantly higher on effector memory CD8 cells than on other CD8-cell subsets. Expression of the three markers on CD8s during primary infection was associated with known surrogates of T-cell exhaustion, such as CD39 expression.
The researchers proposed that "viremia during early HIV infection may drive exhaustion of effector CD8 T cells, impacting upon subsequent disease outcomes."
References
1. Martin GE, Pantazis N, Hoffmann M, et al. Exhaustion of activated CD8 T cells predicts disease progression in primary HIV-1 infection. 21st International AIDS Conference (AIDS 2016). July 18-22, 2016. Durban, South Africa. Abstract WEAA0105LB.
2. Day CL, Kaufmann DE, Kiepiela P, et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature. 2006;443:350-354.
3. Jones RB, Ndhlovu LC, Barbour JD, et al. Tim-3 expression defines a novel population of dysfunctional T cells with highly elevated frequencies in progressive HIV-1 infection. J Exp Med. 2008;205:2763-2779.
4. Trautmann L, Janbazian L, Chomont N, et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med. 2006;12:1198-1202.
5. Chomont N, El-Far M, Ancuta P, et al. HIV reservoir size and persistence are driven by T cell survival and homeostatic proliferation. Nat Med. 2009;15:893-900.
6. SPARTAC Trial Investigators, Fidler S, Porter K, Ewings F, et al. Short-course antiretroviral therapy in primary HIV infection. N Engl J Med. 2013;368:207-217.
http://www.nejm.org/doi/full/10.1056/NEJMoa1110039
--------------

--------------
abstract
Background: The rate at which HIV-1 infected individuals progress to AIDS is highly variable and impacted by T cell immunity. CD8 T cell inhibitory molecules are upregulated in HIV-1 infection and associate with T cell dysfunction. Here we aimed to determine whether CD8 T cell immune checkpoint markers PD-1, Lag-3 and Tim-3 are associated with immune activation and disease progression.
Methods: We evaluated participants (n=122) with primary HIV infection (PHI) randomised to receive short-course ART, or no therapy, in the SPARTAC trial. Expression of PD-1, Tim-3, Lag-3 and CD38 on CD8 T cells from the closest pre-therapy time-point to seroconversion was measured by flow cytometry, and correlated with surrogate markers of HIV disease (HIV-1 plasma viral load (pVL) and CD4 T cell count) and the trial endpoint (time to CD4 count < 350 cells/μl or initiation of long-term antiretroviral therapy). To explore the functional significance of these markers, co-expression of Eomes, T-bet and CD39 was assessed.
Results: Expression of PD-1 on bulk and CD38 CD8 T cells correlated with pVL and CD4 count, and predicted the trial endpoint. Lag-3 expression was associated with pVL but not CD4 count. For all exhaustion markers, expression on CD38 CD8 T cells increased the strength of the associations. In Cox models, progression to the trial endpoint was most marked for PD-1/CD38 co-expressing cells, with evidence for a stronger effect within 12 weeks from seroconversion. The effect of PD-1 and Lag-3 expression on CD8 T cells retained statistical significance in Cox proportional hazards models including antiretroviral therapy and CD4 count, but not pVL as co-variants (HR 1.76; p=0.047 and HR 1.46; p=0.024 respectively). In a cohort of similar individuals with untreated PHI we demonstrated strong associations of PD-1 and Lag-3 with the T-betdim/Eomeshi CD8 population and CD39 expression, suggesting the expression of these markers during PHI represents functional exhaustion.
Conclusions: Expression of ''exhaustion'' or ''immune checkpoint'' markers in early HIV infection is associated with clinical progression, and may be impacted by immune activation and timing of therapy. New markers to identify exhausted T cells and novel interventions to reverse exhaustion may inform the development of new immunotherapeutic approaches.

We know that - the rate at which HIV-infected individuals develop AIDS is highly variable - T cell immunity plays an important role.
T cell dysfunction, or exhaustion, is a hallmark of chronic viral infections including HIV and may contribute to disease outcomes. from [Jules: which is why HCV coinfection increases immune depletion]
These plots show the relationship between time and clinical progression (remind) in SPARTAC participants.
Focus intially on left hand panel
⋅Participants have been split into quartiles based on PD-1 expression levels.
⋅Dose- effect. More PD-1 is faster disease progression
Right hand panel shows this on CD38+ CD8 T cells, this is also seen.
The same was not seen for TIM-3 and Lag-3
This figure from the original SPARTAC analysis shows the progression to endpoint base on time from seroconversion to randomisation.
Demonstrates an intriguing effect of time from seroconversion to randomisation on disease progression - earlier randomisation progressed faster.
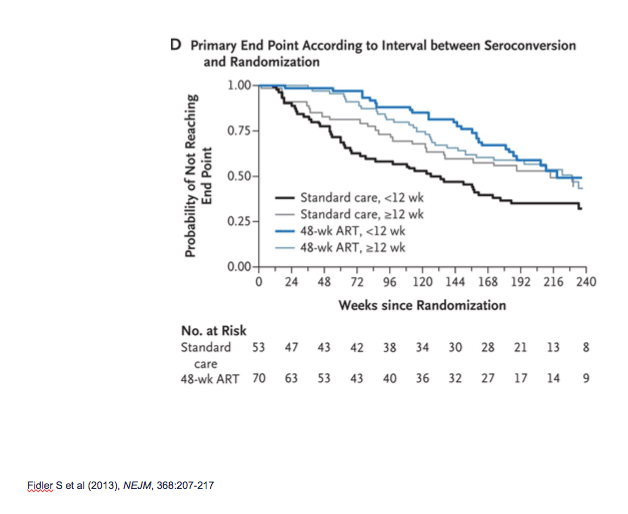
Because of this finding - I will show you this same analysis stratified by time from seroconversion.
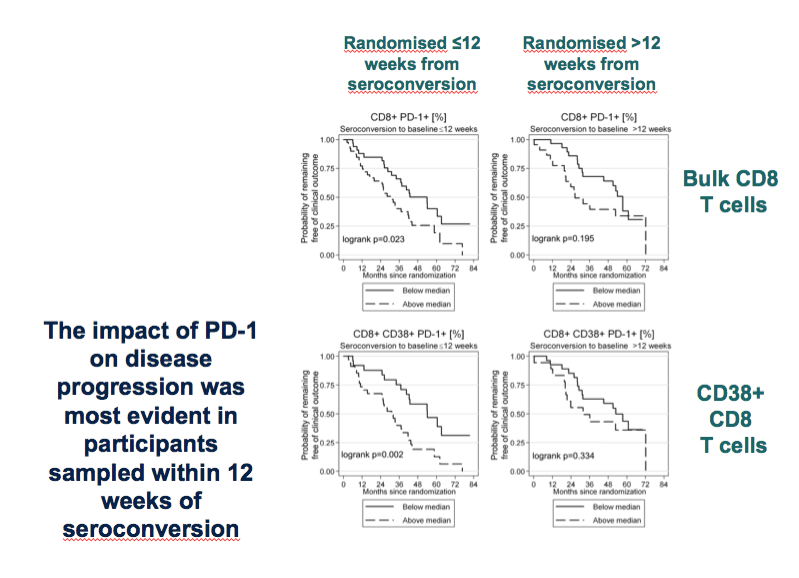
We looked at the relationship between PD-1 expression and clinical progression in individuals who who were within 12 weeks and >12 weeks of seroconversion.
We looked at this on bulk (top panels) or CD38+ (bottom panels) CD8 T cells.
These plots are split at the median, rather than on quartiles with the solid line showing individuals with low PD-1 levels
When we split the analysis this way, we see the ability to predict disease progression is only seen when PD-1 is measured <12 weeks of seroconversion (left panels).
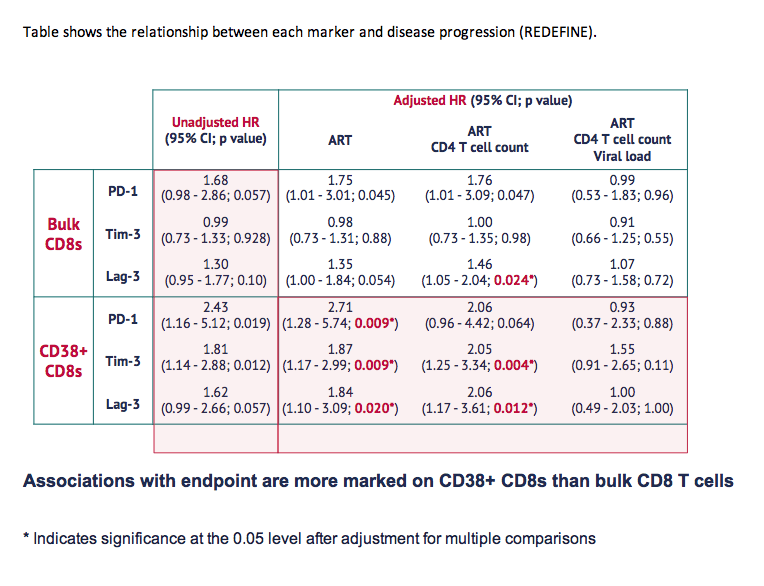
Table shows the relationship between each marker and disease progression (REDEFINE).
Associations with endpoint are more marked on CD38+ CD8s than bulk CD8 T cells
* Indicates significance at the 0.05 level after adjustment for multiple comparisons
To us, this observation raised the question of whether this represents exhaustion or just activation.
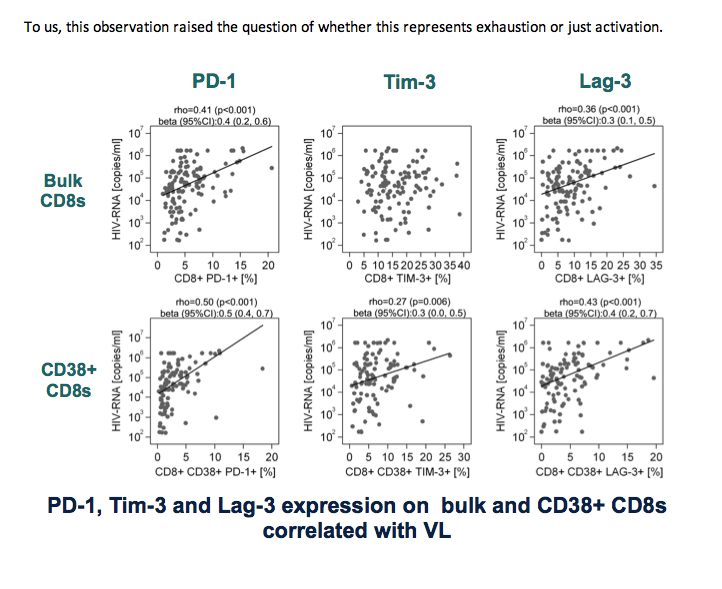
One of our concerns was that this finding could be all due to activation, no exhaustion.
So, sampling limitations - another primary infection cohort.
The way we answered this question was to turn to a different primary infection cohort.
Introduction to HEATHER

To further define if PD-1, Tim-3 and Lag-3 expression in this context represented exhaustion - looked at 2 surrogates of exhaustion !
Combination of transcription factors - shown in murine models and human context to represent functional exhaustion.
CD39 - HIV to represent exhaustion

|
| |
|
 |
 |
|
|