 |
 |
 |
| |
Primary Efficacy Endpoint and Safety Results of Ibalizumab in a Phase 3 Study of Heavily Treatment-Experienced Patients with Multi-Drug Resistant HIV-1 Infection
|
| |
| |
Reported by Jules Levin
IDWeek 2016 Oct 23-26 New Orleans
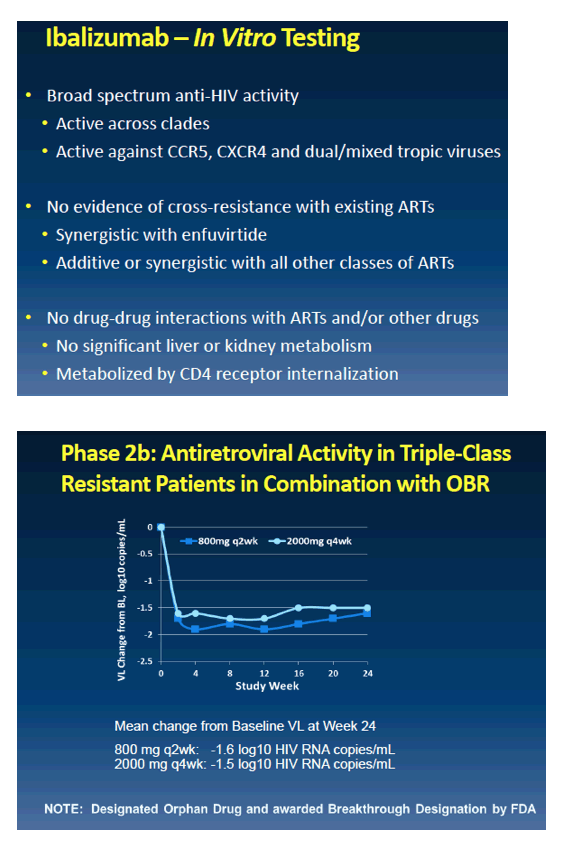
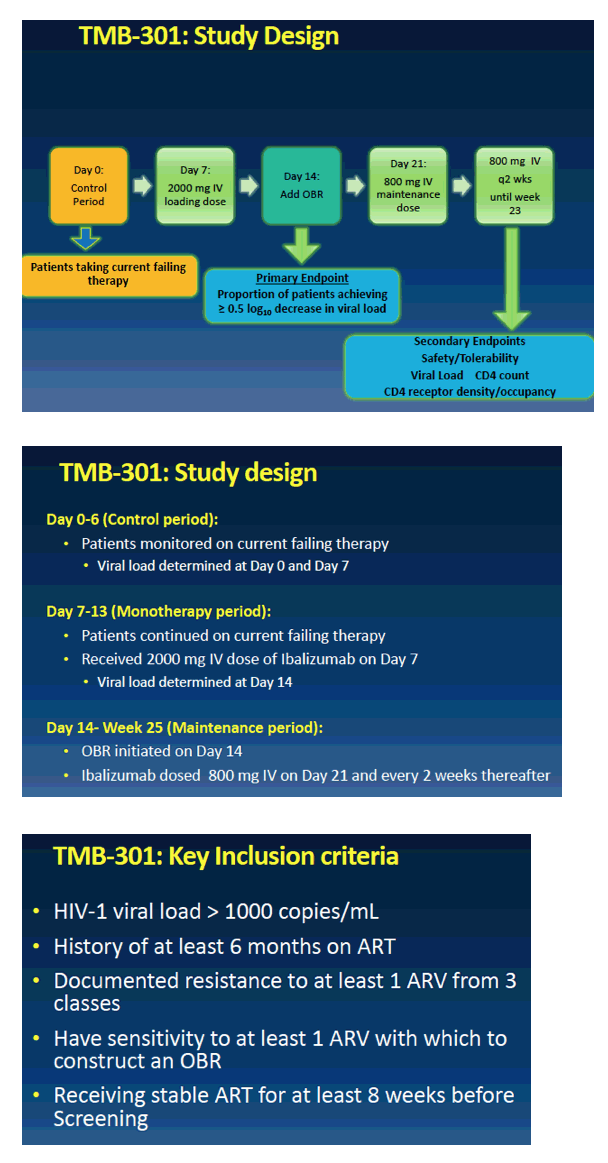
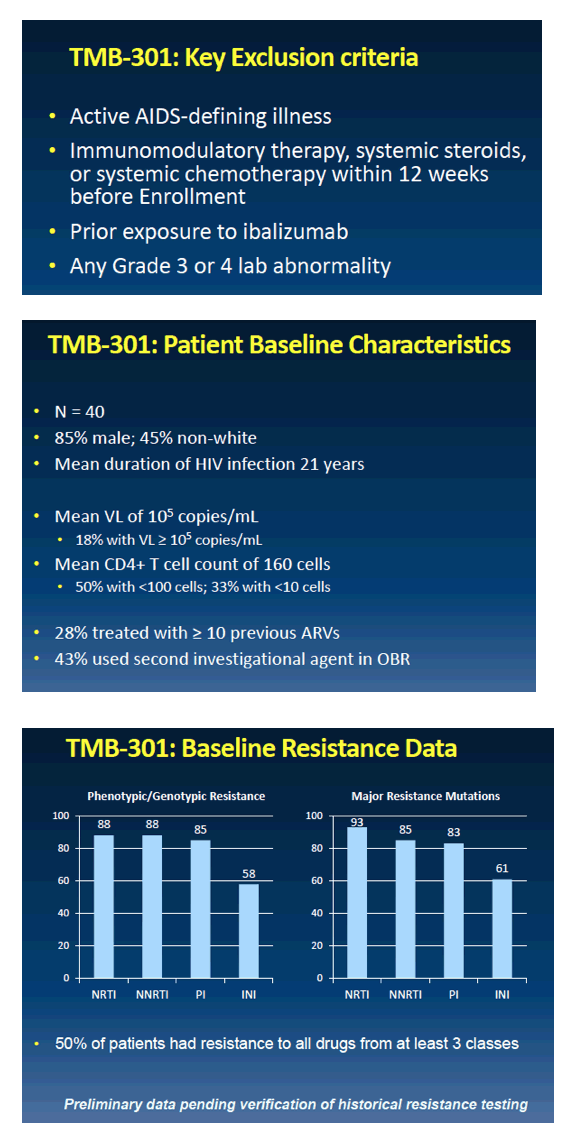
from Jules: the FDA primary endpoint is day 7 after adding this new drug to failing therapy patients were taking, and then patients afterwards can optimize therapy. So here is presented day 7 data & week 24 data will be reported later & submitted to FDA in 1st quarter 2017 with expected FDA approval in the 2nd half of 2017. The drug is infused every 2 weeks so arrangements will be planned on how to provide regular access for patients, company will develop a program, but a patient will be able to receive infusion - the loading dose is 30 minutes & the regular infusion every 2 weeks thereafter is 15 minutes its a 15 minute infusion - in your doctor's office. There are about 25,000 multi drug resistant patients in the USA for whom this therapy might be helpful & 10,000 who are in serious condition and appear to need the drug. As we know there are a number of long acting HIV drugs in development like Cabotegravir. Ibalizumab is the first long acting drug but as others come along we could develop HIV regimens that are completely administered non-orally, by injection and/or infusion, that could be administered in a medical office with visits spaced out over time, without having to take pills perhaps.
Late Breaker Oral presented this morning
Jacob Lalezari, MD Medical Director
Quest Clinical Research, a division of eStudySite


|
| |
|
 |
 |
|
|