 |
 |
 |
| |
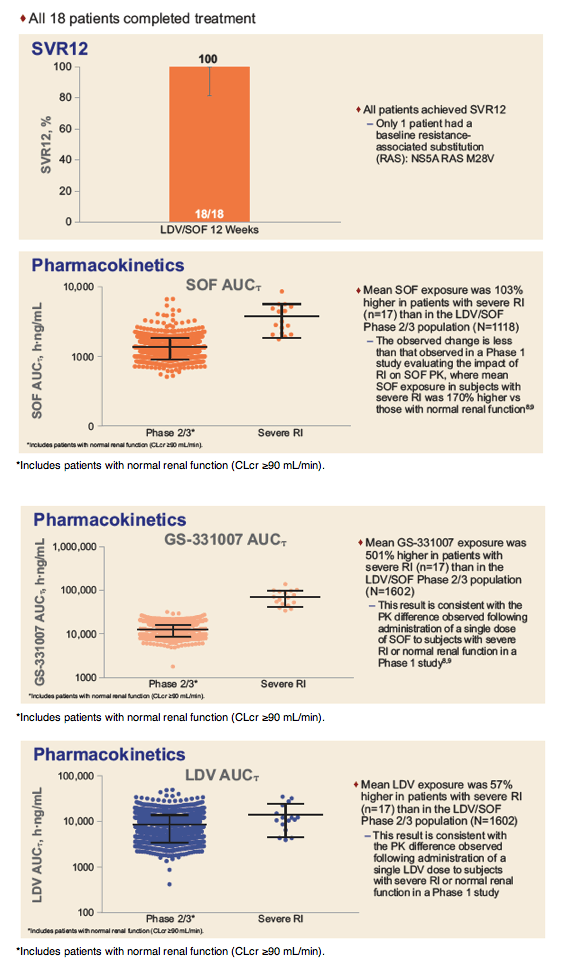
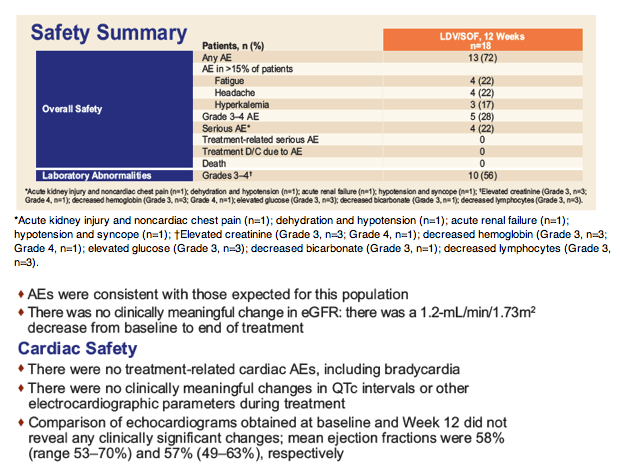
Safety and Efficacy of Treatment With Once-Daily Ledipasvir/Sofosbuvir (90/400 mg) for 12 Weeks in Genotype 1 HCV-Infected Patients With Severe Renal Impairment
|
| |
| |
Reported by Jules Levin
AASLD: The Liver Meeting® 2017, October 20-24, 2017, Washington, DC
Eric Lawitz,1 Charles S. Landis,2 Benedict J. Maliakkal,3 Maurizio Bonacini,4 Grisell Ortiz-Lasanta,5 Jie Zhang,6 Erik Mogalian,6
Shampa De-Oertel,6 Anu O. Osinusi,6 Diana M. Brainard,6 John G. McHutchison,6 Steven L. Flamm,7 Stuart C. Gordon,8 Edward J. Gane9
1UT Health San Antonio, Texas Liver Institute, San Antonio, TX; 2UW Medical Center, Seattle, WA; 3The University of Tennessee, Memphis; 4Mission Gastroenterology & Hepatology, University of California, San Francisco;
5Fundación de Investigación, San Juan, PR; 6Gilead Sciences, Inc., Foster City, CA; 7Northwestern University, Chicago, IL; 8Henry Ford Health System, Detroit, MI; 9Auckland Clinical Studies Ltd, Auckland, New Zealand

References
1. Beinhardt S, et al. Transpl Int 2016;29:999-1007; 2. Bhamidimarri KR, et al. J Hepatol 2015;63:763-5; 3. Dumortier J, et al. Nephrol Dial Transplant 2016 Oct 19 [Epub]; 4. Hundemer GL, et al. Infect Dis 2015;47:924-9; 5. Nazario HE, et al. J Hepatol
2017;66:S507; 6. Saxena V, et al. Liver Int 2016;36:807-16; 7. Singer AW, et al AASLD 2016, poster 804; 8. Cornprobst MT, et al. J Hepatol 2012;56:S389-S548, poster 1101; 9. Mogalian E, et al. J Hepatol 2014;60(suppl):1145A-6A (abstr 1952).
|
| |
|
 |
 |
|
|