 |
 |
 |
| |
Summary from AASLD 2017 for Hepatitis C
HCV: Game over?
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
Department of Medicine I, University Hospital Bonn, Bonn, Germany
Edited by Jules Levin
Introduction
The AASLD Liver Meeting was held in October from 20-24th, 2017 in Washington DC, USA. Many regular AASLD attendees will still remember the completely packed late breaker sessions from recent years, full of phase II and III new direct acting antiviral (DAA) combination trials in varying patient populations. At this year AASLD Liver Meeting, no new DAA combination study results were presented in these sessions but rather were replaced by new drug trials for treatment of NASH or primary sclerosing cholangitis. Indeed, the only two new dual and triple DAA combinations, which were presented at the meeting, were accompanied by press releases from the respective companies Merck and Janssen, which announced the termination of their DAA development programs (1-2). Nevertheless, important new findings from the HCV research space were reported including issues around HCV screening, how to improve linkage to care, and treatment results from many different patient populations and real-life cohorts as well as post HCV cure monitoring. Reassuringly, the high success rates of all oral DAA therapy was further confirmed from a wealth of data particularly from somewhat more challenging HCV patient populations including: PWIDs, patients with concomitant advanced kidney disease, patients before and after kidney or liver transplantation, HCV genotype 3 infection and patients with unfavorable HCV disease characteristics, including compensated and decompensated cirrhosis (3-11). However, there was also data on shorter treatment durations for patients with more favorable HCV disease characteristics such as for non-cirrhotic GT1b patients (12). In addition, there was also new data around retreatment of previous DAA failures (with documented resistance development) with new DAA combinations (13-14). In the area of HIV-coinfection, clearly outcomes of the ACTG trial on treatment of acute hepatitis C in HIV-seropositive individuals for eight weeks with ledipasvir/sofosbuvir (LDV/SOF) were also new and noteworthy (15). Of interest were also reports on the increases in acute HCV infections among HIV-seropositive men who have sex with men (MSM) and the high risk of reinfection in particular patient populations (16-17). Further data was also presented on the question whether there is an increased risk for hepatocellular carcinoma in DAA treated patients from large real-life cohorts and what impact SVR has on clinical endpoints under continued follow-up (18-22).
The following AASLD summary report tries to capture the major new findings and results presented around the topics raised above, as well as to outline some of the status reports of HCV treatment implementation on a global level. Clearly, the successes of modern HCV DAA combination therapy still have not reached all in need for these treatments.
Treatment of acute HCV infection
The optimal duration for the treatment of acute HCV infection is currently unknown. The AASLD/IDSA HCV Treatment Guidance Panel recommends treating acute HCV like chronic infection, while the EASL Guideline Panel recommends 8-12 weeks of LDV/SOF. A previous study in HIV-seropositive individuals with 6 weeks of LD/SOF demonstrated that relapses might occur after treatment duration shortening in particular in patients with high baseline HCV viral loads (23). Cohort-1 of the SWIFT-C investigated sofosbuvir plus ribavirin for treatment of acute HCV for 12 weeks but again showed a high relapse rate (24). Cohort 2 of the SWIFT-C study is a multicenter, single arm trial of the AIDS Clinical Trials Group investigating the safety and efficacy of 8 weeks of LDV/SOF for the treatment of acute HCV infection in HIV-infected participants (15). Acute HCV was defined as the first 24 weeks of HCV infection after diagnosis, with enrollment in the first 12-24 weeks. Criteria for diagnosis were HCV antibody or RNA seroconversion in the preceding 6 months or new liver enzyme elevation (>5X ULN with documented normal baseline ALT or >10X ULN without baseline ALT) and detectable HCV RNA in those without recent HCV testing. Genotypes 1 and 4 were allowed to be enrolled into the study and other liver diseases were excluded (e.g. active hepatitis B). The sample size for the cohort was determined by non-inferiority comparison to the study-defined historical sustained virologic response (SVR) rate of 60%, achieved with 24-48 weeks of pegylated interferon and weight based ribavirin. The primary endpoint was SVR12, defined as HCV RNA target not detected at 12-weeks post-treatment. Overall, 27 subjects were enrolled. The study design is shown below in figure 1.
Figure 1: Study design of the SWIFT-C study
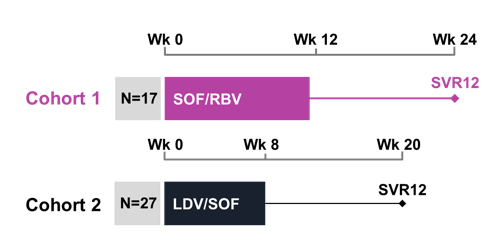
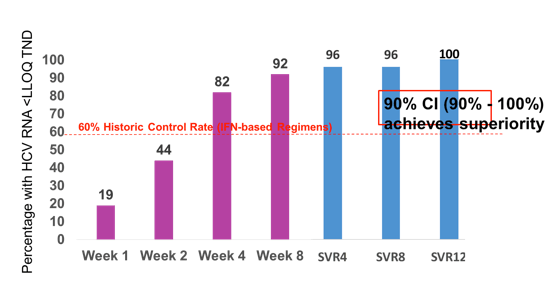
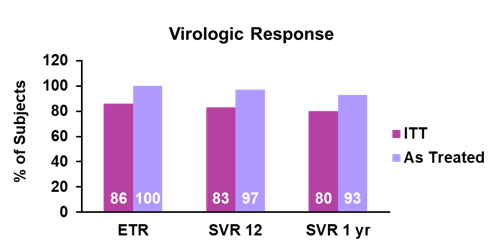
Of the 27 participants enrolled, all were male, median age was 46 years (IQR: 38 - 50), 9 (33%) were Hispanic, and 22(81%) reported having never used IV drugs. The distribution of HCV genotype was: 26 participants (96%) genotype 1 and 1 (4%) genotype 4. Median baseline HCV RNA was 1,490,000 IU/mL (IQR: 32,500 - 3,560,000 IU/mL), with a high baseline RNA of >6 million IU/mL in 6 (22%) participants. Median baseline CD4+ T cell count was 561 cells/mm3. All participants were on antiretrovirals with HIV-1 RNA < LLOQ, of which 10 were on a ritonavir- or cobicistat-boosted regimen with tenofovir disoproxil fumarate (TDF). All 27 participants took 8 weeks of study treatment and all (100%) achieved SVR12. The 90% CI was 90% to 100% showing superiority relative to the historical SVR of 60%. The on-treatment and SVR response rates are summarized in figure 2.
Figure 2: On treatment and SVR rates
Looking at safety outcomes, ten (37%) participants had a Grade 2 or higher adverse event (AE); no nephrotoxicity AEs were reported although a significant number of patients were on a boosted PI and TDF, which comes with higher TDF exposure due to drug-drug interactions.
In summary, the authors concluded that an 8-week course of LDV/SOF achieved 100% SVR12 in this trial of HIV-infected men with acute genotype 1 or 4 HCV infection, regardless of baseline HCV RNA. No renal toxicity was noted in participants on boosted TDF-inclusive HIV regimens. These data support an 8-week duration of LDV/SOF in the treatment of acute HCV infection in HIV-infected persons.
A5327 Sofosbuvir-Containing Regimens Without Interferon for Treatment of Acute HCV in HIV-1-infected Individuals (SWIFT-C) - (10/24/17)
Acute HCV infections on the rise and the risk of HCV reinfections
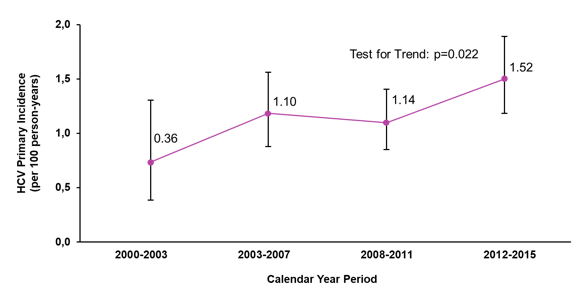
Treatment of acute HCV appears to lead to impressive high SVR rates but in the absence of protective immunity, any patient who is cured of HCV remains at risk for HCV reinfection in particular if ongoing risk behavior for HCV transmission continues. At this year AASLD, various reports addressed the issues of HCV reinfection in different patient populations. The first presentation was from San Diego, which predominantly aimed at determining the HCV primary incidence (seroconversion) among HIV+ MSM in San Diego but also evaluated the HCV reinfection incidence following successful HCV treatment (SVR) among HIV+ MSM again from San Diego (16). The significant increase in acute HCV infections in the studied MSM patient population over the observation period is captured in figure 3. Overall, 3 reinfections were observed during 102.5 person-years of follow-up leading to a reinfection incidence of 2.89/100 person year (95%CI 0.6-8.44). Reinfections occurred at 2.5, 2.7, and 5.9 years post SVR respectively, and because of the long duration between SVR and new infection were considered unlikely to be relapses.
Figure 3: Increasing HCV primary incidence among HIV+ MSM in San Diego
INCIDENCE OF HEPATITIS C AMONG HIV-INFECTED MEN WHO HAVE SEX WITH MEN IN SAN DIEGO, 2000-2015 - (11/10/17)
EACS: High incidence of HCV reinfection in HIV-positive MSM in the DAA era - (10/30/17)
In comparison to reinfection rates from Europe in particular London and Berlin however, the reinfection rates in San Diego so far are considerably lower. Work was presented from Natasha Martin and Patrick Ingiliz which utilized epidemic modeling to assess the impact of scaling-up HCV treatment on HCV incidence, as well as what levels of treatment and behavioral risk reduction are required for HCV elimination among HIV+ MSM in Berlin, Germany (25). The model was calibrated to the Berlin epidemic (rising HCV incidence from 0.56 per 100 person-years (/100py) in 1996-1999 to 2.76/100py in 2008-2012; 9.2% HCV seroprevalence, 8.2/100py reinfection rate 2002-2014). Based on Berlin data, we incorporated high rates of HCV testing (twice/year) and treatment (80% initiated within 6 months of diagnosis from 2002) were incorperated into the model. IFN-based SVR rates were assumed to be around 70%/30% for acute/chronic infection, respectively, and 95% SVR with DAAs from 2015 onwards. Finally, HCV incidence and chronic prevalence among HIV+ MSM in Berlin was modelled until 2030 under the following scenarios: 1) no historical treatment, 2) no change in treatment rates from 2016, 3) scale-up to 100% treated within 6 months of diagnosis and 25%/year of previously diagnosed and untreated from 2016. In addition, level of treatment and risk reduction among the HIV+ MSM population was determined which is required to achieve the World Health Organization elimination target of 90% reduction in HCV incidence by 2030. The corresponding modelling results are depicted below in figure 4. Modeling indicated an estimated HCV incidence of 2.5 per 100 person-years (/100py) among HIV+ MSM in Berlin in 2016, which could have been 3.8/100py in 2016 without treatment (over 50% higher). Continuing current treatment rates but switch to DAAs could have caused incidence to decline in 2016/17, but will only reduce incidence to 1.9/100py by 2030 (see figure 4). Scaling up to treat 100% within 6 months of diagnosis and 25%/year of those previously diagnosed and untreated could reduce incidence by 84% in 2030 to 0.4/100py (see figure 4). Achieving elimination (90% reduction in incidence) by 2030 requires either an additional risk reduction among HIV infected MSM of 10%, or a shortening of the time to treatment initiation to 3 months.
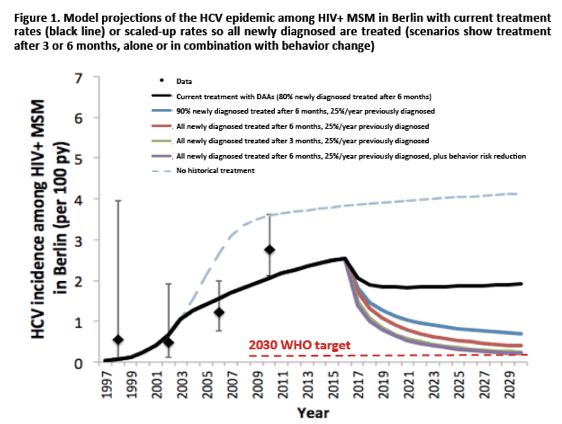
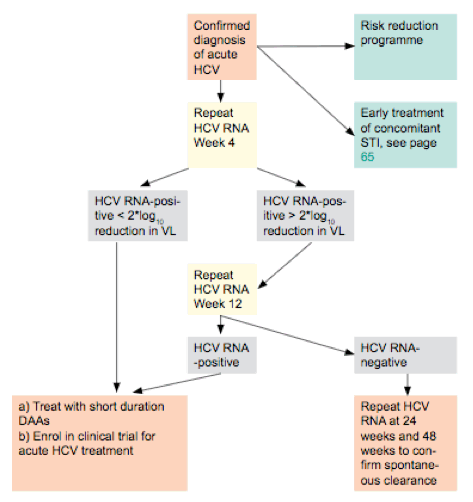
According to this model shortening of time to treatment initiation among patients being newly diagnosed with acute hepatitis C appears to be crucial. The newly included algorithm of the EACS coinfection guidelines for management of acute HCV clearly represents a great step forward in this direction (see figure 5) (26). Indeed, patients after diagnosis should receive a second HCV RNA determination 4 weeks after the initial test, in case there is no drop of at least 2 logs in HCV RNA levels spontaneous eradication of HCV can be excluded and immediate HCV treatment is recommended. This would allow treatment initiation in most patients within the first 3 months of diagnosis and could further contribute to a decrease in HCV transmission and eventually HCV incidence in HIV+ MSM.
Figure 5: Algorithm for management of acute HCV in persons with HIV coinfection (adapted from EACS guidelines version 9.0)
An interesting study on the rate of reinfections over time in IVDU was presented from Greg Dore as a follow-up study to the original Co-STAR study (17, 27). In the original Co-STAR Part A, a phase 3 trial of EBR/GZR for 12 weeks in participants on OAT high SVR12 rates were demonstrated (27). HCV viral recurrence consistent with reinfection was observed in 6/296 participants through follow-up week (FW) 24, with a reinfection rate of 3.4 reinfections/100 person years (95% confidence interval [CI]: 1.3, 7.5). At AASLD the investigators of Co-STAR now provided further analysis of HCV reinfection and injection drug use risk behavior within the ongoing Co-STAR observational study (part B). Co-STAR Part B is a 3-year observational study of participants who received at least one dose of EBR/GZR in Part A (n = 296). During Part B, follow-up occurs every 6 months (6M, 12M, 18M, 24M, 30M, and 36M); if HCV RNA is detected, viral genotype and sequencing are investigated. Participant-reported surveys are also administered at each visit to assess risk behavior. Table 1 summarizes the findings of urine drug screening throughout the different follow-up time-points highlighting that considerable drug use was presented and persisted in the participating study subjects. Overall, 199 participants enrolled in Part B of this study and had data available from the end of treatment through all available follow-up. 74 participants (37%) reported injection drug use during the follow-up versus 125 participants (63%) who reported no injection drug use.
Table 1: C-EDGE Co-STAR Part B: Ongoing risk behavior-urine drug screen
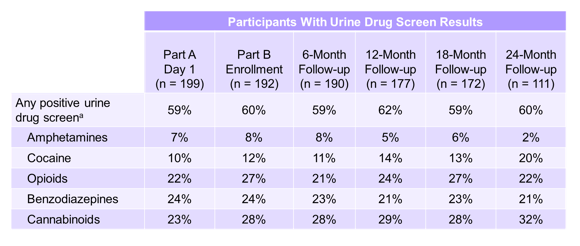
a Excludes methadone and buprenorphine.
The rate of reinfections are shown below in figure 6. Overall, 10 reinfections were documented. Spontaneous clearance was noted in 30% or reinfection cases.
Figure 6: C-EDGE Co-STAR Part B: Incidence of reinfection
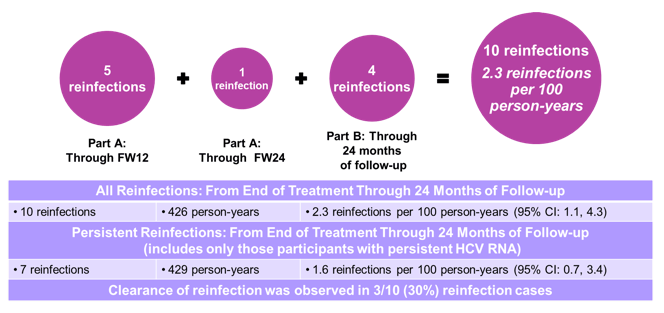
Rate of reinfection was higher in the patients with reporting intravenous drug use (4.2 reinfections/100 person-years 95% CI: 1.5, 9.2) versus patients who did not report intravenous drug use (0.4 reinfections/100 person-years 95% CI: 0.0, 2.3). These findings strongly underline the importance of harm reduction interventions (clean syringe and needle distribution) to prevent reinfections in IVDU.
HEPATITIS C VIRUS REINFECTION AND INJECTING RISK BEHAVIOR FOLLOWING ELBASVIR/GRAZOPREVIR TREATMENT IN PARTICIPANTS ON OPIATE AGONIST THERAPY: C-EDGE CO-STAR PART B - (10/26/17)
Feedback from real world cohorts and clinical trials in special patient populations
Genotype 3
One of the important reasons for analyzing real-life cohorts is clearly their enrichment of patients which would have most likely not be included into clinical trials. In addition, special patient groups that may be under-addressed in clinical trials can be examined in larger patient numbers in extended real-life cohorts. In the DAA development HCV genotype 3 emerged surprisingly as the somewhat more difficult to treat HCV genotype. At this year AASLD several GT3 only analysis were presented. In the ASTRAL 3 study baseline NS5A RAS were associated with a higher risk of virologic failure in GT3 patients receiving 12 weeks of SOF/VEL. The German GECCO cohort attempted to study the impact of additional ribavirin in GT3 patients receiving SOF/VEL therapy (9). The GECCO cohort is a prospective multicenter cohort from 9 sites in Germany. For this analysis, all GT3 infected patients starting SOF/VEL +/-RBV from 8/2016 were included. Overall, 2312 patients have been captured in the GECCO cohort with 25% having a GT3 HCV infection. 232 of the GT3 patients received SOF/VEL+/- RBV (14% = 32 received SOF/VEL + RBV, 86% = 200 only were treated with SOF/VEL). The baseline demographic of this patient population are summarized below in table 2.
Table 2: Baseline demographics of the SOF/VEL cohort in GECCO
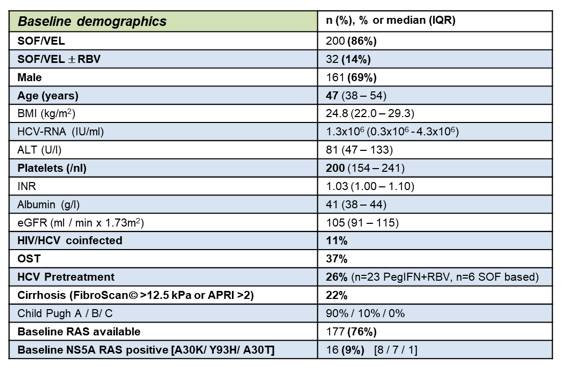
Of note 9% showed baseline NS5A RAS and 22% were cirrhotic. Not all patients had yet full 12 week follow-up data available. Overall, SVR12 results were available for ITT analysis: n=148/232 and for per protocol analysis: n=141/232. The treatment outcome results are shown in figure 7.
Figure 7: SVR 12 in GT3 patients treated with SOF/VEL±RBV for 12 weeks
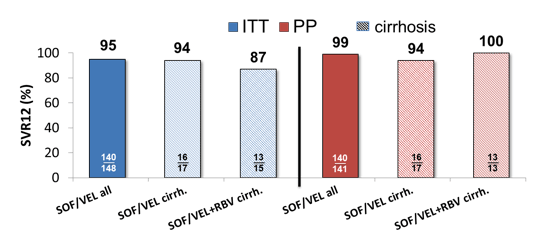
n=3 non-cirrhotic GT3 patients were treated with SOF/VEL+RBV
Only one relapse was noted which occurred in a 54 y old man, HIV-negative, with cirrhosis Child Pugh A who had been pretreated with SOF+RBV for 6 months with relapse. No primary RAS were detected before starting therapy. At time of relapse after SOF/VEL the patient developed a Y93H. The addition of ribavirin did not seem to significantly impact SVR rates. Results by baseline RASs is shown below in figure 8.
Figure 8: SVR 12 in GT3 patients according to RAS at baseline (10% cut off) treated with SOF/VEL±RBV for 12 weeks
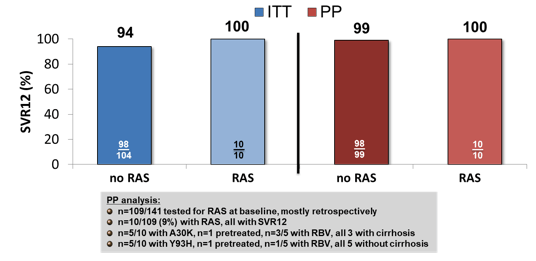
Overall, baseline NS5A RAS with a possible impact on the effectiveness of VEL (A30K or Y93H detected in 10 patients) did not influence the chance for SVR12. Limitation is that not all patients had a genotype performed at baseline and numbers are small. Also the decision to add ribavirin or not was left to the treating physician and may introduce some limitations in comparison to a randomized clinical trial. Therefore, these results should be confirmed in larger datasets.
Do resistance associated substitutions (RAS) or Ribavirin (RBV) use influence treatment success of Sofosbuvir (SOF)/Velpatasvir (VEL) in chronic hepatitis C genotype 3 (GT 3) infection? - Results from the GErman hepatitis C COhort (GECCO) - (10/23/17)
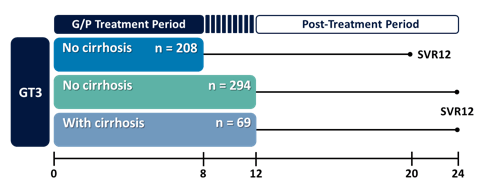
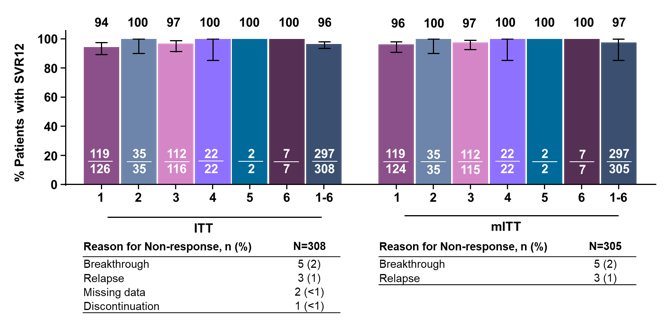
Further data on treatment of GT3 came from an integrated phase II/III analysis of all treatment-naïve GT3 patients receiving glecaprevir/pibrentasvir (G/P) for 8 or 12 weeks (10). Overall, 571 patients across the ENDURANCE-3, EXPEDITION-2, EXPEDITION-4, SURVEYOR-2 (parts 1-3), and MAGELLAN-2 trialswere evaluated. The study design and breakdown of patients according to treatment duration is shown below in figure 9.
Figure 9: Study design
The baseline characteristics for the different treatment duration arms are shown below in table 3.
Table 3a-c: Baseline characteristics
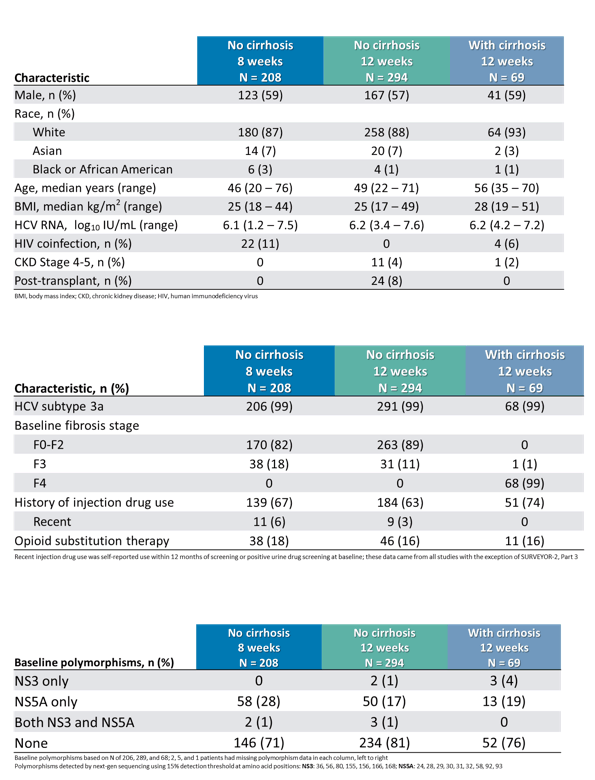
Of note between 17 and 28% of patients had NS5A baseline RAS which potentially could impact outcome of therapy. In addition, the study included a significant number of patients on OST allowing assessing efficacy also in this interesting subpopulation. The treatment outcome results are shown in figure 10.
Figure 10: SVR12 by Intent-to-treat (ITT) and modified ITT (mITT)
In this integrated analyses, G/P achieved high efficacy in treatment-naïve patients with HCV GT3, regardless of cirrhosis status (8 weeks: 95% ITT SVR12 in patients without cirrhosis and 12 weeks: 97% ITT SVR12 in patients with cirrhosis). Extending duration from 8 to 12 weeks had no impact on efficacy for GT3 patients without cirrhosis. G/P was well-tolerated with a safety profile comparable in patients with or without cirrhosis. Baseline RAS had no impact on SVR12 rates.
Efficacy and Safety of Glecaprevir/Pibrentasvir for 8 or 12 Weeks in Treatment-naïve Patients with Chronic HCV Genotype 3: An Integrated Phase 2/3 Analysis - (10/23/17)
EASL: ENDURANCE-3: SAFETY AND EFFICACY OF GLECAPREVIR/PIBRENTASVIR COMPARED TO SOFOSBUVIR PLUS DACLATASVIR IN TREATMENT-NAïVE HCV GENOTYPE 3-INFECTED PATIENTS WITHOUT CIRRHOSIS - (04/21/17)
AASLD: Sofosbuvir/Velpatasvir Fixed-Dose Combination for 12 Weeks Compared to Sofosbuvir with Ribavirin for 24 Weeks in Genotype 3 HCV-Infected Patients: The Randomized Controlled Phase 3 ASTRAL-3 Study - (11/30/15)
AASLD: A Randomized, Phase 3 Trial of Sofosbuvir/Velpatasvir/Voxilaprevir for 8 Weeks and Sofosbuvir/Velpatasvir for 12 Weeks for Patients with Genotype 3 HCV Infection and Cirrhosis: The POLARIS-3 Study - (11/15/16)
Patients with cirrhosis
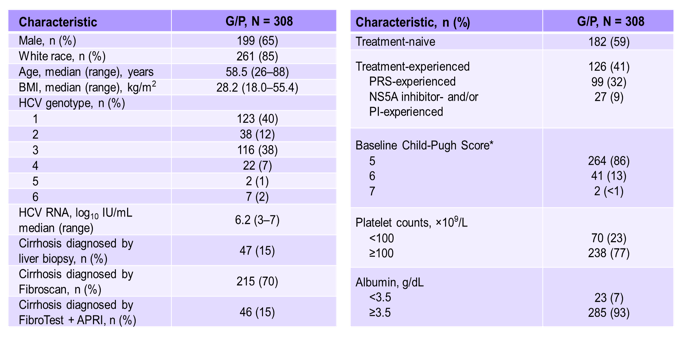
At this year AASLD results from an integrated analysis was presented which evaluated the efficacy, safety and pharmacokinetics of 12 and 16 weeks of G/P treatment in patients with chronic HCV GT1-6 infection and compensated cirrhosis across 4 phase 2 and 3 studies (EXPEDITION-1, EXPEDITION-4, SURVEYOR-2, and MAGELLAN-1 studies) (11). The study included HCV GT1-6-infected adults with compensated cirrhosis, treatment-naïve or previously treated with interferon (IFN), pegylated IFN ± ribavirin (RBV), or sofosbuvir + RBV ± pegIFN, HCV protease inhibitors, or NS5A inhibitors. Cirrhosis was confirmed by liver biopsy, Metavir (or equivalent) score of 4, FibroScan score ≥14.6 kPa or Fibro-Test score ≥0.75 and aspartate aminotransferase (AST)-to platelet ratio >2. The Child-Pugh score was required to be ≤6, with no current or past clinical evidence of liver decompensation. Treatment was G/P 300 mg/120 mg orally once daily, for 12 or 16 weeks. The study design is shown below in figure 11.
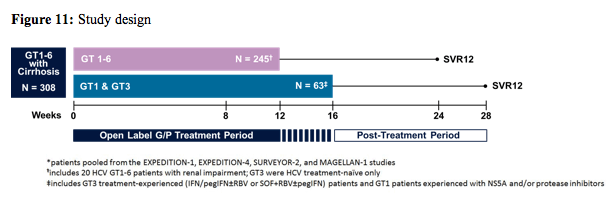
The baseline characteristics are summarized in table 4. Although all genotypes were allowed in only few GT 5 and 6 patients were available. 59% of patients were HCV treatment naïve. The majority of patients had a low Child-Pugh-score as only patients with CHILD A were allowed into the study as G/P is contraindicated in patients with advanced cirrhosis stages.
Table 4: Baseline characteristics
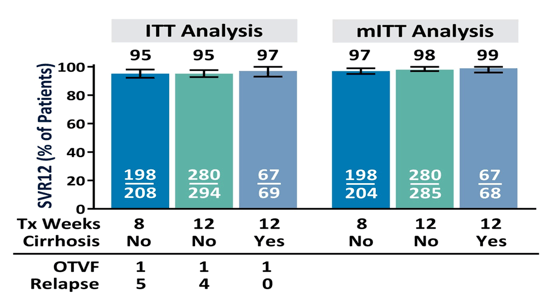
The main findings of this analysis are shown in figure 12.
Figure 12: SVR12: ITT AND mITT ANALYSES
The overall HCV cure rates were quite impressive throughout all genotypes with only 3 relapses in total, indicating excellent outcomes in this more challenging to treat patient population. No difference in outcome was observed between12 and 16 weeks of treatment.
Efficacy, Safety, and Pharmacokinetics of Glecaprevir/Pibrentasvir in Adults With Chronic Genotype 1-6 Hepatitis C Virus Infection and Compensated Cirrhosis: An Integrated Analysis - (10/24/17)
EASL: EXPEDITION-I: Efficacy and Safety of Glecaprevir/Pibrentasvir for Treatment of Chronic Hepatitis C Virus Genotype 1, 2, 4, 5 or 6 Infection in Adults with Compensated Cirrhosis - (04/21/17)
Whereas treatment of compensated cirrhosis seems to work very well as just demonstrated above, the treatment of decompensated cirrhosis remains a controversially discussed topic as clinical benefit of DAA treatment at this late stage of liver disease remains unclear. At this year conference, an analysis on the potential clinical benefit in this challenging patient population was presented (22). Within this study the observed incidence of deaths in patients with hepatic decompensation in the SOLAR studies was compared with mortality predicted by survival models derived from HCV patients with hepatic decompensation prior to DAA availability from the Organ Procurement and Transplantation Network (OPTN). The SOLAR studies were phase 2 trials of sofosbuvir/ledipasvir + RBV for 12 or 24 weeks in patients with Child-Pugh class B or C. The dataset used was to represent the "OBSERVED" data. HCV patients on the waiting list as of January 1, 2007 were used for model development. The model validation cohort was composed from HCV patients on the waiting list as of January 1, 2008. Patients selected met SOLAR study eligibility criteria. This dataset was used to represent the "EXPECTED" data. The baseline patient characteristics for both groups are shown below in table 5.
Table 5: Patient characteristics - see link for clear view of tables
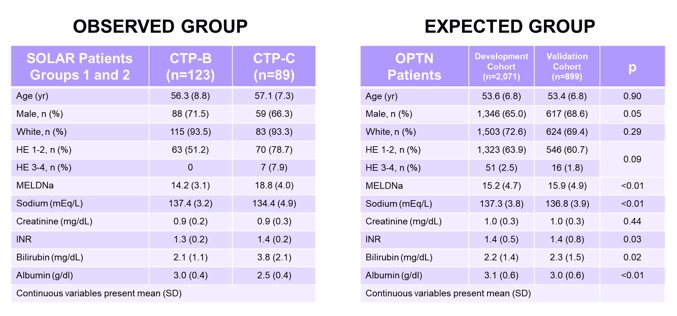
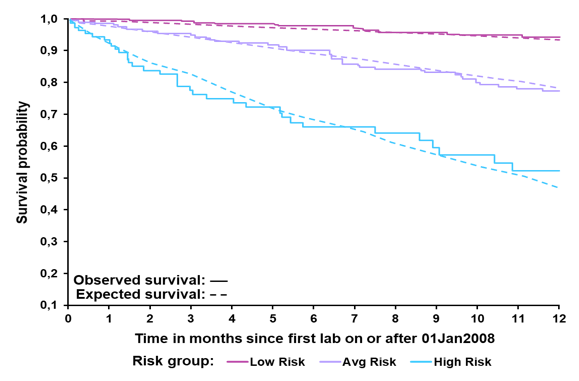
Both study groups were well matched although patients from the expected group were more likely to be male and have slightly higher MELD scores. The comparison between observed and expected survival in the validation group by risk group is shown below in figure 13 and suggests that the model is well validated due to the overlapping curves.
Figure 13: Validation cohort: Observed vs expected survival
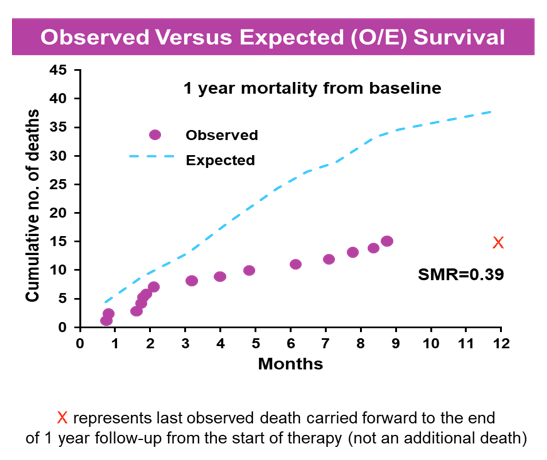
Within the SOLAR data set 15 deaths were recorded within 1 year of therapy. The survival probability between "Observed" versus "Expected" group for the first 100 days showed no difference with a standardized mortality rate (SMR)=0.57. By the 9th death (124 days) a
statistically significant reduction in death (SMR=0.50, 95% CI: 0.26-0.97) was noted. SMR at 1 year was 0.39 (95% CI: 0.24-0.65). When post 12-week treatment data
were used, the model predicted subsequent mortality accurately. The findings are shown correspondingly in figure 14.
Figure 14: Observed vs expected (O/E) survival
In summary, DAA therapy is associated with a significant decrease in mortality risk
in patients with decompensated HCV cirrhosis, by as much as 60% within the first year of therapy.
Survival Benefit of Direct-Acting Antiviral Therapy in Patients with Decompensated Cirrhosis - (11/29/17)
Further real-life cohort experience in treatment of HCV in patients with advanced liver disease was presented from Italy (29). In this prospective real-life observational multicenter cohort study named PITER the impact of DAA treatment on morbidity and mortality in HCV patients with advanced liver disease was compared to patients awaiting treatment. Consecutive DAA treated patients enrolled in PITER, for who follow up data were available, were included in this analysis. Of 1457 DAA patients who completed treatment (median age 60: range 30-80 years; 627 classified as ≤F3 fibrosis by liver elastometry and 830 with cirrhosis by liver elastometry >12.5 kPa and/or clinical cirrhosis. Overall, 1378 (95%) subjects achieved SVR, 79 (5%) failed (25 were in stage F3 and 54 were cirrhotics). Morbidity and mortality were analyzed during a median follow-up of 12 months (range 1-16) in 811 treated patients with cirrhosis and no previously diagnosed HCC before starting DAA therapy. During the same follow-up period, 722 patients with cirrhosis and free of HCC who were awaiting DAA therapy were enrolled into the PITER cohort and evaluated in this study. The baseline demographics for the untreated and treated groups respectively, are summarized below in table 6.
Table 6: Baseline demographics
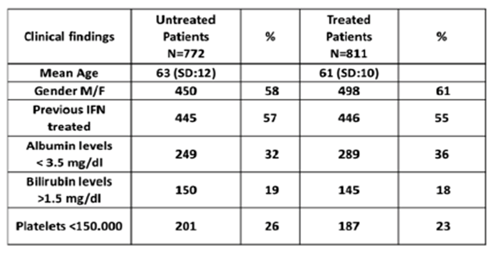
More than half of the patients were treatment experienced and a quarter showed decreased platelets levels suggesting that 75% were in earlier cirrhosis stages. De novo HCC was detected overall in 4% of treated patients (median occurrence time: 12months; range 1-16 months from the end of treatment) and in 2.6% of untreated patients (p=.1). The HCC incidence in patients who failed vs those who achieved SVR was 13.2% (7/53) vs 3.3% (26/758) (p=0.001) respectively. By Kaplan Meier analysis the one year HCC free survival was 97% vs 83% in patients who achieved SVR vs those who didn't (log rank test=0.0001). In addition to failure event in treated patients, other predictors of HCC occurrence by Cox regression analysis were: male gender, older age, and albumin higher than 3 g/dl in untreated patients. Among treated patients who eradicated HCV and untreated patients, 11(1.4%) vs 3 (0.4%) patients underwent OLT and 9 (1.1%) vs 19 (2.5%) patients died (p=0.05) respectively. In 6 ( 70%) of 9 treated vs 11 (60%) of 19 untreated patients who died, death was not liver related.
The authors concluded that these real life data confirm the high efficacy of DDAs treatment in achieving SVR in advanced liver disease patients. The short term HCC rate was similar in treated vs untreated patients, however, it was significantly higher in patients who failed vs those achieving SVR and vs the untreated comparable patients with cirrhosis. Death rate was lower in patients who achieved SVR vs untreated.
Real life clinical outcomes in DAA-treated and untreated patients with HCV advanced liver disease. Interim one year data from the ongoing PITER cohort - (11/17/17)
HCV patients with chronic kidney disease
Elbasvir/grazoprevir (EBR/GZR) is indicated for the treatment of chronic HCV genotypes 1 and 4 infections and has demonstrated high sustained virologic response (SVR) in patients with chronic kidney disease (CKD) (30). At AASLD data was presented from the Veterans Affair population on the real-world effectiveness and use of EBR/GZR in treatment of patients with chronic HCV and chronic kidney disease (CKD) (6). The study population included all HCV-infected patients who initiated EBR/GZR from Feb. 1-Dec. 31, 2016, were ≥18 years, had a positive HCV RNA, ≥1 inpatient or outpatient visit within 1 year prior to treatment and had ≥2 eGFR values, at least 90 days apart (N=5709). Overall, 1528 patients with CHD stage 3-5 were included into the per protocol analysis. The distribution of CKD stages for the entire study population is shown below in figure 15 and the baseline demographics in table 6.
Figure 15: Distribution of Patients by CKD Stages
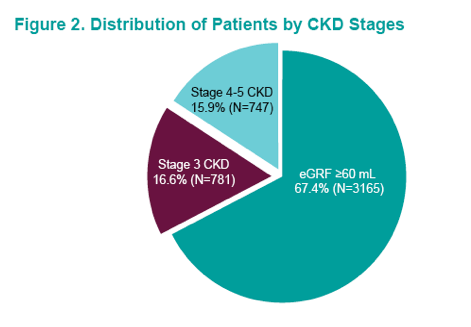
Table 7: Baseline Clinical Characteristics of HCV/CKD Patients Treated with EBR/GZR by CKD Stages
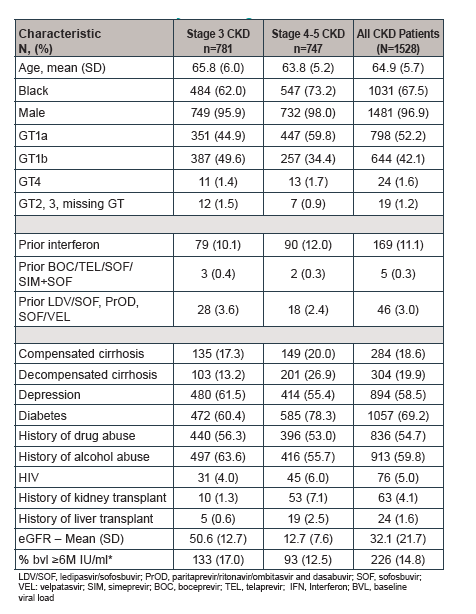
Clearly, this is to date the largest study available with regard to outcomes of DAA therapy in CKD patients. Of note, 18% were categorized as compensated cirrhotics and almost 20% as decompensated cirrhotics. As all PI based DAA regimens EBR/GZR is not indicated in patients with CHILD B or C cirrhosis. Therefore, it remains somewhat unclear how decompensated cirrhosis was coded taking into account the restrictions for EBR/GZR in treatment of advanced cirrhosis stages which would suggest increased presence of decompensated cirrhosis. Could be that these refers to an episode of ascites in general. The SVR rates obtained in the study are shown below in figure 16.
Figure 16: SVR12 rates overall and by CKD stage
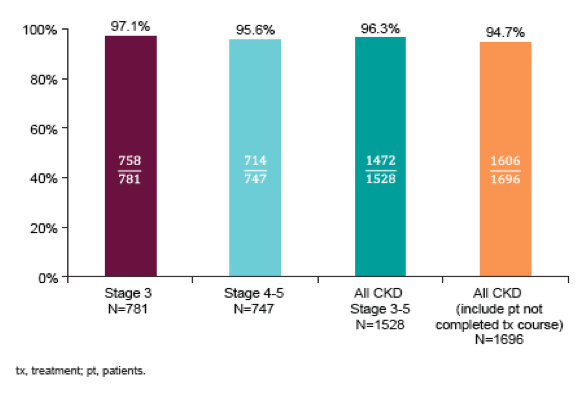
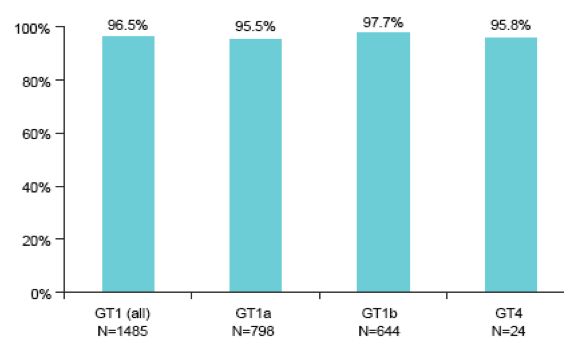
Independent of CKD stage high SVR rates were obtained for all patient groups. HCV cure rates above 95% were found for all respective genotypes in patients with CKD stage 3-5 (see figure 17).
Figure 17: SVR Rates by Genotype in EBR/GZR Patients with CKD Stages 3-5
In summary, EBR/GZR was highly effective in HCV-infected patients with moderate to severe CKD with SVR (PP) of 96.3% overall. Reassuringly, SVR rates were high across select patient subgroups in a large clinical setting.
Effectiveness of Elbasvir/Grazoprevir in Patients With Chronic Hepatitis C and Chronic Kidney Disease:Results From the Veterans Affairs System - (11/27/17)
HCV therapy in PWIDs
Even in the presence of access to HCV therapy, linkage to treatment remains challenging among PWIDS. At AASLD a study was presented which evaluated the impact of cash incentives or peer mentors to improve HCV linkage and treatment among HIV/HCV coinfected persons who inject drugs and were so far not engaged in HCV care (The CHAMPS Study) (4). HCV treatment-naïve PWIDs with genotype 1 infection were eligible if they received HIV care at Johns Hopkins and had not been evaluated for HCV care within 8 months, had a CD4 count >100 mm3, and no evidence of decompensated liver disease. Eligible participants were randomized (1:2:2) to 1) Usual Care (UC, nurse supervision); 2) UC + Peer-Mentors; 3) UC + Cash Incentives. All participants were provided 12 weeks of ledipasvir/sofosbuvir (LDV/SOF) at no cost. Overall 904 individuals were assessed for eligibility of whom 144 participated. 460 patients were already engaged in HCV care, 13 declined participation and 287 could not be reached. The baseline characteristics of the study participants is shown below in table 8.
Table 8: Baseline characteristics of the CHAMPS study
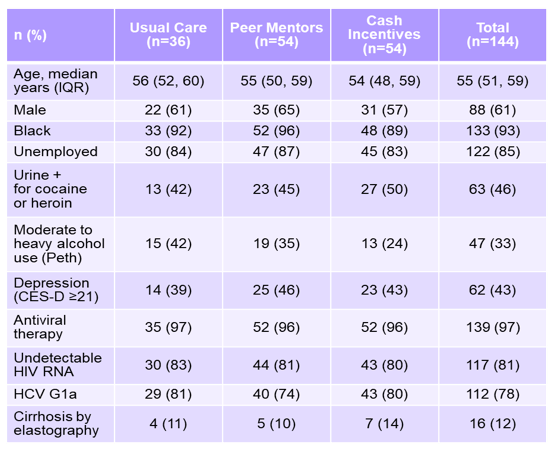
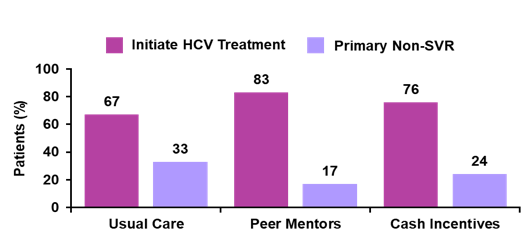
Clearly, this patient population represented a real-life challenging PWID group. 85% of the participants were unemployed with high rates of ongoing substance and alcohol abuse. The majority had a genotype 1a infection and only 12% were cirrhotic. Despite access, ∼1/3 of HIV/HCV + PWID did not initiate treatment with usual care. Figure 18 shows the percentage of patients which initiated HCV therapy for the different intervention groups, respectively.
Figure 18: Who started therapy in the CHAMPS study
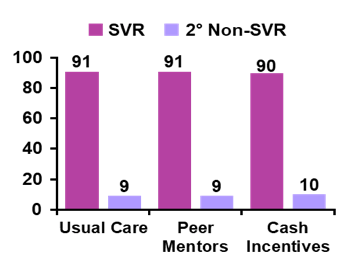
According to the study results, peer mentors appear to increase uptake. SVR12 was achieved by 91% of all study subjects who started HCV therapy%). One reinfection (1b to 1a) was noted as well as two relapses. Six patients stopped therapy early (5 prior to Day 28), three due to side effects and three due to non-adherence. One death occurred after completion of LDV/SOF (HCV RNA not detected). Figure 19 shows the SVR12 results.
Figure 19: SVR12 results of the CHAMPS study
In summary, almost a quarter of 144 coinfected PWIDs did not initiate HCV treatment despite access to expert clinicians and LDV/SOF at no cost. Access to DAAs is necessary but may not be sufficient to achieve HCV cure among populations of PWIDs; these data suggest peer-mentors may effectively increase HCV linkage and treatment.
Randomized Controlled Trial of Cash Incentives or Peer Mentors to Improve HCV Linkage and Treatment Among HIV/HCV Coinfected Persons Who Inject Drugs: The CHAMPS Study - (10/24/17)
Among the poster session confirmatory data on high SVR rates in PWIDs who participate in opioid substitution programs was reported (31). Thirty-five HCV patients from a single methadone clinic received HCV treatment with PrOD which was administered
with daily methadone and electronic pill box. Of note five subjects receiving ribavirin discontinued treatment: 1 due to hemolytic anemia and 4 due to GI intolerance.
One subject was lost to follow up in the 12-week post treatment period and another was lost prior to undergoing 1 yr post-treatment testing. There were no virologic relapses. SVR rates are shown below in figure 20.
Figure 20: SVR treatment outcome
The authors concluded that add-on HCV treatment to methadone makes sense. As no reinfection occurred up to one year follow-up these data support that concurrent treatment of opiate use disorder may lower reinfection risk.
No Evidence of 1-Year Reinfection after Treating HCV at a Methadone Program - (11/29/17)
HCV therapy in organ transplant recipients
The availability of all oral IFN free DAA therapy for treatment of HCV recurrence in liver transplant recipients is obviously very attractive. So far little data is available for newer pangenotypic DAA combinations. At AASLD a study was presented which evaluated the safety and efficacy of SOF/VEL in adult patients with recurrent chronic GT 1-6 HCV infection post-liver transplant (8). In this single-arm, open-label phase 2 study conducted in Europe 79 subjects were included. The baseline characteristics are shown below in table 9.
Table 9: Baseline characteristics
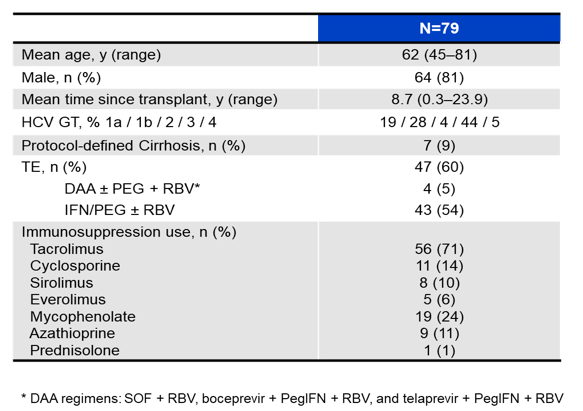
Most of the treated patients had GT1 or 3 infection with high amounts of prior treatment failures (60%) but low percentage of cirrhotics (9%). Overall high SVR rates were achieved (see figure 21).
Figure 21: SOF/VEL for 12 Weeks in GT 1-4 HCV-Infected Post-Liver Transplant Patients
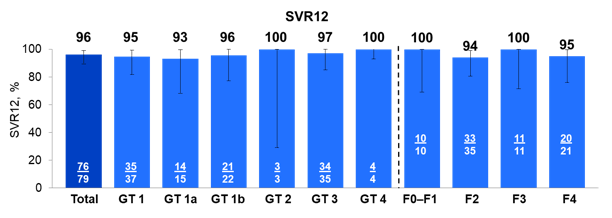
Only three patients did not achieve SVR: one early D/C and 2 relapses. 4/4 patients with baseline Y93H RASs (3 GT 3 and 1 GT 1b) achieved SVR12. No changes in immunosuppression were needed for rejection or suspected drug-drug interactions. As Pis are more prone to lead to DDIs the availability of a pan-genotypic NS5a based regimen is of great importance.
Sofosbuvir/Velpatasvir for 12 Weeks in Genotype 1-4 HCV-Infected Liver Transplant Recipients - (11/27/17)
New treatment options for treatment of Gt1b
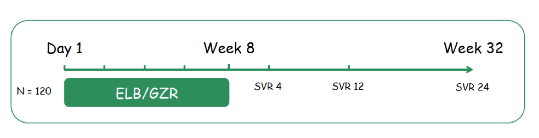
Genotype 1b is the most common HCV genotype globally, accounting for the largest proportion of infections in Europe, Latin America, Russia, Turkey, and East Asia. Reducing treatment duration can improve adherence and reduce drug exposure. In addition, in cost sensitive markets this may help to broaden access to DAA therapy independent of fibrosis stage. Accordingly, within the STREAGER trial the efficacy of 8 weeks of the protease inhibitor grazoprevir 100mg/d (GZR) and NS5A inhibitor elbasvir 50 mg/d (EBR) was evaluated among treatment-naïve HCV patients with non-severe fibrosis. The study design is shown below in figure 22.
Figure 22: Study design of the STRAEGER trial
Overall, 53 patients were available for full analysis at time of AASLD presentation. The baseline demographics are shown below in Table 10.
Table 10: Baseline demographics

The SVR4 and SVR 12 rates are shown below in figure 23. Overall, two relapses were noted. One relapse was in a GT1b patient with a particularly high baseline viral load of 14.000.000 IU/mL.
Figure 23: SVR results
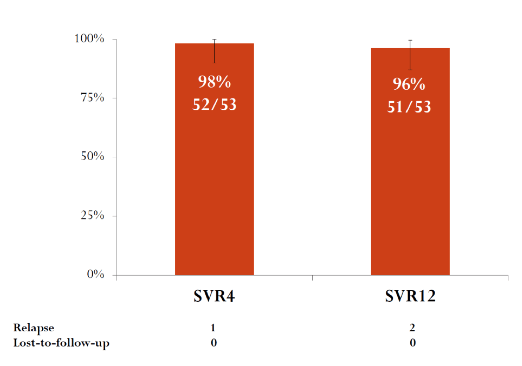
In conclusion, high SVR12 (51/53, 96%) was achieved in a TN non-severe fibrosis GT1b-infected population in patients treated for 8 weeks by the combination of grazoprevir and elbasvir.
A Phrase 3, French Multicenter, Open-Label Study to Investigate the Efficacy of Elbasvir/Grazoprevir Fixed-Dose Combination for 8 Weeks in Treatment-Naïve HCV GT1b-Infected Patients with non-severe Fibrosis: STREAGER study - (10/24/17)
Treatment of prior DAA failures
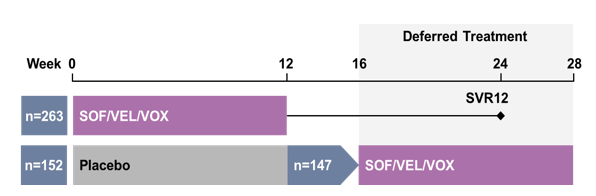
At AASLD new data around retreatment of previous DAA failures (with documented resistance development) with new DAA combinations was presented (13-14). In the phase 3 POLARIS-1 study, sofosbuvir/velpatasvir/voxilaprevir (SOF/VEL/VOX), a fixed-dose combination containing pangenotypic inhibitors of the NS5B, NS5A, and NS3/4A HCV viral proteins, respectively, demonstrated high efficacy in HCV-infected patients previously treated with an NS5A inhibitor (32). This analysis describes the safety and efficacy for SOF/VEL/VOX in patients initially randomized to placebo treatment who subsequently received SOF/VEL/VOX for 12 weeks in a deferred treatment substudy. HCV-infected patients who received placebo treatment in the POLARIS-1 study subsequently received SOF/VEL/VOX (400mg/100mg/100mg)daily for 12 weeks. The endpoints were the sustained
virologic response 12 weeks after cessation of treatment (SVR12) and the assessment of adverse events and laboratory analyses. The study design is shown below in figure 24.
Figure 24: Study design
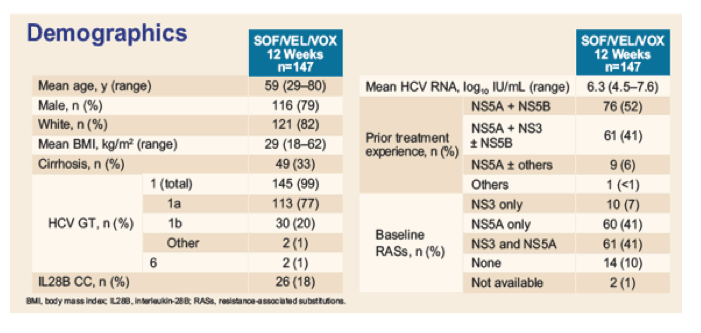
The baseline demographics for the participating subjects in this new subanalyses from the POLARIS study are summarized in table 11.
Table 11: Baseline characteristics from the POLARIS-1 study
Around half of the study subjects had failed a prior NS5A+NS5B combination, and 41% of patients had NS5A baseline RASs only, whereas another 41% had both, NS5A and NS3 mutations. Clearly, this again is a challenging group of patients to treat with quite extensive baseline resistance. The SVR rates by genotype are shown below in figure 25.
Figure 25: Polaris-1 study: SVR12 by genotype/subtype
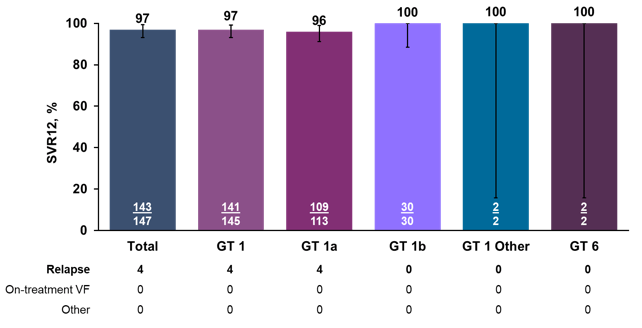
Most impressively, a total of 97% of patients achieved SVR12 despite prior DAA failure and extensive resistance at baseline. Only in GT1a patients were four relapses recorded. Figure 26 shows the previous NS5A exposure by compound used as well as the SVR12 rates by prior DAA exposure.
Figure 26: Polaris-1 study: Prior NS5A exposure and SVR12
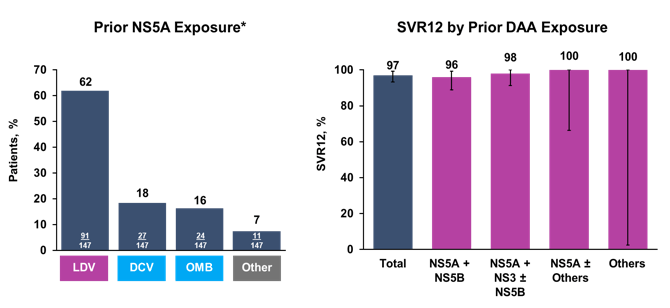
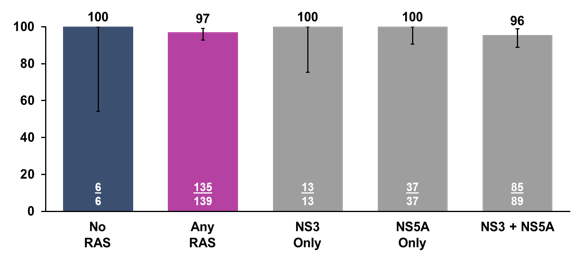
High SVR12 rates were obtained independently from the DAA drugs used before. In addition RASs at baseline also did not have any impact on the likelihood of achieving SVR12 as shown in figure 27.
Figure 27: POLARIS-1 study: SVR12 by Baseline RAS
Of the 4 patients who relapsed, 2 developed treatment-emergent resistance. 1 had treatment-emergent NS3 Y56H, D168A/V, and NS5A L31L/M and 1 had treatment-emergent NS3 V36V/A.
Overall, these new results confirm that the single tablet regimen of SOF/VEL/VOX for 12 weeks is safe, well tolerated, and highly effective for patients who have previously failed NS5A inhibitor-containing DAA regimens.
SOF/VEL/VOX for 12 Weeks in NS5A-Inhibitor-Experienced HCV-Infected Patients: Results of the Deferred Treatment Group in the Phase 3 POLARIS-1 Study - (10/23/17)
Susceptibility to Voxilaprevir of NS3 Resistance-Associated Substitutions and Clinical Isolates From Direct-Acting Antiviral-Experienced and -Naïve Patients - (10/26/17)
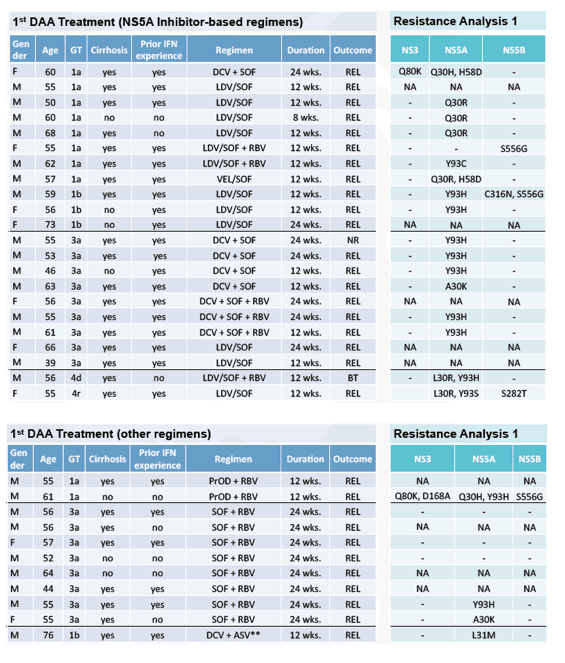
Beyond, the above summarized findings from a randomized controlled trial for retreatment of prior DAA combination treatment failures there also was increasing feedback from retreatment of prior DAA failures from the real-world setting. Professor Vermehren (14) presented data from an international study, which assessed the clinical and virological characteristics and retreatment options of patients who failed multiple courses of DAA therapies in a real-world setting. Clearly, resistance-wise this is the most challenging to treat patient group one can imagine. Clinical and virological data were collected from patients who failed at least two IFN-free DAA regimens in a large European HCV resistance database. Resistance associated substitutions (RASs) were determined by means of population sequencing of the NS3, NS5A, and NS5B coding regions after each treatment failure where available. Retreatment was primarily based on guideline recommendations, i.e. Patients who failed on a regimen containing an NS5A inhibitor were retreated with a protease inhibitor and sofosbuvir, and patients who failed on a protease inhibitor-based regimen were retreated with an NS5A inhibitor and sofosbuvir. However, detailed recommendations for retreatment were also given based on the resistance analysis, where available. All patients were retreated at the discretion of the treating physician. Thus, some patients did not receive guideline recommended and/or resistancebased regimens. A total of 33 patients with failure to multiple courses of IFN-free DAA therapies were identified. RASs were detectable in the majority of patients (77%) after the first DAA therapy. Table 12 summarizes the clinical characteristics and results from the first resistance test after DAA failure.
Table 12: Clinical characteristics and results from first resistance test
The table 13 shows the selected retreatment DAA combination and in how far this met guideline recommendations. Also results from a second resistance test if available is captured.
Table 13: 2nd DAA therapy and resistance test
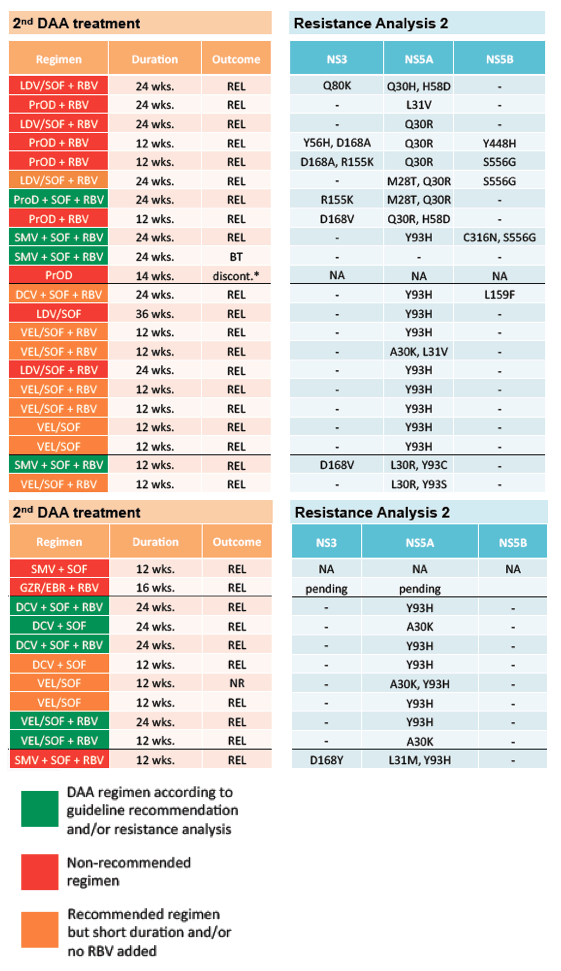
Clearly, the majority of retreatments did not occur based on guidelines recommendations or resistance test which at least partially may be explained by access limitations to some of the newer drugs at that time. The majority of patients who did not respond to multiple DAA therapies were male (76%) and had cirrhosis (79%). Resistance testing was performed in 71% of patients after the first failed course of DAA therapy. Despite this, retreatment was not consistently tailored to the RASs detected. Importantly, treatment failure was even observed in patients in whom the best available retreatment option was chosen. Therefore, although probably a very rare event, some patients who do not respond to multi-targeted rescue therapies may never be cured. The outcome and respective 3rd DAA combinations chosen are shown in table 14.
Table 14: 3rd DAA combination and outcome
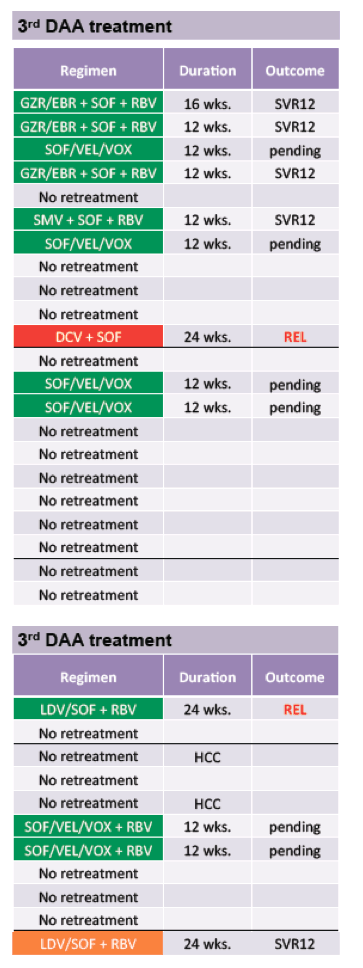
Overall, 13/33 of 2 DA treatment failures were retreated for a third time with 5/7 with SVR12 follow-up achieving HCV cure. Of note, all patients who used recommended salvage regimens achieved SVR12.
Failure to multiple Interferon-free direct Antiviral therapies: could Chronic hepatitis C become incurable? - (11/27/17)
New dual and triplet HCV DAA combinations
At this year AASLD only few new studies on new DAA combinations were presented. Indeed, already prior to the liver meeting Merck and Janssen independently had issued press releases informing that they were going to stop their further DAA development programs. Clearly, with SVR rates above 95% with various pangenotypic regimens new drugs have ambitious targets to meet. Following the impressive high SVR rates observed in the C-SURGE trials which evaluated the new fixed-dose combination of grazoprevir (GZR), ruzasvir (RZR), and uprifosbuvir (UPR) in HCV patients with prior DAA treatment failure, the company now presented results from the ongoing studies with the dual combination of ruzasvir and uprifosbuvir (1,33 ). The C-BREEZE-1 study was a phase 2, 12-week, open-label, clinical trial in which all participants received RZR 60mg + UPR 450mg once daily as separate medications, administered under fasting conditions (33). Overall, 60 patients were enrolled (GT1a, n=54); GT1b, n=15; GT2, n=29; GT3, n=39; GT4, n=20; GT5, n=0; GT6, n=3), of whom 50 (31%) had cirrhosis. The study design is depicted below in figure 28.
Figure 28: Study design of C-BREEZE-1
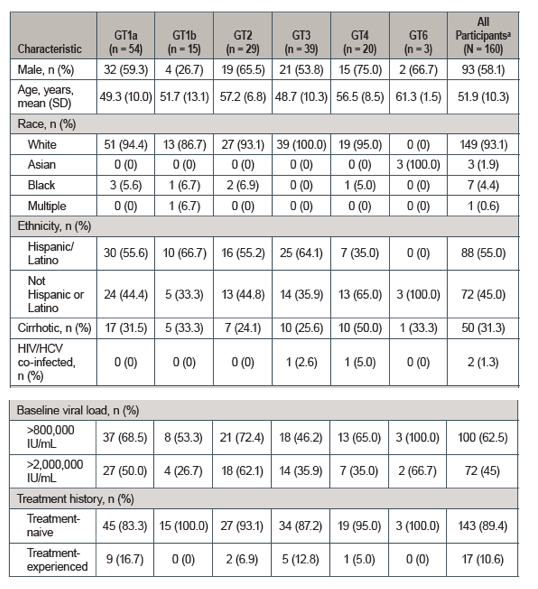
The baseline characteristics are shown below in table 15. Few HIV-coinfected patients were included. 10.6% were treatment experienced.
Table 15: Baseline characteristics with the BREEZE-1 study
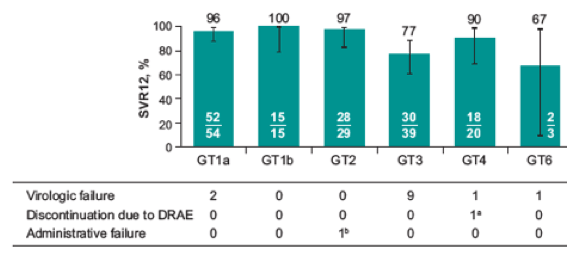
The main treatment results are depicted below in figure 29. Of note, a relatively high virological failure rate was noted in the genotype 3 patients; but there were also two virological failures in the genotype 1a patients. Clearly, these results were suboptimal and suggested limited efficacy for some genotypes which most likely impacted the decision to stop further development of this particular combination.
Figure 29: Study results from the BREEZE-1 trial
C-BREEZE 1: Efficacy and Safety of Ruzasvir 60 mg and Uprifosbuvir 450 mg for 12 Weeks in Adults With Chronic Hepatitis C Virus Genotype 1, 2, 3, 4, or 6 Infection - (11/29/17)
The BREEZE-2 study was an open-label, single-arm trial of combination regimen of 12 weeks of RZR 180 mg and UPR 450 mg administered as single entities. Of note this was twice the RZR dosage which was studied in the above summarized BREEZE-1 trial. The study consisted of two enrollment cohorts: initial enrollment of 50 participant, pause to assess safety, and if no safety criteria are met, continued enrollment for a total of n=250. The study design is shown below in figure 30.
Figure 30: Study design of the BREEZE-2 trial
The baseline characteristics for the study population are shown below in table 16. All HV genotypes were allowed into the study.
Table 16: Baseline characteristics of the BREEZE-2 trial
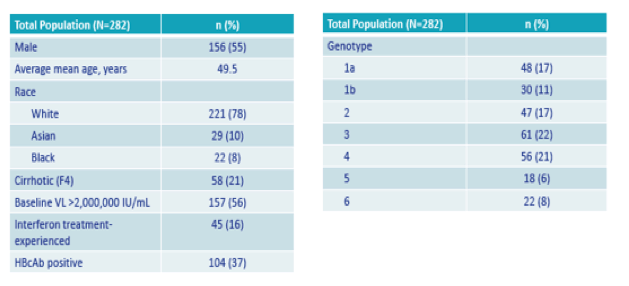
Good cross genotype representation was achieved in this study. Only 21% were cirrhotic and 16% IFN-experienced. The overall SVR12 rates for the full analysis set as well as the modified analysis set ae shown below in figure 31.
Figure 31: Overall efficacy in the BREEZE trial
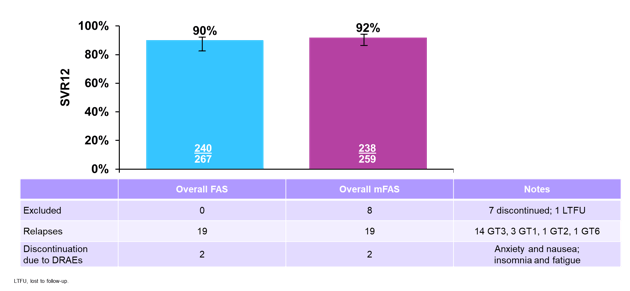
Only a disappointing 90% overall achieved SVR12. In total 19 relapses were recorded, again just like in BREEZE-1 the majority of relapses occurred in GT3 patients and to a lesser extent in GT1a patients. A breakdown of SVR12 rate by genotype is shown in figure 32.
Figure 32: C-BREEZE-2 study: SVR12 rate by genotype
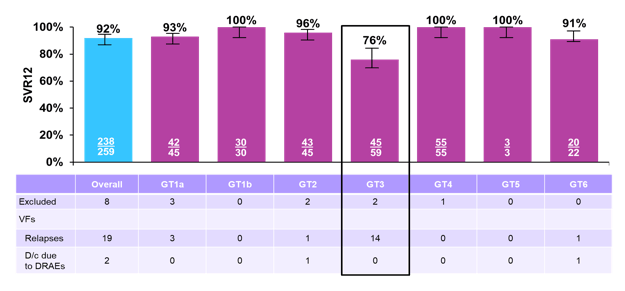
The overall efficacy in GT3 patients was 76%. An even lower SVR12 rate of 68% was observed in the subset of cirrhotic GT3 patients, whereas SVR12 rate in non-cirrhotic GT3 patients was 80%. The overall efficacy for GT1a was also relatively low with 93%. Most interestingly, the lower SVR rates in GT 3 patients were observed with and without baseline RASs as shown below in figure 33.
Figure 33: C-BREEZE-2 study: Lower Efficacy in GT3-Infected Participants With and Without Baseline RASs
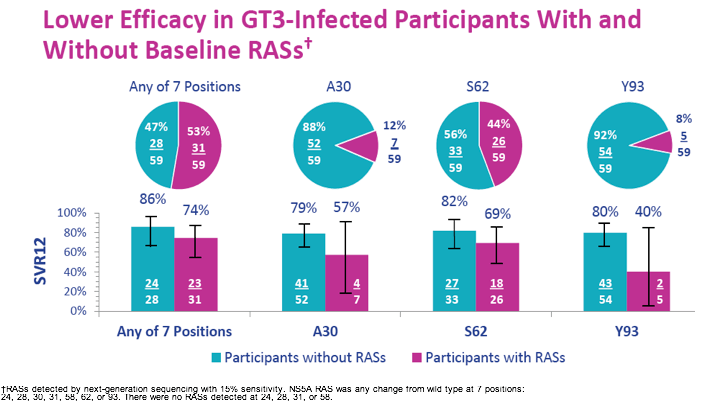
In contrast in GT1a patients the presence of the 93H RAS at baseline was associated with a lower likelihood of obtaining SVR12 (see figure 34).
Figure 34: C-BREEZE-2 study: Lower Efficacy in Only Those GT1a-Infected Participants With Y93 Baseline RAS
The main safety results are summarized in table 17. Overall, this dual DAA combination was tolearated very well with hardly any discontinuations because of drug related adverse events.
Table 17: C-BREEZE-2 study: Safety - Adverse event summary
In summary, the two-drug combination of RZR 180 mg + UPR 450 mg led to an overall SVR12 rate of 90%. Higher relapse rates were observed in GT3 patients but also in GT1a patients in particular if they had the Y93H RAS at baseline. The company, which has evaluated this combination, has announced their termination of this DAA development program.
C-BREEZE-2: Efficacy and Safety of a Two-Drug Direct-Acting Antiviral Agent Regimen Ruzasvir 180 mg and Uprifosbuvir 450 mg for 12 Weeks in Adults With Chronic Hepatitis C Virus Genotype 1, 2, 3, 4, 5, or 6 - (10/23/17)
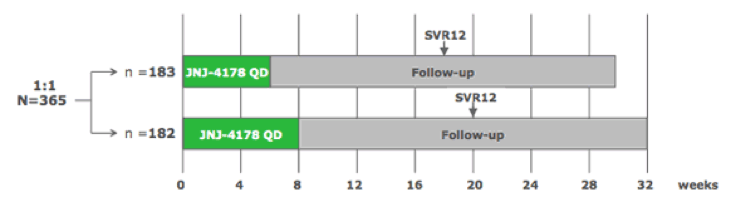
The only other new DAA combination presented at this year AASLD was the 3 DAA combination of AL-335 (novel uridine-based nucleotide analog HCV NS5B polymerase inhibitor), odalasvir [ODV] (a novel HCV NS5A inhibitor) and simeprevir (a licensed HCV PI). The combination of AL-335 800 mg, ODV 25 mg, and SMV 75 mg once daily (QD) (also known as JNJ-4178) was being further evaluated in the Phase IIb OMEGA-1 study (NCT02765490). This multicenter, randomized (1:1), open-label study included treatment-naïve pts or pts previously treated with (peg)interferon (IFN) (18-70 years of age) with HCV GT1, 2, 4, 5, or 6 infection, without cirrhosis. Exclusion criteria included prior DAA treatment and HCV GT3 infection. Pts received JNJ-4178 for 6 or 8 weeks. The study design of the OMEGA-1 study is shown below in figure 35.
Figure 35: Study design of the OMEGA-1 trial.
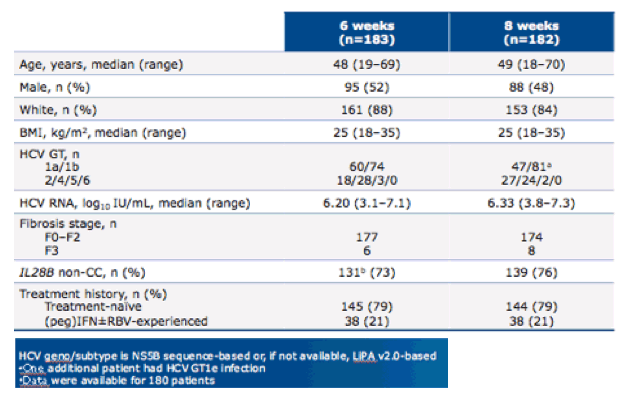
The baseline characteristics of the study population are shown below in table 18. The majority of patients were treatment-naive (79%) and had early fibrosis stages (F0-F2).
Table 18: baseline characteristics
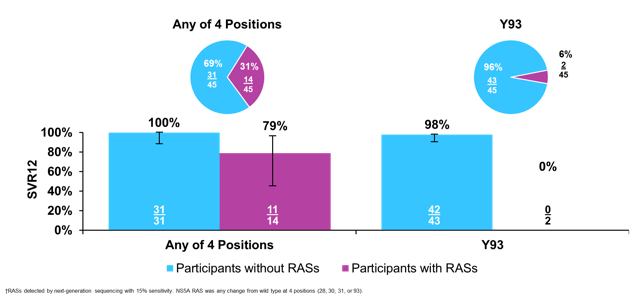
The main SVR12 results are shown in figure 36.
Figure 36: OMEGA-1 study: SVR12 (ITT population)
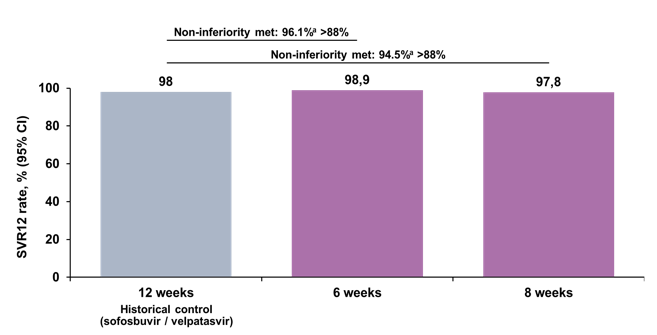
Overall, impressive SVR rates were achieved for both study arms above 97%. Moreover, non-inferiority was determined in comparison to a historical control of SOF/VEL. A breakdown of SVR12 rate by HCV genotype and subtype is shown in figure 37.
Figure 37: OMEGA-1 study: SVR12 by geno/subtype (excluding non-virologic failures)
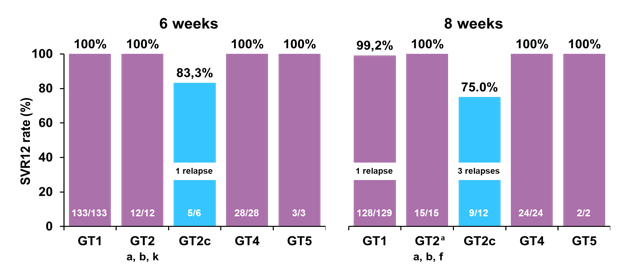
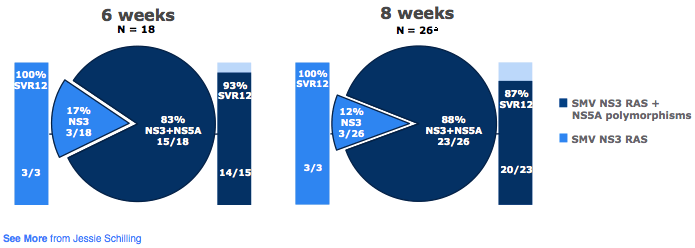
Of note of the 5 relapses noted during the trial 4/5 were reported in GT2c patients. All GT2 infected patients had baseline SMV NS3 RASs and NS5A polymorphisms. All GT2-infected patients who relapsed had at baseline an NS5A F28C polymorphism in addition to SMV RASs; but not all patients with F28C had a relapse (7/11 SVR12). The prevalence and impact of baseline RAS for GT2 patients is shown below in figure 38.
Figure 38: OMEGA-1 study: GT2-infected patients - Prevalence and impact of baseline SMV NS3 RASs and NS5A polymorphisms
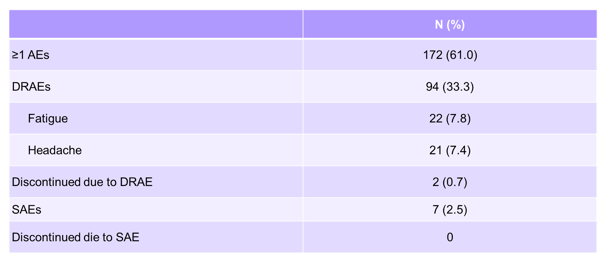
The safety findings are summarized in table 19.
Table 19: OMEGA-1 study: Adverse events of interest
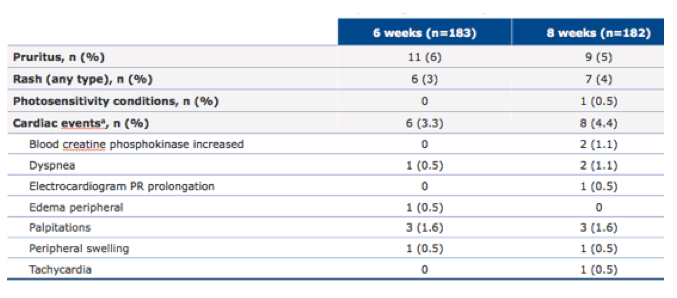
Isolated asymptomatic increases in PR interval (>200 ms) were seen in a low number of patients (n=8, 2.2%) and were not considered clinically relevant. Some of the skin reactions can be attributed to the well-known adverse event profile from simeprevir. Overall, tolerability was good with no discontinuations because of adverse events.
In summary, this new 3 DAA combination promised good efficacy above 97%. However, the developing company nonetheless decided to terminate their further DAA development program because of the already available very effective existing DAA combinations. Maybe the lack of activity for GT3, as well as the observed relapses in GT2c patients has further influenced this decision.
Evaluation of the efficacy and tolerability of JNJ-4178 (AL-335, odalasvir, and simeprevir) in hepatitis C virus-infected patients without cirrhosis: The Phase IIb OMEGA-1 study - (10/23/17)
Virology analyses from HCV-infected patients treated with the combination of AL-335 and odalasvir, with and without simeprevir, in Phase II studies - (12/01/17)
Impact of Sustained Virologic Response with Direct-Acting Antiviral Treatment on Mortality and Hepatocellular Carcinoma
Reduced all-cause mortality remains the ultimate goal of direct-acting antiviral (DAA) treatment. Therefore, the impact of sustained virologic response (SVR) achieved with oral DAAs on all-cause mortality and incident hepatocellular carcinoma (HCC) was evaluated in veterans with and without advanced chronic liver disease (ACLD) (20). In this impressive analysis 15,069 patients with advanced liver disease (FIB 4 >3.25) and 41,226 patients with less advanced disease were included. All-cause mortality rates and incident HCC rates were determined for all veterans in the HCV registry treated with DAAs by 30 September 2016 with laboratory-confirmed SVR or No SVR. SVR was associated with a significant reduction in mortality as well as a reduction in incident HCCs (see figure 39).
Figure 39: K-M curves for ACLD deaths and HCCs
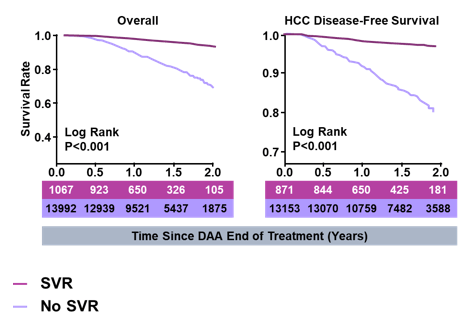
In both ACLD and non-ACLD patients, SVR was strongly associated with delayed time until death (p<0.001) and until development of first HCC (p<0.001). The reductions in mortality and HCC with SVR were consistent across genotypes and were similar when considering one-year mortality and HCC rates. Reduced odds of death occurred in both ACLD and non-ACLD patients with SVR compared to No SVR (ACLD: odds ratio (OR) 0.20, 95% confidence interval (CI) 0.16-0.25, p<0.001; non-ACLD: OR 0.24, 95%CI 0.18-0.33, p<0.001).
In conclusion, ACLD and non-ACLD patients achieving SVR after DAA treatment had significantly lower all-cause mortality and lower incident HCC rates than those without SVR.
Impact of Sustained Virologic Response with Direct-Acting Antiviral Treatment on Mortality and Hepatocellular Carcinoma - significantly lower mortality, HCC 60% to 84% - (10/24/17)
What are the remaining hurdles in access to cure?
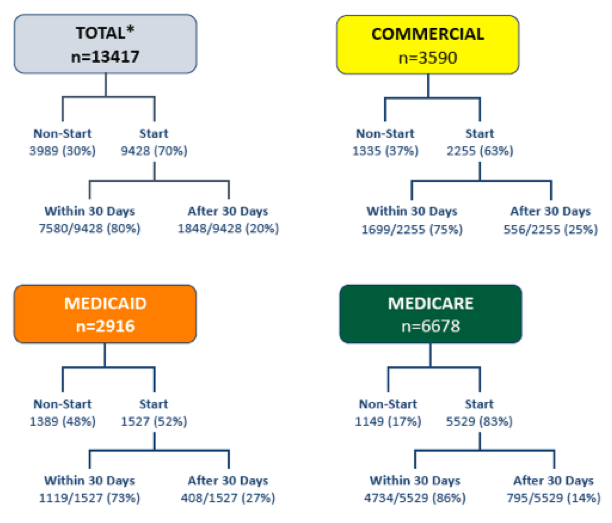
Since 2013, the fraction of patients starting prescribed curative therapies for HCV has decreased because of insurance denials and/or processes. At this year AASLD a study was presented which examined both non-start rates and time to fill by payer type to better understand the larger populations facing hurdles in access to cure (34). Data collected from providers and specialty pharmacies through Trio Health's disease management program were limited to patients (n=13417) who received an anti-HCV prescription in 2016. 3989 (30%) of 13417 patients did not start prescribed therapy. Non-start reasons were specified in 2535/3989 (64%) of patients with most indicated as insurance denial (83%, 2114/2535). Failure to start therapy occurred in 37% (1335/3590) commercial (COM), 48% (1389/2916) Medicaid (MCD), and 17% (1149/6678) Medicare (MCR) (see figure 40). Cure access varied by payer type. Start rates ranged from 52% to 83%. Of the patients who started, 73% to 86% received drug within 30 days of prescription.
Figure 40: TRIO: Patient disposition
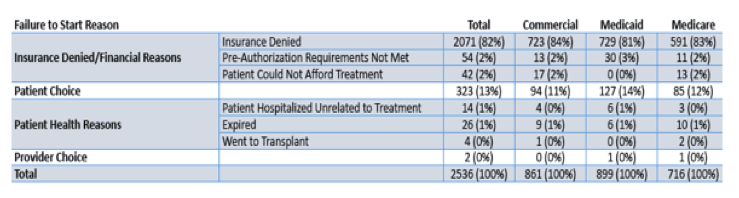
Non-start reasons were specified in 2535/3989 (64%) of patients and are shown below in table 20. Overall, insurance denials and processes accounted for 84% (2125/2535) of non-start reasons.
Table 20: TRIO: Reasons not to start DAA therapy
Variables associated with non-start and delayed start populations include COM or MCD coverage and less severe disease as based on fibrosis, platelets, viral load, and reduced presence of comorbids diabetes and hypertension. Accounting for both non-starts and delayed time to fill, 53% (1891/3590) of COM, 62% (1797/2916) of MCD, and 29% (1944/6678) of MCR patients face barriers in access to cure. Clearly, in light of the high SVR rates achievable with modern DAA therapy and the clinical benefit associated with SVR this is not acceptable and calls for improvement.
Cure denied and cure delayed in Chronic Hepatitis C; monitoring non-start rates and increased time to start using real-world data from the TRIO Network - (11/20/17)
Access to HCV Care for 31,154 Patients in the United States Real-world experience from the TRIO Network - (11/20/17)
Summary
° An 8-week course of LDV/SOF achieved 100% SVR12 in a trial of HIV-infected men with acute genotype 1 or 4 HCV infection, regardless of baseline HCV RNA. No renal toxicity was noted in participants on boosted TDF-inclusive HIV regimens. These data support an 8-week duration of LDV/SOF in the treatment of acute HCV infection in HIV-infected persons.
° High and recently (2008-2015) increasing HCV incidence was reported among HIV+ MSM in San Diego. MSM with a history of methamphetamine use and/or IDU were at particularly high risk for HCV acquisition. After SVR HIV+MSM remain at elevated HCV reinfection risk.
° Despite increases over calendar time, HCV incidence among HIV+ MSM, could have been 50% higher in Berlin in 2016 without existing levels of treatment.
° In the Co-STAR study the reinfection rate overall was 2.3/100 person-years, with a persistent reinfection rate of 1.6/100 person-years. Reinfection rates were higher in persons with reported intravenous drug use. Harm reduction interventions remain crucial also after obtaining SVR.
° High SVR12 rates from clinical phase III studies could be confirmed in GECCO in GT3 patients treated with SOF/VEL +/- RBV for 12 weeks. The addition of RBV to SOF/VEL in cirrhotic GT3 patients did not seem to have further benefit. Baseline NS5A RAS with a possible impact on the effectiveness of VEL (A30K or Y93H detected in 10 patients) did not influence the chance for SVR12.
° G/P achieved high efficacy in treatment-naïve patients with HCV GT3, regardless of cirrhosis status (8 weeks: 95% ITT SVR12 in patients without cirrhosis and 12 weeks: 97% ITT SVR12 in patients with cirrhosis). Extending duration from 8 to 12 weeks had no impact on efficacy for GT3 patients without cirrhosis. G/P was well-tolerated with a safety profile comparable in patients with or without cirrhosis. Baseline RAS had no impact on SVR12 rates.
° G/P for 12 and 16 weeks achieved high efficacy in patients with HCV GT 1-6 infection and compensated cirrhosis, including those with renal impairment (96% (ITT) and 97% (mITT) overall SVR12 rates).
° EBR/GZR was highly effective in HCV-infected patients from the Veterans Affair System with moderate to severe CKD with SVR (PP) of 96.3% overall. SVR rates were high across select patient subgroups in a large clinical setting.
° DAA therapy is associated with a significant decrease in mortality risk
in patients with decompensated HCV cirrhosis, by as much as 60%
within the first year of therapy.
° Almost a quarter of 144 coinfected PWIDs did not initiate HCV treatment despite access to expert clinicians and LDV/SOF at no cost in the CHAMPS study. The study results suggest peer-mentors may effectively increase HCV linkage and treatment.
° The single tablet regimen of SOF/VEL/VOX for 12 weeks was safe, well tolerated, and highly effective for patients who had previously failed NS5A inhibitor-containing DAA regimens.
° The two-drug combination of RZR 180 mg + UPR 450 mg led to an overall SVR12 rate of 90%. Higher relapse rates were observed in GT3 patients but also in GT1a patients in particular if they had the Y93H RAS at baseline. The company, which has evaluated this combination, has announced their termination of this DAA development program.
° The combination of AL-335 800 mg, ODV 25 mg, and SMV 75 mg once daily (QD) (also known as JNJ-4178) achieved good efficacy with SVR12 rates above 97%. However, the developing company nonetheless decided to terminate their further DAA development program because of the already available very effective existing DAA combinations. Maybe the lack of activity for GT3 as well as the observed relapses in GT2c patients have further influenced this decision.
° In the Veterans cohort patients with advanced chronic liver disease (ACLD) and non-ACLD patients achieving SVR after DAA treatment had significantly lower all-cause mortality and lower incident HCC rates than those without SVR, regardless of genotype.
References
1. Lawitz E et al. C-BREEZE 2: Efficacy and Safety of a Two-Drug Direct-Acting Antiviral Agent (DAA) Regimen Ruzasvir 180 mg and Uprifosbuvir 450 mg for 12 Weeks in Adults With Chronic Hepatitis C Virus (HCV) Genotype (GT)1, 2, 3, 4, or 6. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 61
2. Zeuzem S et al. Evaluation of the efficacy and tolerability of JNJ-4178 (AL-335, odalasvir, and simeprevir) in hepatitis C virus-infected patients without cirrhosis: The Phase IIb OMEGA-1 study. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 65
3. Günter R et al. High SVR-12 rates in patients with and without opioid substitution therapy (OST) in daily practice after SOF-based therapy regimens despite cannabis and alcohol consumption in both groups. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1073
4. Ward K et al. Randomized controlled trial of cash incentives or peer mentors to improve HCV linkage and treatment among HIV/HCV coinfected persons who inject drugs: The CHAMPS Study. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 76
5. Lawitz E et al. Safety and Efficacy of Treatment with Once-Daily Ledipasvir/Sofosbuvir (90/400 mg) for 12 Weeks in Genotype 1 HCV-Infected Patients with Severe Renal Impairment. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1587
6. Kramer JR et al. Effectiveness of Elbasvir/Grazoprevir in patients with Chronic Hepatitis C and Chronic Kidney Disease: Results from the Veterans Affairs system. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1113
7. Chasca DM et al. Impact of HCV Treatment before vs. after Renal Transplantation on Time from Listing to Transplantation: A Multi-Center Study. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 197
8. Argawal K et al. Sofosbuvir/Velpatasvir for 12 Weeks in Genotype 1-4 HCV-Infected Liver Transplant Recipients. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1069
9. Christensen S et al. Do resistance-associated substitutions (RAS) or Ribavirin (RBV) use influence treatment success of Sofosbuvir (SOF)/Velpatasvir (VEL) in chronic hepatitis C genotype 3 (GT 3) infection? - Results from the GErman hepatitis C COhort (GECCO). 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 63
10. Flamm SL et al. Efficacy and Safety of Glecaprevir/Pibrentasvir for 8 or 12 Weeks in Treatment-Naïve Patients with Chronic HCV Genotype 3: An Integrated Phase 2/3 Analysis 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 62
11. Gane E et al. Efficacy, Safety, and Pharmacokinetics of Glecaprevir/Pibrentasvir in Adults With Chronic Genotype 1-6 Hepatitis C Virus Infection and Compensated Cirrhosis: An Integrated Analysis. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 74
12. Abergal A et al. High Efficacy and Safety of the combination HCV Regimen Grazoprevir and Elbasvir for 8 Weeks in Treatment-Naive, non-severe fibrosis HCV GT1b-Infected Patients: Interim Results of the STREAGER study. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract LB-5
13. Bourlière M et al. SOF/VEL/VOX for 12 Weeks in NS5A-Inhibitor Experienced HCV-Infected Patients: Results of the Deferred Treatment Group in the Phase 3 POLARIS-1Study. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1178
14. Vermehren J et al. Failure to multiple Interferon-free direct antiviral therapies: could chronic hepatitis C become incurable? 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1605
15. Naggie S et al.: 100% SVR with 8 weeks of ledipasvir/sofosbuvir in HIV-infected Men with Acute HCV Infection: Results from the SWIFT-C Trial (Sofosbuvir-Containing Regimens without Interferon for Treatment of Acute HCV in HIV-1 Infected Individuals). 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 196
16. Chaillon A et al. Increasing Primary Incidence of Hepatitis C Among HIV-Infected Men Who Have Sex with Men in San Diego; a Pooled Analysis of Two Large Clinics from 2000-2015. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 124
17. Dore G et al. Hepatitis C Virus (HCV) Reinfection and Injecting Risk Behavior Following Elbasvir (EBR)/Grazoprevir (GZR) Treatment in Participants on Opiate Agonist Therapy (OAT): Co-STAR Part B. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 195
18. Nakagawa M et al. Evaluation of an early occurrence and recurrence of hepatocellular carcinoma in chronic hepatitis C patients treated with All-Oral DAAs - propensity score-matched analysis of a prospective database. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 66
19. Degasperi E et al. Low rates of de novo or recurrent hepatocellular carcinoma in HCV cirrhotic patients treated with direct-acting antivirals (DAAs): a single-center experience. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 77
20. Backus LI et al. Impact of Sustained Virologic Response with Direct-Acting Antiviral Treatment on Mortality and Hepatocellular Carcinoma. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 78
21. Delmonte RJ et al. Treatment of HCV with direct acting antivirals significantly reduces liver-related hospitalizations in patients with cirrhosis. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 75
22. Kim WR et al. Survival benefit of direct-acting antiviral therapy in patients
with decompensated cirrhosis. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract LB-27
23. Rockstroh JK, et al. Ledipasvir-sofosbuvir for 6 weeks to treat acute hepatitis C virus genotype 1 or 4 infection in patients with HIV coinfection: an open-label, single-arm trial. Lancet Gastroenterol Hepatol. 2017;2:347-353.
24. Naggie S et al.: AIDS Clinical Trials Group (ACTG) A5327 Study Team. Sofosbuvir Plus Ribavirin Without Interferon for Treatment of Acute Hepatitis C Virus Infection in HIV-1-Infected Individuals: SWIFT-C. Clin Infect Dis. 2017;64:1035-1042.
25. Martin N et al. Can HCV be eliminated among HIV-infected MSM in Berlin? Modeling a setting with increasing incidence and high treatment rates. 68th Annual Meeting of the American Association for the Study of Liver diseases, October 20-24, 2017, Washington DC, USA; abstract 1050
26. www.eacsguidelines.org
27. Dore G et al. C-EDGE CO-STAR Study Group. Elbasvir-Grazoprevir to Treat Hepatitis C Virus Infection in Persons Receiving Opioid Agonist Therapy: A Randomized Trial. Ann Intern Med. 2016;165:625-634.
28. Foster GR et al., ASTRAL-2 Investigators; ASTRAL-3 Investigators. Sofosbuvir and Velpatasvir for HCV Genotype 2 and 3 Infection. N Engl J Med 2015; 373:2608-2617
29. Kondili LA et al. Real life clinical outcomes in DAA-treated and untreated patients with HCV advanced liver disease. Interim one year data from the ongoing PITER cohort. 68th AASLD; Washington, DC; October 20-24, 2017; Abst. 1020
30. Roth D et al. Grazoprevir plus elbasvir in treatment-naive and treatment-experienced patients with hepatitis C virus genotype 1 infection and stage 4-5 chronic kidney disease (the C-SURFER study): a combination phase 3 study. Lancet 2015;386(10003):1537-45.
31. Sylvestre D, et al. No Evidence of 1-Year Reinfection after Treating HCV at a Methadone Program. 68th AASLD; Washington, DC; October 20-24, 2017; Abst. LB-18
32. Bourliere M et al. POLARIS-1 and POLARIS-4 Investigators. Sofosbuvir, Velpatasvir, and Voxilaprevir for Previously Treated HCV Infection. N Engl J Med. 2017;376:2134-2146.
33. Lawitz E et al. C-BREEZE 1: Efficacy and Safety of Ruzasvir 60 mg Plus Uprifosbuvir 450 mg for 12 Weeks in Adults With Chronic Hepatitis C Virus (HCV) Genotype (GT)1, 2, 3, 4, or 6 Infection. 68th AASLD; Washington, DC; October 20-24, 2017; Abst. 1175
34. Zobair M et al. Cure denied and cure delayed in Chronic Hepatitis C; monitoring non-start rates and increased time to start using real-world data from the TRIO Network. 68th AASLD; Washington, DC; October 20-24, 2017; Abst. 1115
|
| |
|
 |
 |
|
|