 |
 |
 |
| |
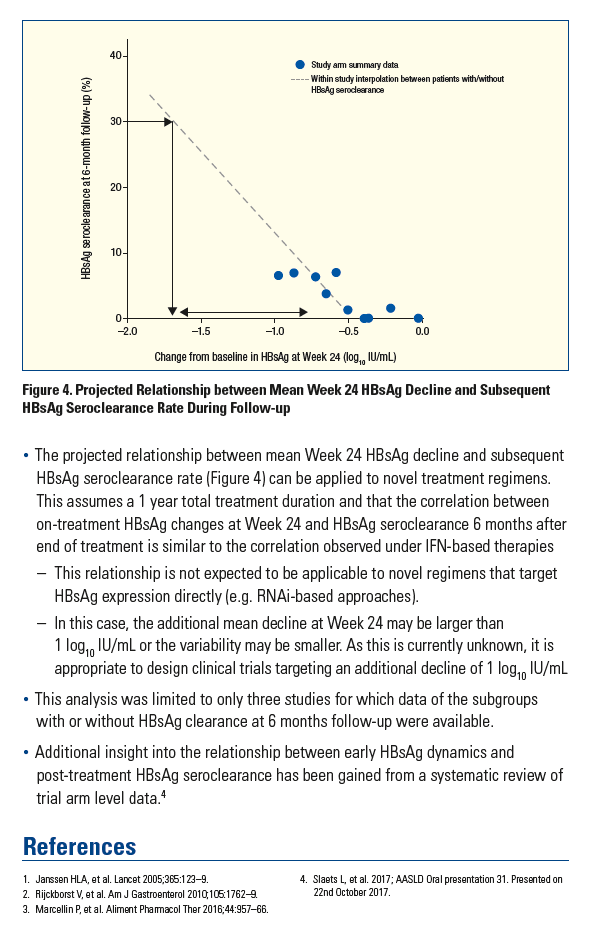
Mean Week 24 HBsAg Declines Predict Subsequent Rate of HBsAg Seroclearance and Could Be a Valuable Endpoint for Early Development HBV Trials
|
| |
| |
AASLD: Safety, Tolerability, Pharmacokinetics, and Antiviral Activity of JNJ-56136379, a Novel HBV Capsid Assembly Modulator, in Non-cirrhotic, Treatment-naïve Patients with Chronic Hepatitis B. - (11/01/17)
AASLD/2016: Capsid assembly modulator JNJ-56136379 prevents de novo infection of primary human hepatocytes with hepatitis B virus - (11/22/16)
Safety, tolerability and pharmacokinetics of JNJ-56136379, a novel HBV capsid assembly modulator, in healthy subjects - (11/22/16)
Reported by Jules Levin
AASLD, Washington DC, USA, October 20-24 2017
Filip De Ridder1, Milan J Sonneveld2, Oliver Lenz1, Harry LA Janssen3, Willem Talloen1, Bettina E Hansen2-4
1Janssen Research & Development, Beerse, Belgium; 2Department of Gastroenterology and Hepatology, Erasmus MC University Medical Center, Rotterdam, The Netherlands; 3Toronto Centre for Liver disease, Francis Family Liver Clinic, Toronto General Hospital, Toronto, ON, Canada; 4IHPME, University of Toronto, Toronto, ON, Canada


|
| |
|
 |
 |
|
|