 |
 |
 |
| |
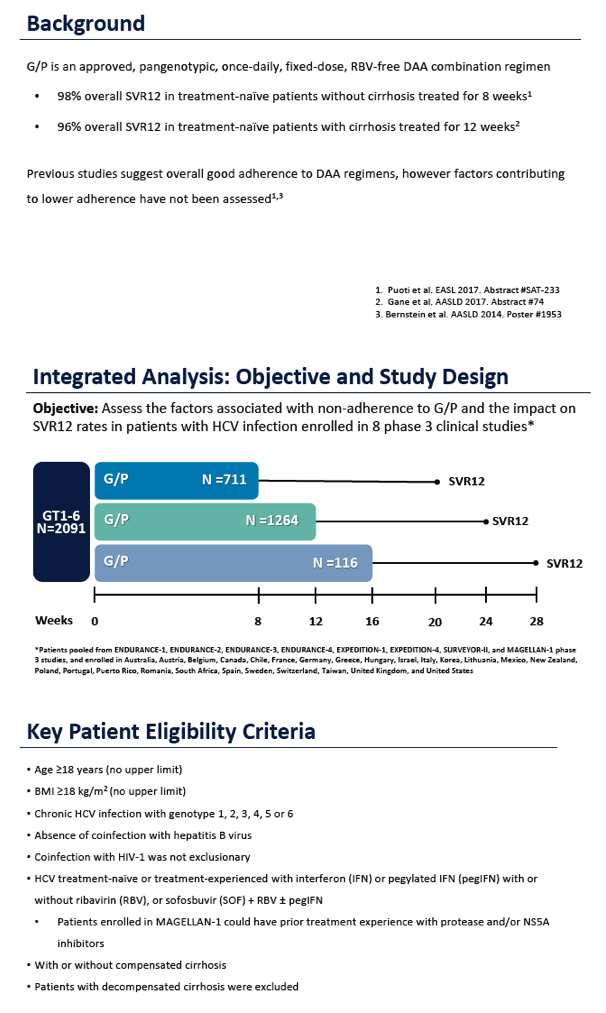
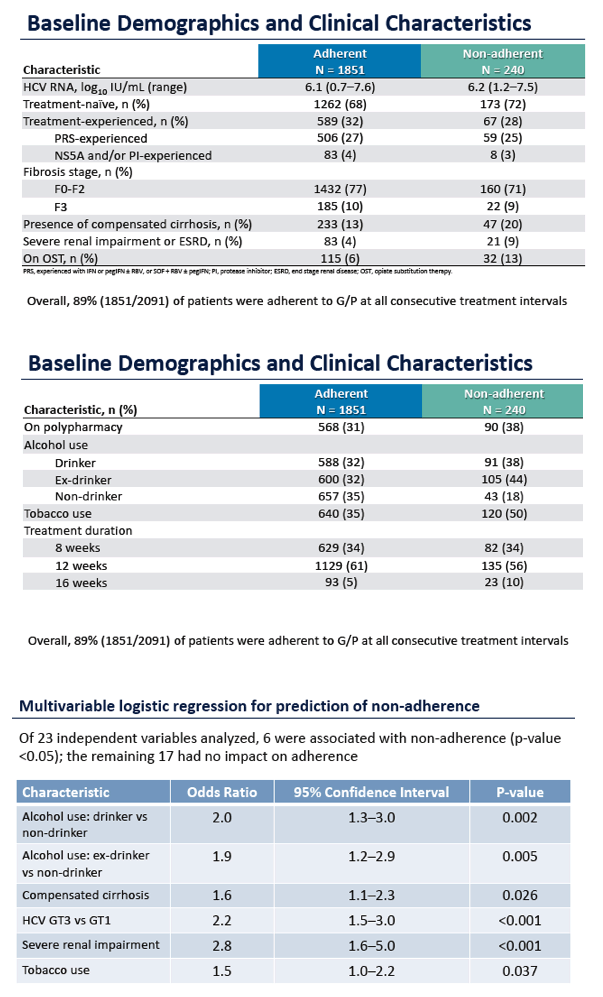
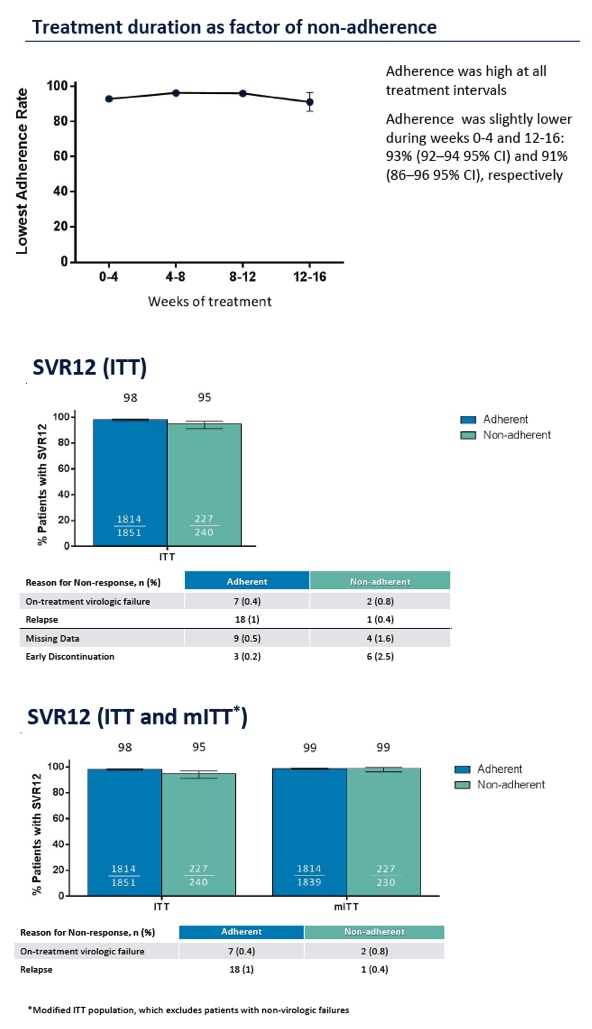
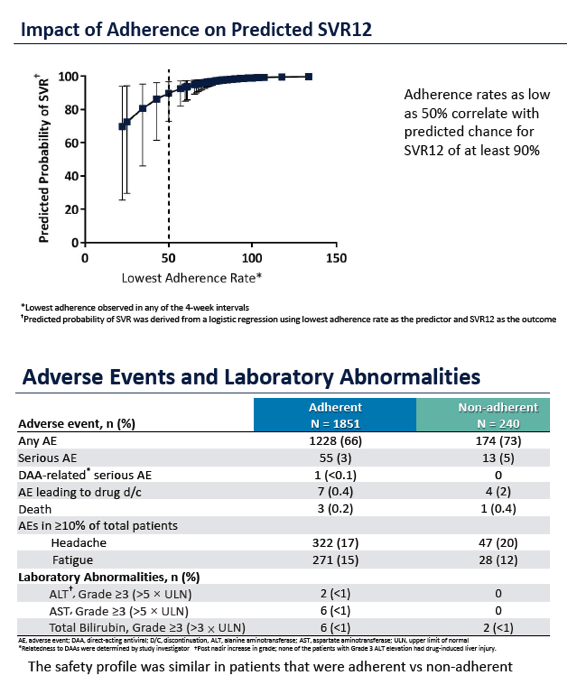
Adherence to Pangenotypic Glecaprevir/Pibrentasvir Treatment and SVR12 in HCV-infected Patients: An Integrated Analysis of the Phase 2/3 Clinical Trial Program
|
| |
| |
Reported by Jules Levin
AASLD: The Liver Meeting 2017, October 20-24, 2017, Washington, DC
Brown A1, Welzel TM2, Conway B3, Negro F4, Bräu N5, Grebely J6, Puoti M7, Aghemo A8, Kleine H9, Pugatch D9, Mensa F9, Chen YJ9, Lei Y9, Lawitz E10
1Imperial College Healthcare, London, United Kingdom; 2J. W. Goethe University Hospital, Frankfurt, Germany; 3Vancouver Infectious Diseases Center, Vancouver, Canada; 4Divisions of Gastroenterology and Hepatology and Clinical Pathology, University Hospital, Geneva, Switzerland; 5James J. Peters VA Medical Center, Bronx, NY, United States; 6The Kirby Institute, UNSW Sydney, Sydney, Australia; 7Department of Infectious Diseases, AO Niguarda Ca' Granda, Milan, Italy; 8Humanitas Clinical and Research Center, Milan, Italy; 9AbbVie, North Chicago, IL, United States; 10Texas Liver Institute, University of Texas Health, San Antonio, TX, United States

*Patients should take G/P as directed; it is important not to miss or skip doses. If a dose is missed and it is <18 hours from the usual time taken, take the dose as soon as possible. If a dose is missed and it is >18 hours from the usual time taken, do not to take the missed dose and take the next dose at the usual time.


|
| |
|
 |
 |
|
|