 |
 |
 |
| |
Cost-effectiveness of HCV Screening and Linkage in MMT: Relevance of HIV Co-infection
|
| |
| |
CROI 2017 Feb 14-17 Seattle, WA
Bruce R. Schackman,1 Jared A. Leff,1Jake R. Morgan,2Sarah Gutkind,1Czarina N. Behrends,1Kevin L. Delucchi,3Courtney McKnight,4David C. Perlman,4Carmen L. Masson,3Benjamin P. Linas2
1Department of Healthcare Policy & Research, Weill Cornell Medical College, New York, NY, USA, 2Boston Medical Center, Boston, MA, 3Department of Psychiatry, UCSF School of Medicine, San Francisco, CA, USA; 4Icahn School of Medicine at Mount Sinai, New York, NY, USA

Abstract Body:
We evaluated the cost-effectiveness of an HCV screening and linkage to care intervention in US methadone maintenance treatment (MMT) patients using data from a randomized trial conducted in New York City and San Francisco.
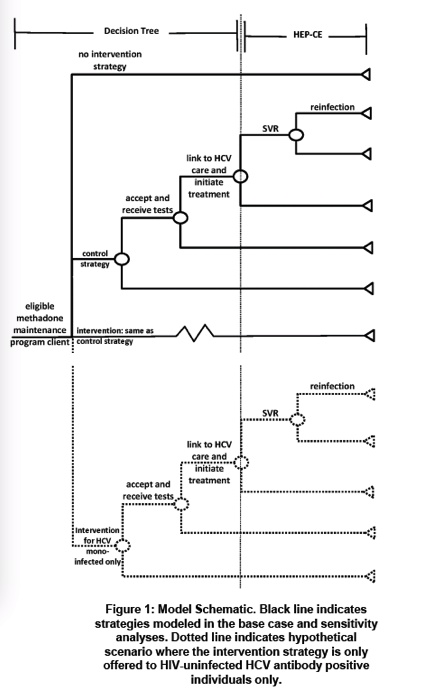
We used a decision analytic model to compare the cost-effectiveness of 4 strategies: 1) no intervention; 2) HCV screening and education (control); 3) HCV screening and education for all and care coordination for all HCV-infected 4) HCV screening and education for all with care coordination only for HCV mono-infected patients (to explore trial results indicating that HIV co-infected participants linked through other systems of care). Trial data include population characteristics (67% male, mean age 48, 50% HCV mono-infected, 8% HCV/HIV co-infected) and linkage rates for HCV mono-infected (34% control, 68% intervention) and HCV/HIV co-infected (79% control, 87% intervention) individuals. Data from published sources include treatment efficacy and HCV re-infection risk. Clinical outcomes include proportions with chronic HCV linked and achieving SVR. We projected quality-adjusted life expectancy (QALYs) and lifetime medical costs using an established model of HCV (HEP-CE). Incremental cost-effectiveness ratios (ICERs) are in 2015 US$/QALY discounted 3% annually.
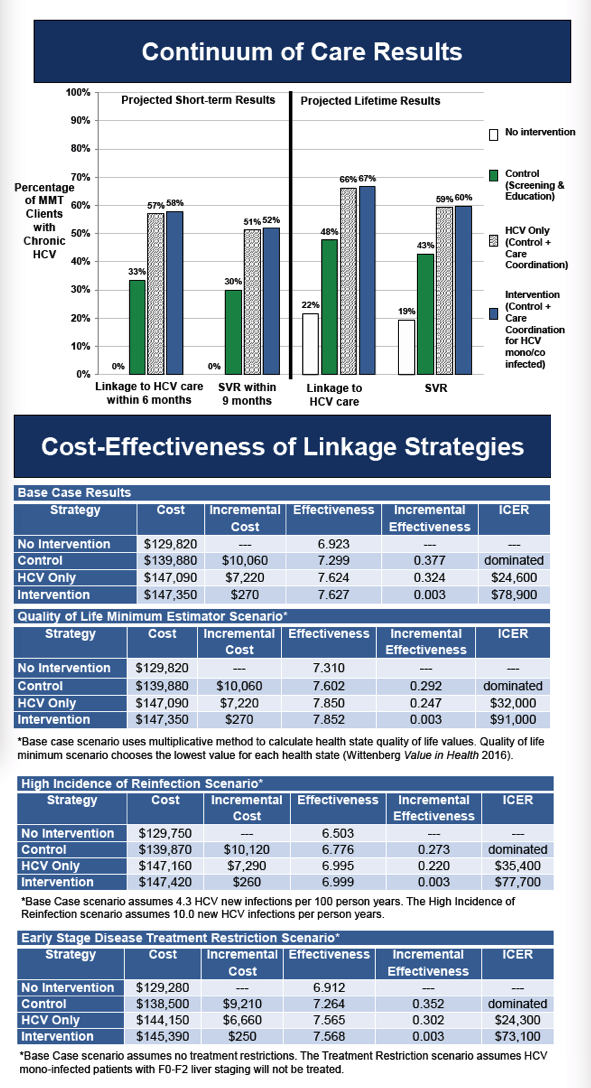
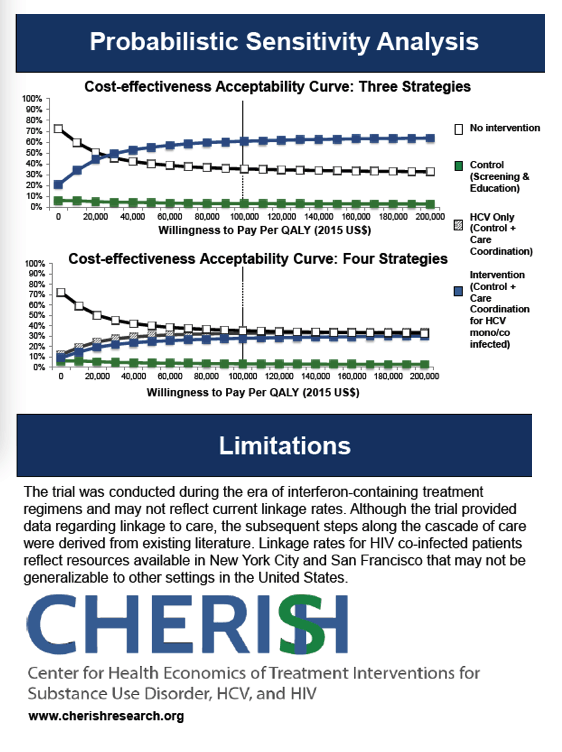
The control strategy resulted in a projected 34% linking to care within 6 months and 30% achieving SVR (Figure). The cost was $156/person screened. HCV care coordination for HCV mono-infected patients resulted in 57% linkage and 51% achieving SVR at a cost of $470/person screened. Care coordination for all increased projected linkage to 58% and SVR to 52% at a cost of $514/person screened. The care coordination for all strategy was more efficient than (dominated) the care coordination for HCV-mono infected patients only strategy and the control strategy, and had an ICER of $21,600/QALY compared to no intervention. In sensitivity analyses varying the risk of re-infection, results were not highly sensitive to increasing the risk of reinfection (resulting in triple the number of reinfections) as long as re-infected patients were eligible for retreatment (resulting in an additional 8 treatments per 1000 patients treated).
HCV care coordination interventions that include screening, education and linkage to care in MMT settings are likely cost-effective at a conventional $100,000/QALY threshold for both HCV mono-infected and HIV co-infected patients.

|
| |
|
 |
 |
|
|